Week 07: Genetic Circuits Part 2
Assignment Part 1: Intracellular Artificial Neural Networks (IANNs)
- What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions?
Traditional genetic circuits primarily rely on Boolean logic (AND, OR, NOT gates), which results in “all-or-nothing” digital responses. Intracellular Artificial Neural Networks (IANNs) offer several distinct advantages:
- Non-linear Signal Integration: Unlike Boolean gates that require strict thresholds, IANNs use activation functions (like Hill functions) to process analog chemical gradients, allowing for more nuanced environmental sensing.
- Weighted Inputs: IANNs allow for “tunable” inputs. By varying promoter strength or ribosome binding site (RBS) efficiency, the cell can assign different weights (w) to various biological signals, prioritizing one metabolite over another.
- Noise Filtering: Biological environments are inherently “noisy.” The summation and thresholding architecture of a perceptron acts as a natural buffer, preventing the circuit from misfiring due to minor stochastic fluctuations in gene expression.
- Computational Density: A single-layer IANN can perform complex classifications that would require a much larger and more metabolically taxing combination of traditional logic gates.
- Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
Application: An engineered E. coli strain that acts as a therapeutic diagnostic tool within the human gut.
- Input/Output Behavior: A) Inputs (Xn): The system senses multiple biomarkers of inflammation simultaneously, such as Nitric Oxide (X1), Thiosulfate (X2), and Calprotectin (X3). B) Processing: The IANN integrates these concentrations. Only if the weighted sum of these inflammatory markers exceeds a specific threshold (indicating a disease state rather than a transient spike) does the “neuron” fire. C) Output (Y): The controlled secretion of an anti-inflammatory cytokine (e.g., IL-10) or a visual reporter like GFP for diagnostic stool analysis.
- Limitations: A) Metabolic Burden: Expressing multiple sensing proteins and processing machinery can redirect significant resources away from cellular growth (chassis stress). B) Orthogonality: Ensuring that the synthetic components do not cross-react with the host cell’s native RNA processing machinery is a major design challenge.
- Below is a diagram depicting an intracellular single-layer perceptron where the X1 input is DNA encoding for the Csy4 endoribonuclease and the X2 input is DNA encoding for a fluorescent protein output whose mRNA is regulated by Csy4. Tx: transcription; Tl: translation. Draw a diagram for an intracellular multilayer perceptron where layer 1 outputs an endoribonuclease that regulates a fluorescent protein output in layer 2. The diagram illustrates a two-layer genetic cascade functioning as an artificial neural network within a cellular chassis.
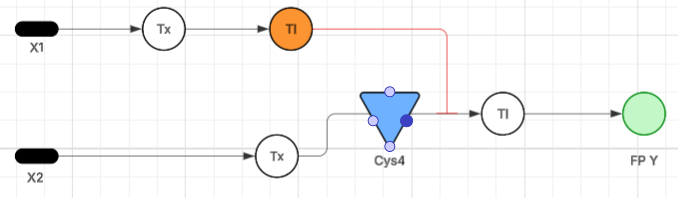

Layer 1 (Input Processing): Genetic input X1 undergoes transcription (Tx) and translation (Tl) to produce the endoribonuclease Csy4 (represented by the node in Layer 2). Layer 2 (Signal Integration): Genetic input X2 is transcribed into mRNA. The Csy4 protein produced in Layer 1 acts as a negative regulatory weight, targeting and cleaving the X2 mRNA transcript. This site-specific cleavage inhibits the subsequent translation (Tl) of the final output. Output (Y): The system results in the expression of Fluorescent Protein (FP Y) only in the absence of Csy4 and the presence of X2 stimulus, effectively mimicking a logic gate with tunable biochemical weights.
Assignment Part 2: Fungal Materials
- What are some examples of existing fungal materials and what are they used for? What are their advantages and disadvantages over traditional counterparts?
Fungal materials, often referred to as mycomaterials, are a rapidly growing field of sustainable engineering. These materials are typically grown by inoculating agricultural waste with mycelium (the root-like structure of fungi), which acts as a natural biological glue.
Some existing fungal materials are:
- Myco-Foam: Used as a direct replacement for Polystyrene (Styrofoam). Companies like Ecovative Design grow custom-molded packaging that is fully compostable.
- Myco-Bricks: Mycelium is grown into bricks or insulation panels. These are used in experimental architecture for their thermal and acoustic properties ( because of its porous and fibrous nature).
- Myco-Leather: Brands like Mylo or Reishi produce a material that mimics the texture and durability of animal leather for the fashion industry
In terms of Sustainability fungal materials are carbon-negative and fully biodegradable. They grow on agricultural “waste” (like corn husks or wood chips), turning low-value byproducts into high-value materials; on the other hand traditional materials like plastics are petroleum-based and contribute to long-term microplastic pollution. Animal leather has a massive carbon footprint due to the land and water required for livestock.
