Homework
Weekly homework submissions:
Week 1 HW: Principles and Practices
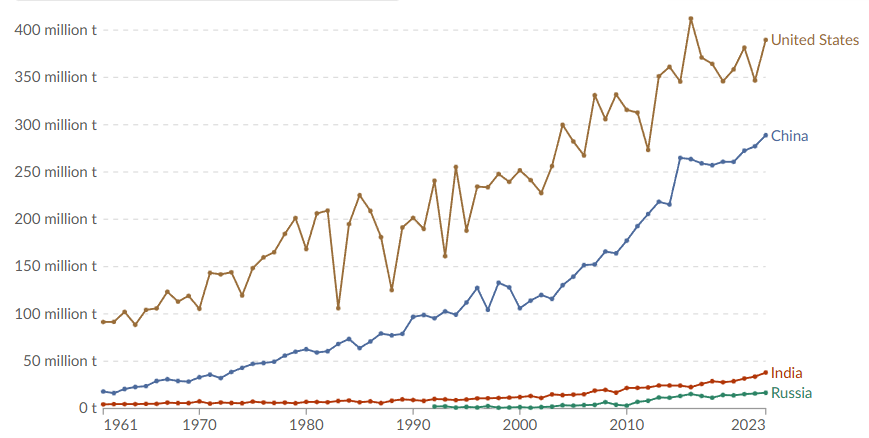
Week 1 Biological Engineering application: governance, policy; Abstract: Raising the same crop species over large areas and long time periods increases the risk of disease and pest outbursts, limits biodiversity and requires huge amounts of amendments and fertilizers. By genetically modifying the already socially approved off and profitable maize specie, global warming can be slowed down.
Week 2 HW: DNA Read, Write & Edit
