Week 7 HW: Genetic Circuits Part II
Part1: Intracellular Artificial Neural Networks
- What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions?
Traditional genetic circuits treat inputs as binary. This works for simple logic but breaks down when you need nuanced, graded decisions based on multiple continuous signals. Biology itself is almost never binary; cells exist on spectrums of gene expression and signalling intensity. IANNs overcome this by operating in the analog domain. An IANN computes a weighted sum of all inputs and applies a nonlinear activation function, exactly like an artificial neuron. The same molecular parts can be reused to implement completely different decision boundaries just by changing the weights, without engineering new biological parts from scratch. IANNs can also be stacked into multiple layers, enabling hierarchical computation that is completely impossible with single-layer Boolean circuits.
- Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
Application: multi-signal tumour detection A compelling use case is engineering a cancer-detecting IANN in CAR-T cells that triggers apoptosis only when multiple tumour markers are simultaneously present at the right levels, while ignoring healthy cells that express some markers at lower concentrations. Three inputs (HER2, MUC1, HIF-1a) drive promoters at strengths proportional to their concentration. Those promoters produce endoribonucleases whose expression encodes the weighted input combination. Layer 1 outputs Csy4, whose concentration reflects the weighted sum. In layer 2, a caspase gene carries a Csy4-recognition hairpin in its 5’ UTR. If Csy4 is below threshold, the hairpin is intact and the cell triggers apoptosis in the target. If Csy4 is high, it cleaves the mRNA and nothing happens. Limitations: the number of well-characterised orthogonal ERNs is small, capping practical input dimensionality. The system is also sensitive to transcriptional noise at low signal concentrations, and tuning promoter strengths reliably across cell types is difficult.
- Draw a diagram for an intracellular multilayer perceptron where layer 1 outputs an endoribonuclease that regulates a fluorescent protein output in layer 2. cover image
Part 2: Fungal Materials