Regarding Growth Time fungal Can be grown and “manufactured” in days to weeks while traditional Leather requires years for an animal to mature, plastic production is nearly instant, the geological time required to create the oil it comes from is millions of years.
Also, Fungal materials are naturally fire-resistant and do not off-gas Volatile Organic Compounds (VOCs), which are common in synthetic foams and glues; any traditional foams are highly flammable and release toxic fumes during combustion or over time through degradation.
Despite their potential, fungal materials face specific engineering hurdles. In case of fungal, as we know, biological systems are inherently variable. Factors like humidity, temperature, and substrate consistency can lead to biological “noise”, making it difficult to produce perfectly uniform batches; industrial processes for plastics and metals are highly standardized, ensuring every unit is identical.
Because they are designed to be biodegradable, fungal are sensitive to moisture. If not properly sealed, they can begin to decay if used in outdoor or high-humidity environments; materials like PVC or high-density polyethylene are extremely durable and resist decay, which is their greatest strength during use but their biggest flaw as waste.
In Fungal moving from lab-scale prototypes to massive industrial throughput requires significant infrastructure. Furthermore, there is often a “yuck factor” or stigma associated with using “mushrooms” for clothing or housing that must be overcome.
- What might you want to genetically engineer fungi to do and why? What are the advantages of doing synthetic biology in fungi as opposed to bacteria?
An application for genetically engineering fungi is the development of wearable, autonomous biosensors (Biostickers) for industrial safety, specifically in mining environments. I want to engineer filamentous fungi (such as Aspergillus nidulans) to sense sub-lethal concentrations of toxic gases (CO, CH4). Using an Intracellular Artificial Neural Network (IANN), the fungi would integrate chemical signals from the mine’s atmosphere. When a specific safety threshold is reached, the circuit triggers a visible phenotypic change, such as the expression of high-intensity chromoproteins (e.g., amilCP for a dark blue/purple color) or bioluminescence. This provides a zero-power, spark-safe, and low-cost early warning system for miners. Unlike electronic sensors, a “living sticker” on a helmet is intrinsically safe in explosive atmospheres and highly resistant to the physical rigors of a mine.
- What are the advantages of doing synthetic biology in fungi as opposed to bacteria?
While E. coli is often the default chassis for synthetic biology, fungi offer unique mechanical and biological advantages for a mining Biosticker:
- Environmental Resilience: Fungi are naturally evolved to thrive in harsh, low-moisture, and variable pH environments. In a mine, where humidity fluctuates and surfaces are abrasive, the fungal cell wall (chitin-based) provides superior structural integrity compared to the fragile membranes of bacteria.
- 3D Morphological Engineering (Mycelium): Fungi grow in complex hyphal networks. We can engineer the branching density of the mycelium to create a “living fabric” within the sticker. This allows for a higher surface area for gas diffusion and a more robust physical form factor that can be integrated into a wearable adhesive.
- Eukaryotic Transcriptional Control: Fungi possess sophisticated eukaryotic gene regulation. This allows for the implementation of complex, multi-layered IANNs with post-translational modifications, which are necessary for the accurate folding of advanced reporter proteins that bacteria might struggle to produce.
- Secretory Power and Matrix Integration: Fungi are masters of secretion. They can be engineered to secrete protective proteins into the surrounding hydrogel matrix of the sticker, effectively “engineering their own environment” to remain viable on a miner’s helmet for weeks without external maintenance.
Assignment Part 3: First DNA Twist Order
- Review Part 3: DNA Design Challenge of the week 2 homework. Design at least 1 insert sequence and place it into the Benchling/Kernel/Other folder you shared in the Google Form above. Document the backbone vector it will be synthesized in on your website.
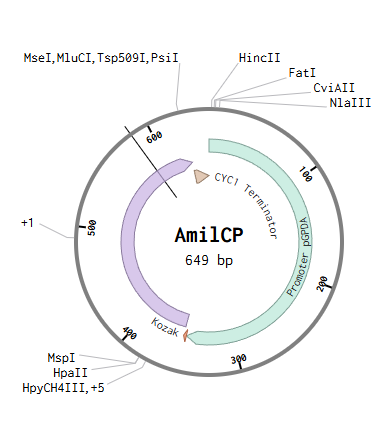

The insert contains a fungal expression cassette designed for a biosensing system in mining environments. The PgpdA promoter from Aspergillus drives expression of the AmilCP chromoprotein reporter. When environmental stress caused by toxic gases occurs, the fungus produces a visible blue signal. The construct includes a Kozak sequence for translation initiation and a transcription terminator.