Week 4 HW: Protien Design Part I

HW Questions
Assuming that the meat is ~20% protein, thus for 500 g meat ≈ 100 g protein. Average amino acid ≈ 100 Da = 100 g/mol, we can find the Moles of amino acids by ≈ 100 g (protein) ÷ 100 g/mol = 1 mol. According to Avogadro’s Number of molecules ≈ 6.02 × 10²³ amino acids in 1 mol.
Why eating beef doesn’t turn me into a cow? Proteins are digested into amino acids in the stomach and intestine and broken down before absorption, then transferred in the blood and reassembled in each cell based on our DNA and mRNA commands into different proteins. food’s structure is always broken down, that’s why insulin can’t be taken orally.
Why only 20 natural amino acids exist naturally? Many reasons, including Evolution induced minimal but sufficient set that already cover diverse properties: polar, nonpolar, charged, aromatic. Also, Genetic code constraints (triplet codons) stabilized this set and prevented increasing error rates without major benefit.
Non-natural amino acids? Yes, we can design them. For example: Bulky hydrophobic amino acid: extended aromatic rings with a side chain of 3 phenol substitutions. Or Metal-binding amino acid: includes extra histidine-like rings that bind to metals and increase stability or provide a catalytic function.
Origin of amino acids before life • Formed via prebiotic chemistry (e.g., Miller–Urey-type reactions) • Sources: lightning, volcanic gases, meteorites • Found in space (e.g., meteorites like Murchison meteorite) • Accumulated before enzymes existed
Why most helices are right-handed Because proteins use L-amino acids, whose stereochemistry favors right-handed packing with low steric effects .
Why β-sheets aggregate • Backbone forms strong hydrogen bonds • Flat structure allows stacking
Why amyloid diseases come form β-sheets? Misfolded proteins reorganize into stable β-sheet-rich fibrils Examples include Alzheimer’s disease Can they be used as materials? Yes, Extremely strong (like nanofibers) or Used in biomaterials, nanotechnology, and tissue scaffolds
Designing a β-sheet motif Key principles: we Alternate hydrophobic (H) and polar (P) residues, Example pattern: H–P–H–P–H–P–H–P. The machanisims were a Hydrophobic side chains align on one face that drives stacking, Adding turns can happen with a Gly–Pro to stabilize sheet folding . The Result: ordered, self-assembling β-sheet structure.
PART A
I choose lactone synthase, an enzyme responsible for the synthesis of compounds containing lactone which are bio adhesive, protective and good smelling.
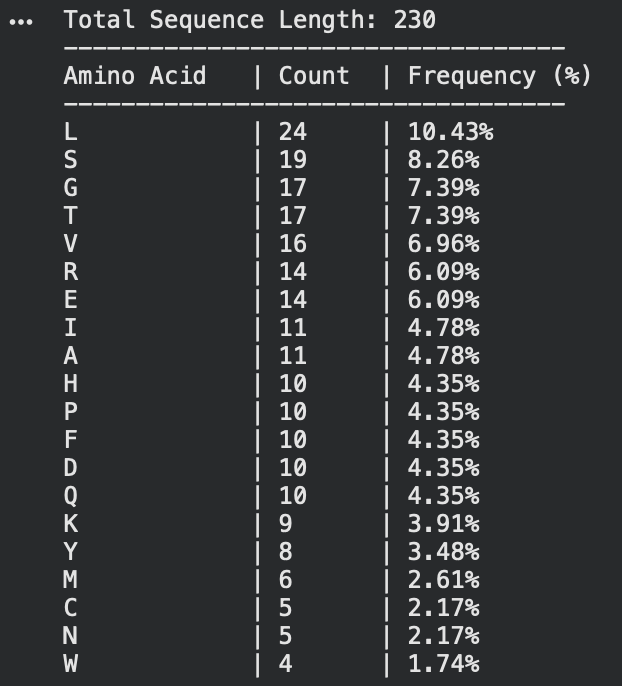
2. Its 230 aa, the most frequent one is Leucine. like shown below
 3. there was around 250 homologue sequences there for it.
4. Transferase family of finctionality.
3. there was around 250 homologue sequences there for it.
4. Transferase family of finctionality.
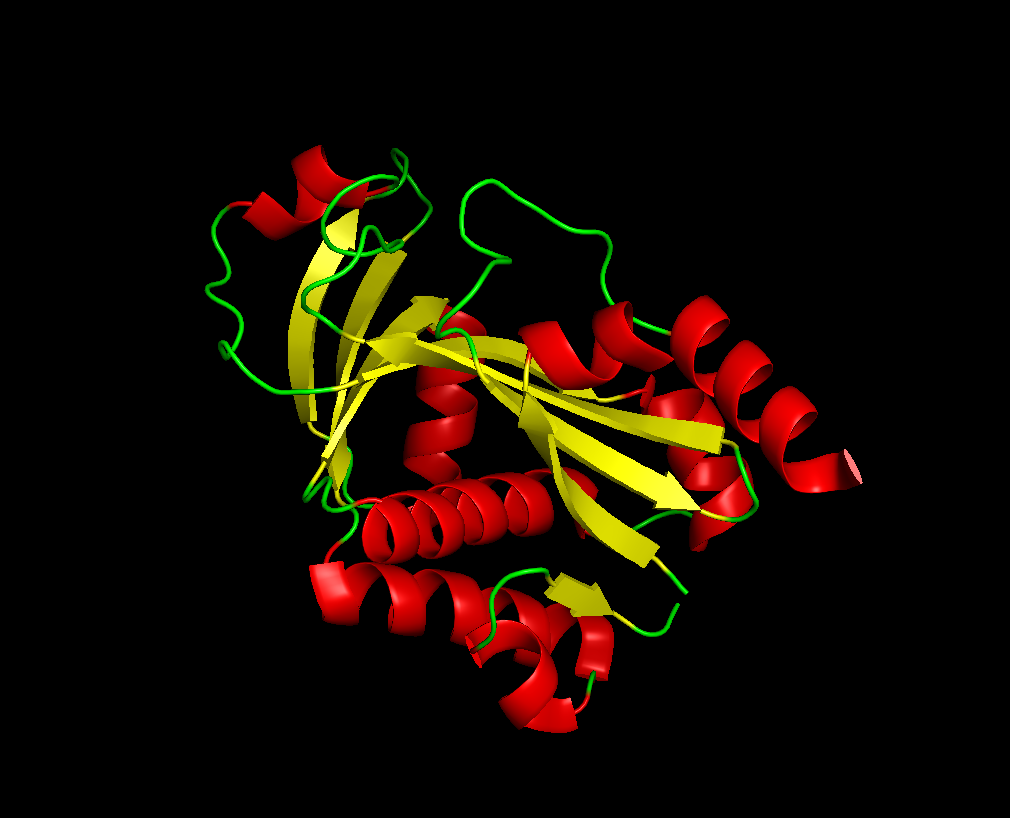
Identifying the structure page of the protein in RCSB!
well, There is no other molecules in the protein. and It was solved in 2002, with a good resolution of 1.8Å It is an autoinducer synthase family.
after the varoius types of visualization of my protien, I acquired the following informations: Total main 5 alpha helix and 3 mini, while the beta sheet were 7 large and one mini. So the name of helix and sheets, with sheets larger. The protein has many hydrophobic and hydrophilic properties, the hydrophilic amino acids are more oriented towards the outer layers of the protein. There was around 4 cavities that might work as binding pockets
