Week 1 HW: Principles and Practices

1. Describe a biological engineering application or tool you want to develop and why
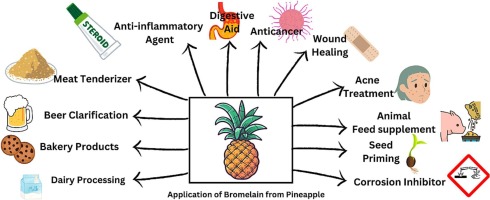
One biological engineering application I would like to develop at HTGAA is an active dressing or biomaterial for treating superficial wounds that incorporates bromelain, a proteolytic enzyme derived from pineapple.
This biomaterial would primarily promote gentle enzymatic debridement, reduce local inflammation, and support the healing process, using a naturally occurring and potentially accessible compound. The interest in this application stems from the fact that bromelain has demonstrated anti-inflammatory and proteolytic properties, but its direct, uncontrolled use can cause irritation or tissue damage. Therefore, integrating this compound into a controlled biomaterial would maximize therapeutic benefits and minimize potential risks, especially in primary healthcare settings or areas with limited access to healthcare facilities.
2. Describe one or more governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future, like ensuring non-malfeasance (preventing harm). Break big goals down into two or more specific sub-goals
Main Objective
To ensure that the development and use of a bromelain-containing biomaterial is safe, ethical, and does not cause harm to patients.
Sub-objectives
a. To ensure the safety and biocompatibility of the biomaterial before its clinical or educational use.
b. To prevent misuse or unregulated use, such as unsupervised home application, inappropriate concentrations, or self-medication.
c. To promote responsible and evidence-based use, especially in low-resource or educational settings.
3. Describe at least three different potential governance “actions” by considering the four aspects below (Purpose, Design, Assumptions, Risks of Failure & “Success”)
Action 1: Establishment of Minimum Formulation and Use Protocols
Purpose
Currently, bromelain can be acquired and used in biomedical applications without clear regulatory oversight. This action proposes the development of minimum standardized protocols for bromelain concentration, encapsulation, and safe use in biomaterials, in order to reduce potential risks and misuse.
Design
This action requires the participation of universities, biomaterials faculty, and ethics committees. Technical guidelines for academic and experimental use would be developed and subsequently reviewed and validated by institutional regulatory bodies or internal review committees.
Assumptions
It is assumed that safe concentration ranges for bromelain can be clearly defined. However, variability in enzymatic activity may occur depending on the source and processing of bromelain, which introduces uncertainty.
Risks of Failure and “Success”
- Failure: Protocols are not consistently followed or are perceived as unnecessary by users.
- Unintended success: The existence of protocols is used to justify premature or unauthorized clinical applications without sufficient preclinical or clinical trials.
Action 2: Mandatory Biocompatibility and Degradation Assessment
Purpose
The purpose of this action is to ensure that bromelain-containing biomaterials do not cause tissue damage, adverse biological reactions, or uncontrolled degradation when applied to biological environments.
Design
This action involves conducting basic in vitro evaluations, such as cytotoxicity and degradation tests. Additionally, these assessments would be incorporated as a standard requirement in biomedical engineering curricula and supervised by faculty members and academic committees.
Assumptions
It is assumed that in vitro assays provide an adequate approximation of in vivo behavior and that participating institutions possess the minimum technical and financial resources required to perform these tests.
Risks of Failure and “Success”
- Failure: Experimental results are misinterpreted or oversimplified by students or developers.
- Unintended success: Positive in vitro results generate a false sense of safety, leading to unauthorized testing in human subjects.
Action 3: Regulation of Discourse and Dissemination of the Product
Purpose
This action aims to prevent bromelain-based biomaterials from being promoted as inherently “natural” or “safe” without sufficient scientific evidence supporting such claims.
Design
Ethical communication guidelines would be established for academic and preclinical projects. This includes reviewing the language used in presentations, posters, publications, and online materials, with oversight from faculty members and evaluators.
Assumptions
It is assumed that non-expert audiences may misinterpret terms such as “natural” or “enzymatic,” and that developers are aware of and willing to uphold their responsibility for accurate and ethical communication.
Risks of Failure and “Success”
- Failure: Continued dissemination of exaggerated or misleading claims regarding safety or efficacy.
- Unintended success: Increased popularity and informal adoption of the product without adequate regulatory supervision.
4. Score (from 1-3 with, 1 as the best, or n/a) each of your governance actions against your rubric of policy goals
| Does the option: | Option 1 | Option 2 | Option 3 |
|---|---|---|---|
| Enhance Biosecurity | |||
| • By preventing incidents | 2 | 1 | 3 |
| • By helping respond | 2 | 1 | 3 |
| Foster Lab Safety | |||
| • By preventing incident | 1 | 2 | 3 |
| • By helping respond | 2 | 1 | 3 |
| Protect the environment | |||
| • By preventing incidents | 1 | 3 | 2 |
| • By helping respond | 1 | 3 | 2 |
| Other considerations | |||
| • Minimizing costs and burdens to stakeholders | 3 | 2 | 1 |
| • Feasibility | 2 | 3 | 1 |
| • Not impede research | 1 | 2 | 3 |
| • Promote constructive applications | 2 | 1 | 3 |
5. Drawing upon this scoring, describe which governance option, or combination of options, you would prioritize, and why. Outline any trade-offs you considered as well as assumptions and uncertainties
Based on the scoring, Action 2 (Mandatory Biocompatibility and Degradation Assessment) should be prioritized, as it most directly supports non-malfeasance by reducing the risk of biological harm through early safety evaluation.
Action 1 (Minimum Formulation and Use Protocols) should be implemented alongside it, as standardized guidelines help prevent misuse and ensure consistency across academic and experimental settings.
Action 3 (Regulation of Discourse and Dissemination) serves as a complementary measure, addressing risks related to misinformation and informal adoption.
The main trade-off is the increased cost and effort associated with testing and protocol enforcement; however, this is justified by the ethical need to prioritize safety. This approach assumes institutional capacity for basic testing and acknowledges uncertainty in translating in vitro results to real-world use.
Bibliography
- Ahmad, T., Ismail, A., Ahmad, S.A. et al. Extraction, characterization and molecular structure of bovine skin gelatin extracted with plant enzymes bromelain and zingibain. J Food Sci Technol 57, 3772–3781 (2020). https://doi.org/10.1007/s13197-020-04409-2
- Kansakar, U., Trimarco, V., Manzi, M. V., Cervi, E., Mone, P., & Santulli, G. (2024). Exploring the Therapeutic Potential of Bromelain: Applications, Benefits, and Mechanisms. Nutrients, 16(13), 2060. https://doi.org/10.3390/nu16132060