Conceptual questions
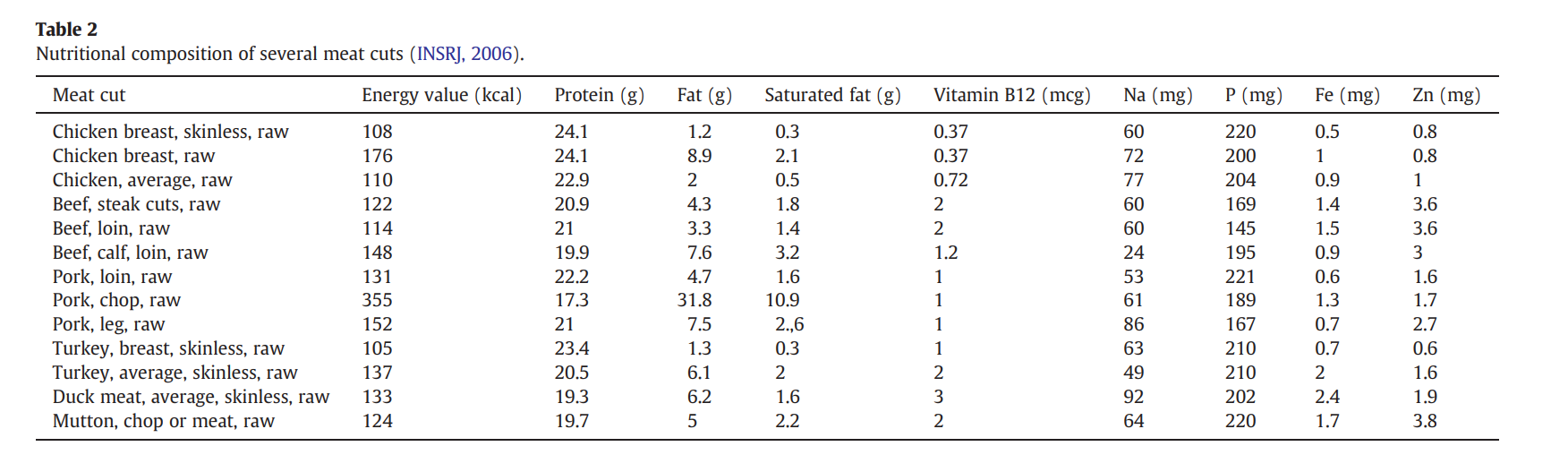
How many molecules of amino acids do you take with a piece of 500 grams of meat? (on average an amino acid is ~100 Daltons)
A: Averaging out the nutritional composition of common meats (like the data from Pereira & Vicente, 2013), meat is about 21.25% protein by weight.If you eat a 500-gram steak, you are consuming roughly 106.25 grams of pure protein. Since 1 Da is exactly 1 g/mol, an average amino acid weighing 100 Da has a molar mass of 100 g/mol.By dividing your total protein (106.25 g) by the average amino acid mass (100 g/mol), we get 1.0625 moles of amino acids. Multiply that by Avogadro’s number 6.022e+23, and you get approximately 6.399e+23 molecules of amino acids

Why do humans eat beef but do not become a cow, eat fish but do not become fish?
A: Because exogenous DNA is either degraded before consumption or degraded by our own enzymes during the digestion process, it cannot be processed to alter our cells. So, the only materials our body takes are the disassembled amino acids; then, it rebuilds them into human proteins, following our human genome.
Why are there only 20 natural amino acids?
A: Certainly, there are only 20 amino acids in Choanoflagellatea, and this set of amino acids provides just enough chemical diversity (acidic, basic, hydrophobic, and hydrophilic shapes) to build complex and versatile proteins. However, I believe it’s a little bit biased to say they are the only “natural” ones, because there are also species of methanogenic archaea that use other amino acids, like selenocysteine and pyrrolysine.
Can you make other non-natural amino acids? Design some new amino acids.
A: Yes, it’s possible to synthesize new amino acids with synthetic biology or organic chemistry. You could also find them as non-canonical amino acids, like 2-azatyrosine, 3-azatyrosine, or azetidine-2-carboxylic acid.
Where did amino acids come from before enzymes that make them, and before life started?
A: It’s believed that they formed completely abiotically, as was proven in the famous Miller-Urey experiment, where they showed that if you mix simple gases present on early Earth (like methane, ammonia, and water) and strike them with electricity (simulating lightning), amino acids form naturally and spontaneously.
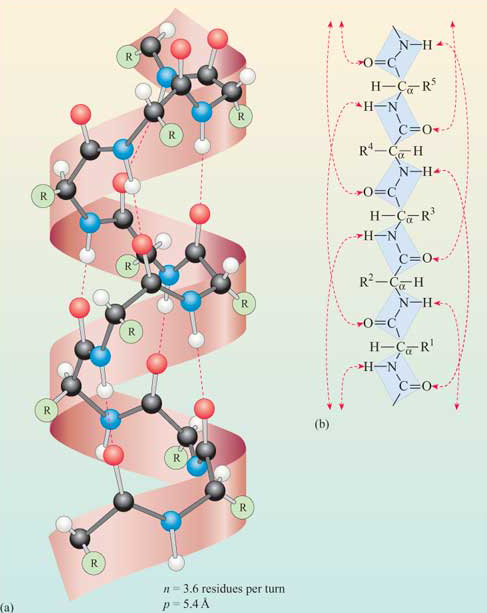
If you make an α-helix using D-amino acids, what handedness (right or left) would you expect?
A: Left. Because D-amino acids are enantiomers of our natural L-amino acids, the most energetically stable way for them to fold is also completely mirrored.
Why are most molecular helices right-handed?
A: This conformation is much more energetically favorable because it minimizes atomic collisions (steric hindrance) between side chains and the backbone of the L-amino acids.

Why do β-sheets tend to aggregate? What is the driving force for β-sheet aggregation?
A: Because the unpaired hydrogen bond donors and acceptors of a beta strand attain a highly stable conformation by bonding with another strand, reaching a lower energy state. The driving forces are hydrogen bonds and hydrophobic interactions.
Why do many amyloid diseases form β-sheets? Can you use amyloid β-sheets as materials?
A: Because amyloid diseases are characterized by protein denaturation or misfolding, these proteins expose hydrofobic regions in this process so the end up crashing ther strands together and “zippering up” into tightly packed cross-β-sheet structures. Yes, because of its stability and self-assembly capacity, we could use it as diverse materials: nanowires, hydrogel, scaffolds, etc.
References:
Pereira, P. M. de C. C., & Vicente, A. F. dos R. B. (2013). Meat nutritional composition and nutritive role in the human diet. Meat Science, 93(3), 586–592. https://doi.org/10.1016/j.meatsci.2012.09.018
4.2: Secondary structure and loops. (2018). Biology LibreTexts; Libretexts. https://bio.libretexts.org/Bookshelves/Biochemistry/Fundamentals_of_Biochemistry_(Jakubowski_and_Flatt)/01%3A_Unit_I-_Structure_and_Catalysis/04%3A_The_Three-Dimensional_Structure_of_Proteins/4.02%3A_Secondary_Structure_and_Loops