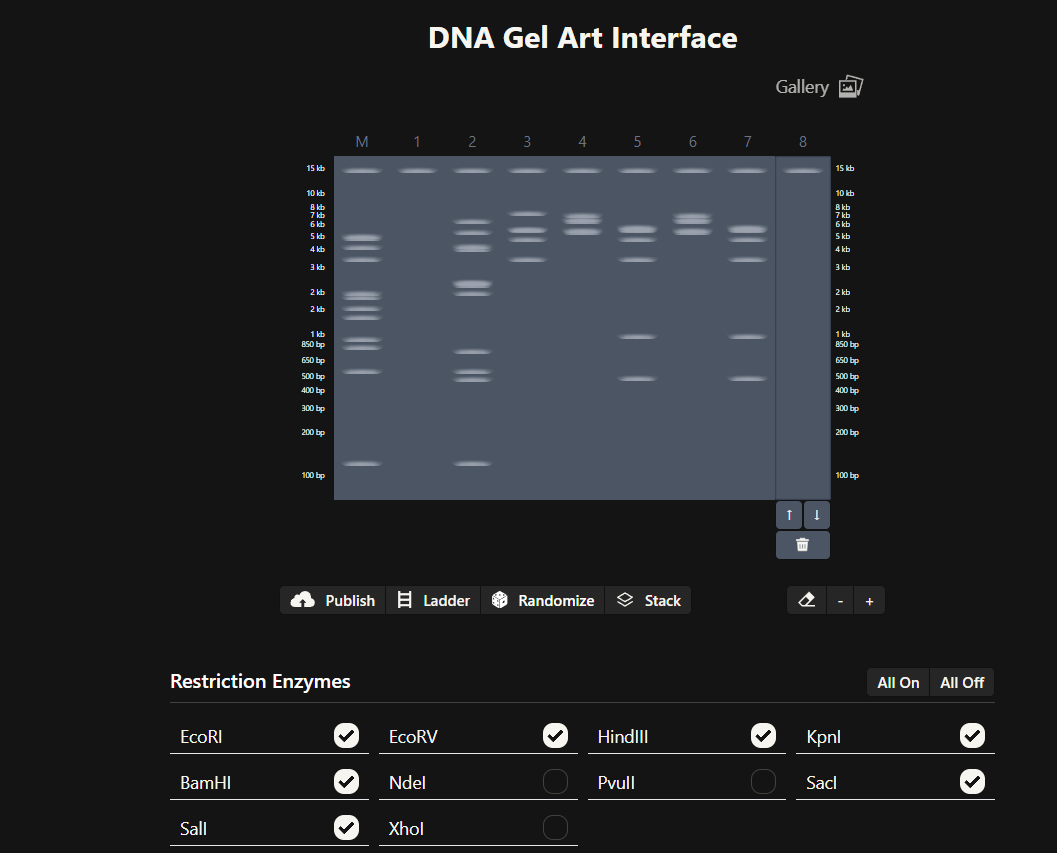
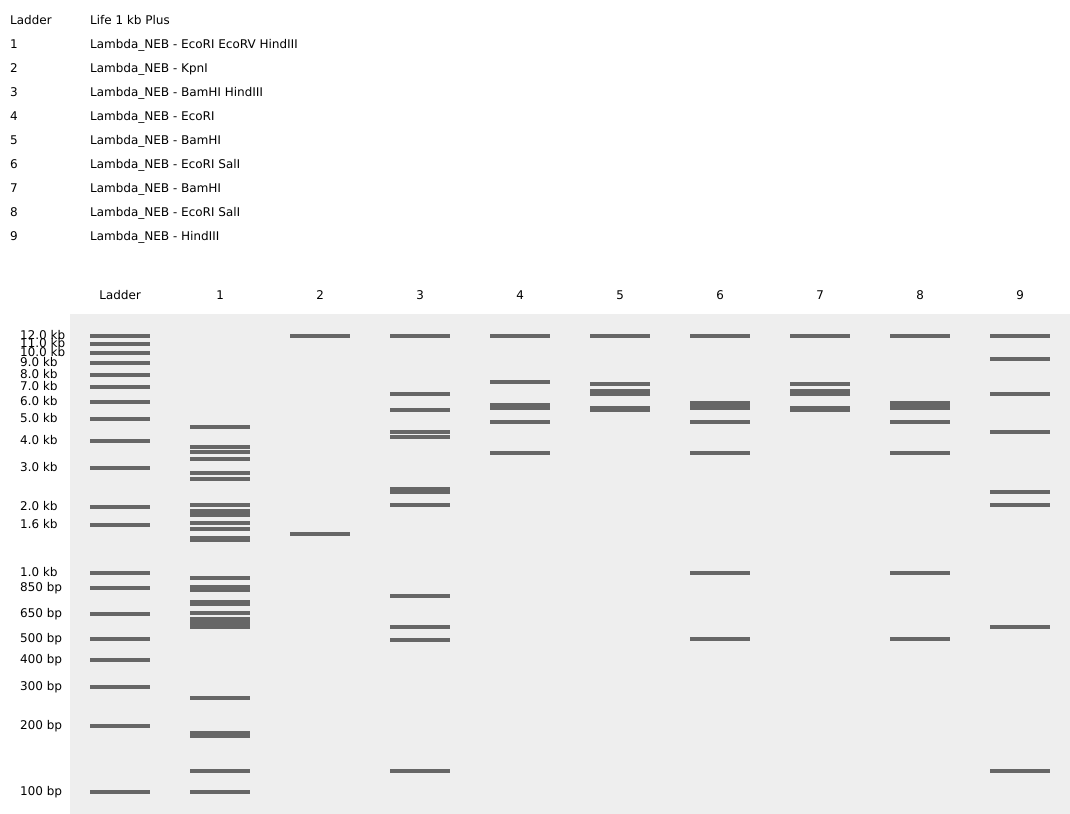
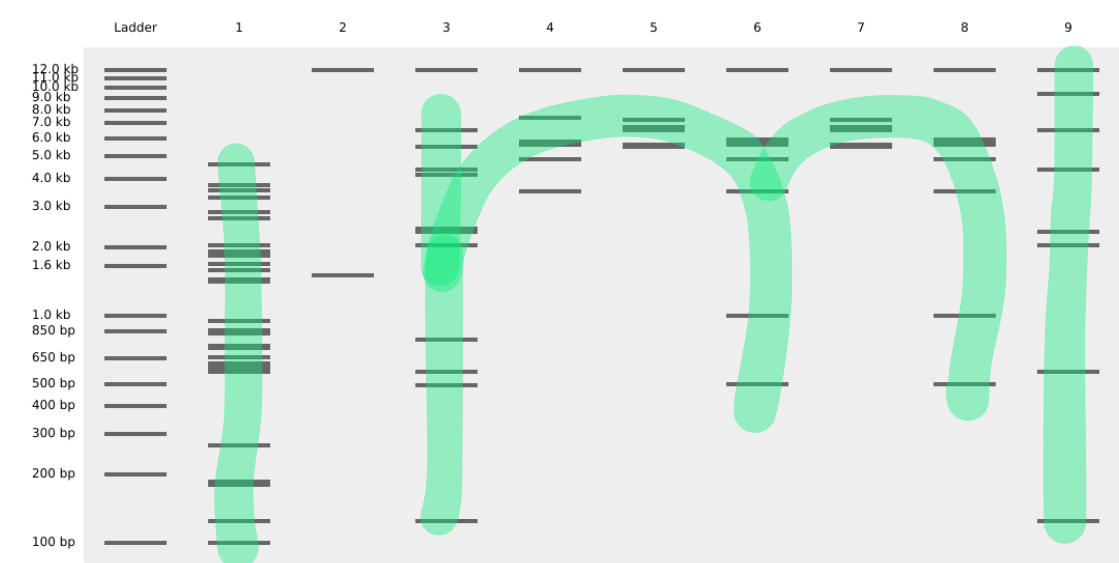
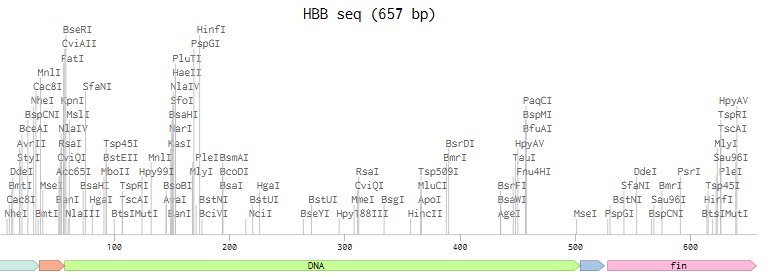
Week 2 Gel art, DNA processing; Abstract: The DNA molecule is an "information-packed" polymer, it encodes for proteins' monomer- amino acids. Even more so, DNA is electronegatively charged and so, using electrophoresis, DNA fragments' migration to the positively charged part of container can be controlled to constitute "gel art". I: I used rcdonovan to iterate a design (after unselecting