- What are some examples of existing fungal materials and what are they used for? What are their advantages and disadvantages over traditional counterparts?
The most developed fungal material is mycelium composite, where filaments of fungi like Ganoderma are grown through agricultural waste substrates like corn stalks and grain husks. The mycelium binds these particles into a solid mass that can be moulded. Ecovative Design uses this for packaging foam replacing expanded polystyrene. Bolt Threads grows mycelium leather sheets (Mylo) used by fashion brands, and Mogu produces acoustic wall panels and floor tiles.
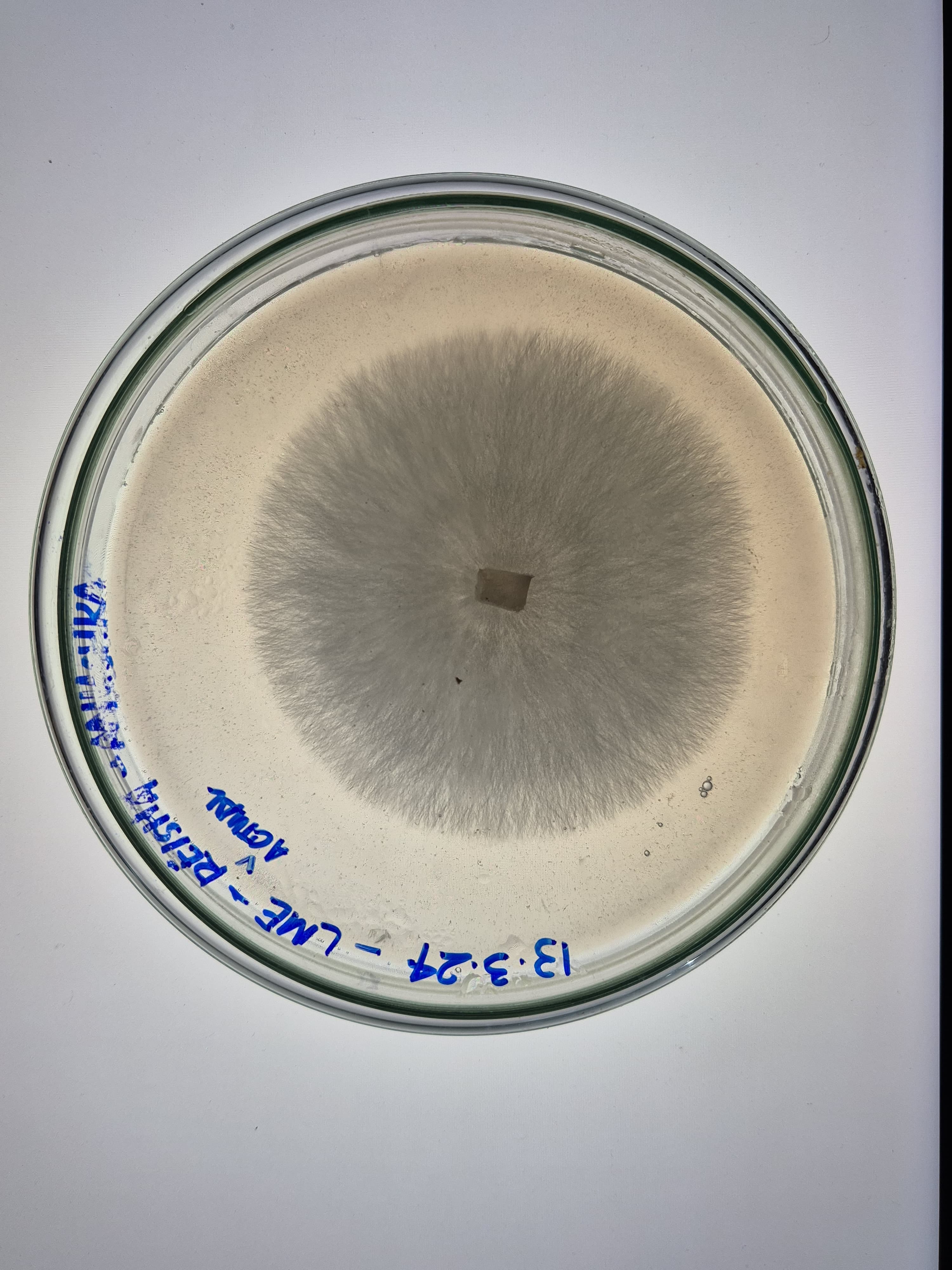
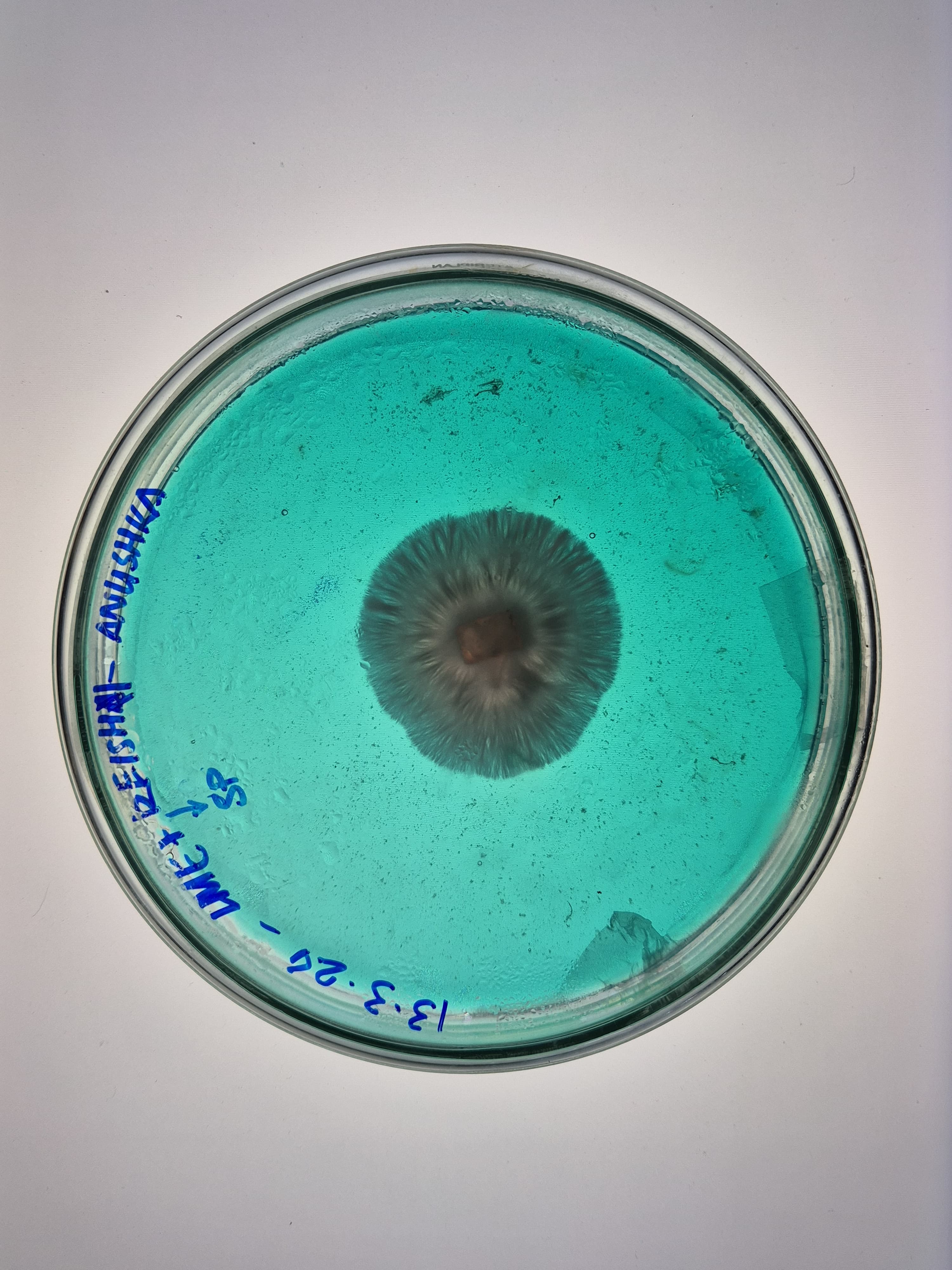
Here are some samples of mycelium I grew in 2024. The strains used are reishi and florida oyester mushroom strains.
Mycelium composites are biodegradable, grown on agricultural waste with no petrochemical inputs, naturally fire-resistant, and thermally insulating. Mycelium leather avoids tanning chemicals and animal welfare concerns, and unlike synthetic PU leather it does not shed microplastics. Disadvantages: the material must be heat-killed at the end of growth to stop fungal activity, causing dehydration and shrinkage that can warp precision shapes. Moisture resistance is limited without coatings. The growth process is sensitive to contamination. And mechanical properties like tensile strength still fall short of high-performance synthetics.
- What might you want to genetically engineer fungi to do and why? What are the advantages of doing synthetic biology in fungi as opposed to bacteria?
The most impactful engineering target is producing complex therapeutic glycoproteins that bacteria cannot make correctly. Beyond therapeutics, engineering mycelium to produce chitin fibres with controlled orientation or to express spider silk proteins could yield composites with dramatically improved mechanical properties. Fungi could also be engineered for mycoremediation, bioaccumulating heavy metals from contaminated soil. Fungi secrete proteins at rates 10 to 1000 times higher than bacteria, survive harsh conditions like low pH and desiccation, and build three-dimensional hyphal networks enabling solid-state fermentation without large water volumes.