Week 3 HW: Lab Automation
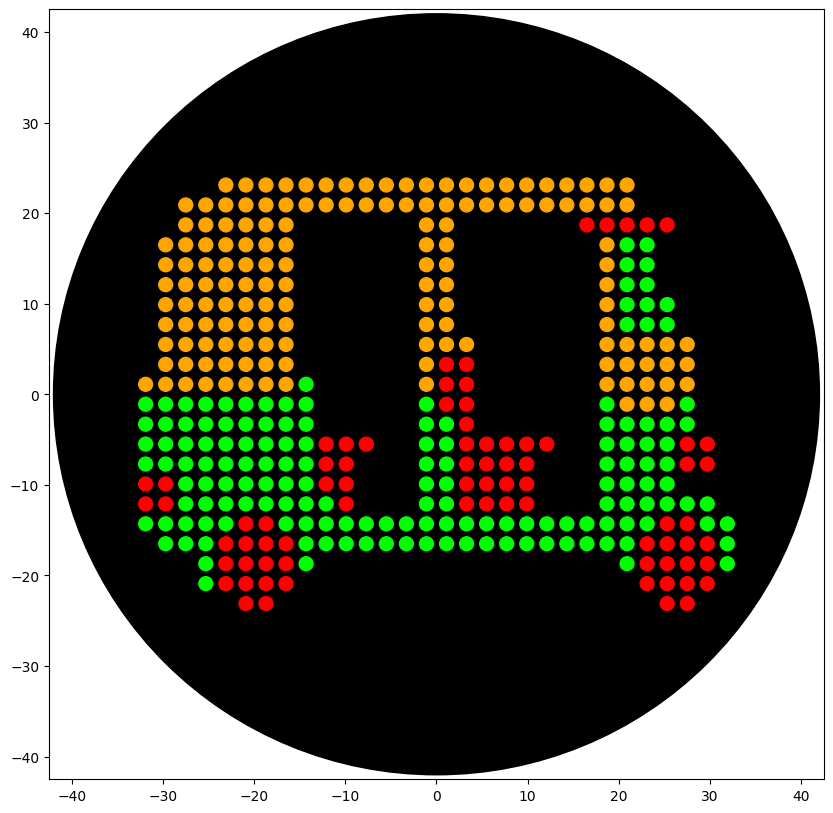
Part 1: Python Script Opentron Artwork
Abhishek Udawat Copy of HTGAA26 Opentrons Colab
Fluorescent TukTuk

Part 2: Post-Lab Questions
Question 1. Find and describe a published paper that utilizes the Opentrons or an automation tool to achieve novel biological applications.
- Since my interest in cell-free systems and biosensors, I found a relevant paper titled “Semi-automated Production of Cell-free Biosensors”
The Problem: Manually pipetting cell-free reactions is slow, inconsistent across different researchers, and difficult to scale for field use
Their Solution: The Authors used the Opentrons OT-2 to automate the assembly of flouride riboswitch biosensors
The Novelty: They didn’t just automate the liquid moving, they optimized the robot’s physical parameters (like blowout height and dispense speed) to handle the viscosity of cell-free extracts. This allowed them to produce 384 consistent, functional sensors in 30 minutes- something that would take a researcher much longer and with higher risk of error.
The Result: They proved that low-cost robotics can produce “shelf-stable” diagnostics that perform nearly as well as those made by expert humans, making it possible to manufacture sensors for environmental toxins anywhere in the world.
Question 2: What do you intend to do with automation tools for your final project?
- Project Context: My project, DermLogic, invovles creating a smart biopolymer hydrogel patch that utilizes cell-free genetic circuits to detect and treat cutaneous HPV.
- Automation Plan:
I intend to use the Opentons OT-2 to standardize and scale the production of these “Living” patches. I envision that the primary challenge with DermLogic will be ensuring the precise ratio of cell-free extracts, DNA logic gates, and biopolymer precursors (hydrogel) to maintain therapeutic consistency.
Here are some lessons taken from the above paper:
Viscosity-Optimized Liquid Handling: Following the methodology in Brown et al. (2024), I will program specific robotic parameters to handle the high viscosity of the hydrogel-extract mixture. This includes tuning the aspirate/dispense rates and implementing touch-tip/blowout sequences to prevent the “bubbling” that often occurs during manual pipetting of cell-free systems.
High-Throughput Logic Testing: I will use automation to screen an array of genetic circuits designed to detect HPV oncoproteins. The robot will facilitate a “Master Mix” approach, where the base cell-free machinery is distributed across 96-well plate, followed by the addition of unique DNA constructs. This allows me to test hundreds of logic-gate variants in a single run to see which provides the sharpest “ON” trigger in the presence of HPV markers.
Automated Lyophilization Prep: To meet governance goals of a “cell-free mandate” for safety proposed in week 1’s assignment, i will use the Opentrons to dispense the final reactions into patch molds before they are frozen and lyophilized. Automation ensures that every patch has a uniform concentration of the therapeutic peptide, which is critical for clinical safety and efficacy.
- Pseudocode for DermLogic Patch Assembly:
Final Project Ideas Slide