Ndel, PvulI and Xhol enzymes), that I could later modify to my liking. I used ↑and ↓ to change specific ladders.
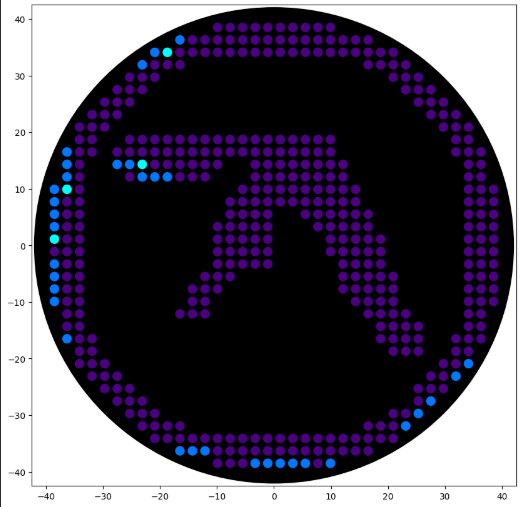
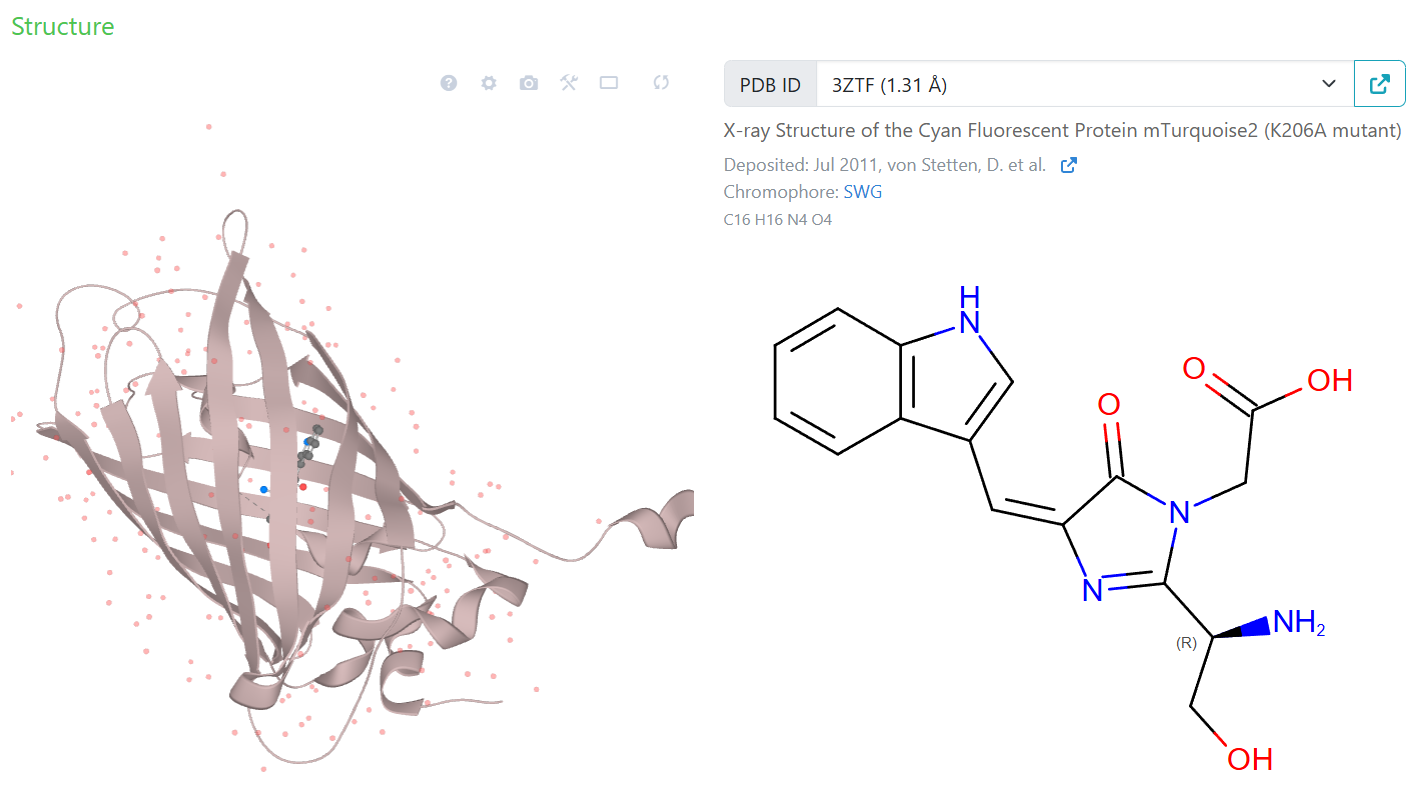
Week 3 Lab automation: Opentrons artwork; Abstract: Laboratory experiments and follow of protocols can lead to errors, thus in wrong assumptions. Automating lab work not only saves precious time, but also ensures quality of information delivered. I: I used rcdonovan’s site to iterate a design. Instead of manually creating the image I chose to