Individual Final Project
#MycoTint: Stress Responsive Mycelium
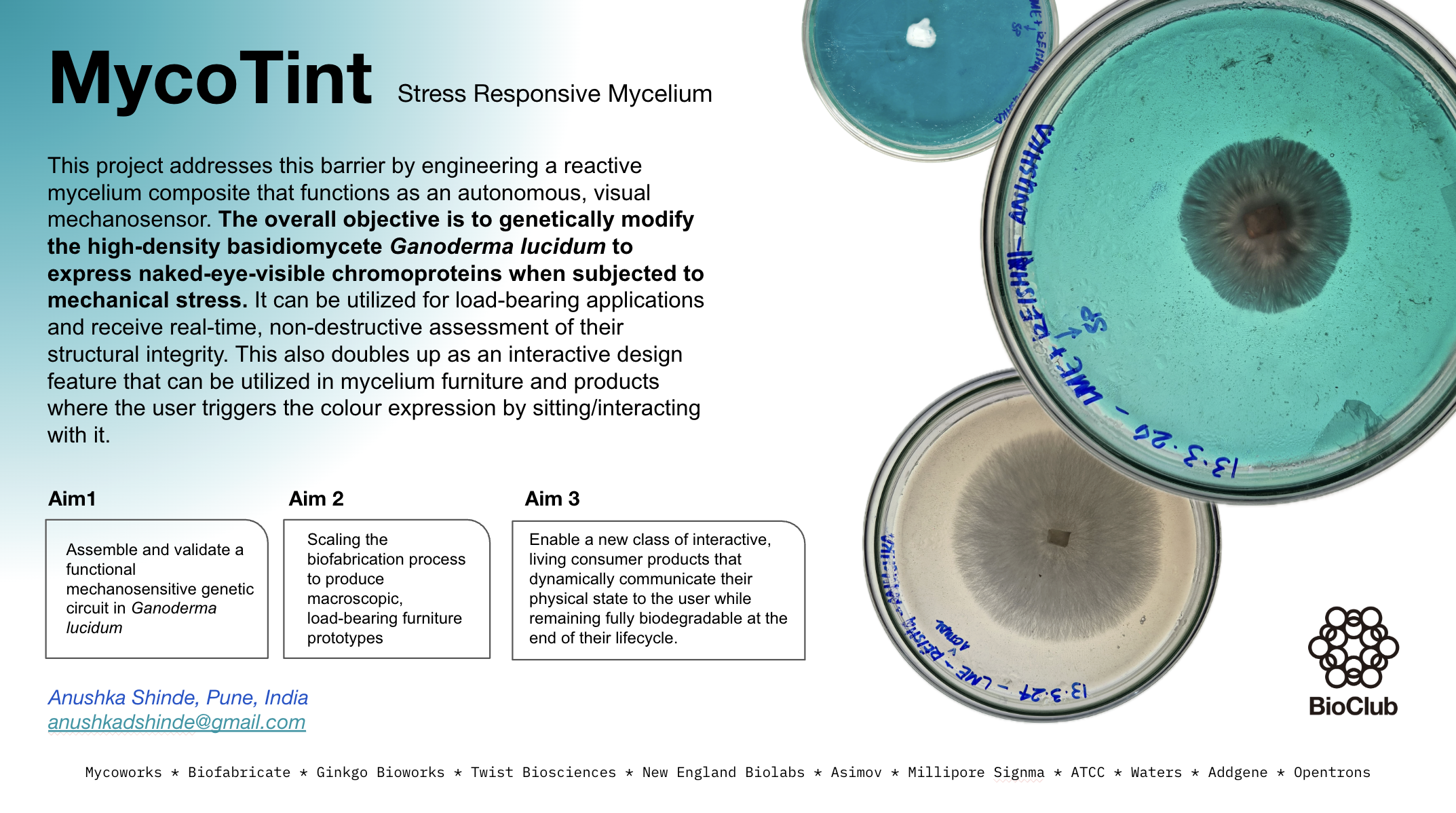

MycoTint is the conceptualization, genetic engineering, and biofabrication of reactive, living mycelium furniture capable of displaying visible color changes in response to mechanical stress. The objective is to engineer a mechanosensitive synthetic genetic circuit within the fungal host that actuates the expression of vivid, naked-eye-visible chromoproteins when specific points of the furniture are subjected to weight or impact. This document details the selection of the optimal biological chassis, the intricate design of the synthetic genetic circuit, fungal transformation protocols, specialized biofabrication and long-term viability maintenance strategies, practical feasibility assessments, alternative non-biological methods, and an analysis of precedent projects shaping the ELM landscape.
This project addresses this barrier by engineering a reactive mycelium composite that functions as an autonomous, visual mechanosensor. The overall objective is to genetically modify the high-density basidiomycete Ganoderma lucidum to express naked-eye-visible chromoproteins when subjected to mechanical stress. It can be utilized for load-bearing applications and receive real-time, non-destructive assessment of their structural integrity. This also doubles up as an interactive design feature that can be utilized in mycelium furniture and products where the user triggers the colour expression by sitting/interacting with it.
The different methods I see using to achieve this result are:
Mechanosensitive Genetic Circuits: Filamentous fungi like Ganoderma lucidum can be genetically engineered to hijack their innate cell wall integrity pathway, triggering the biological expression of visible anthozoan chromoproteins specifically when mechanical pressure is applied.
Bacterial Co-cultivation: Structural fungal mycelium can be grown alongside genetically engineered bacterial consortia that act as localized sensors, detecting environmental or physical cues to produce fluorescent reporters or color changes within the composite.
Engineered Melanin Synthesis: Fungal strains such as Aspergillus niger can be genetically modified to place melanin synthesis genes under the control of specific inducible promoters, causing the living material to display distinct color intensities when exposed to specific chemical triggers.
Embedding cell-free protein synthesis (CFPS) machinery and DNA encoding chromoproteins within hydrogels or polymers integrated throughout an inert mycelium scaffold. Upon mechanical stress, embedded microcapsules can release water or specific chemical triggers to rehydrate and activate the CFPS system, initiating the in vitro synthesis of visible pigments without the need to maintain living fungal cells.
The ideal organism must exhibit vigorous vegetative growth on low-cost lignocellulosic substrates, possess high mechanical compressive strength, resist environmental contamination, and, crucially, be highly amenable to genetic modification. Two white-rot basidiomycetes dominate the current landscape of mycelium biofabrication and serve as the primary candidates for this application: Pleurotus ostreatus (the oyster mushroom) and Ganoderma lucidum (the reishi mushroom).