import it from google and then make as many changes as I desired. Shown image is Aphex Twin’s, a famous British musician (“pioneering figure”), logo. Image copied from wikipedia. used colors: mkate2; electra2; mturquoise2.
Week 4 Protein Design part I; A. Conceptual questions answers 1. How many molecules of amino acids do you take with a piece of 500 grams of meat? (on average an amino acid is ~100 Daltons) a Dalton is a unit of measure, equal to 1/12 of carbon-12 atom weight; approx. 1.66 x 10-27 Kg.
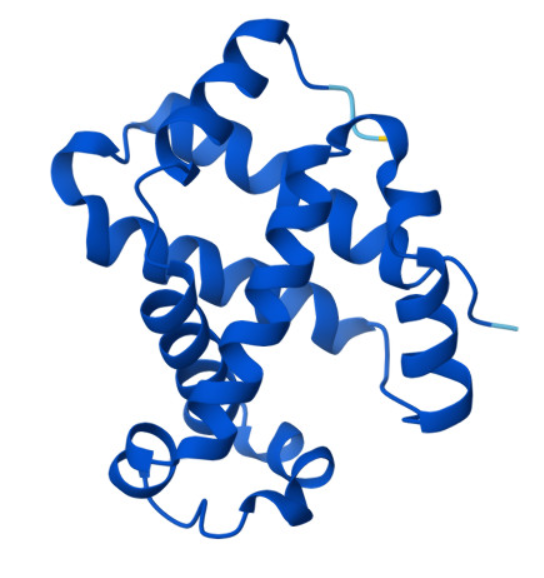
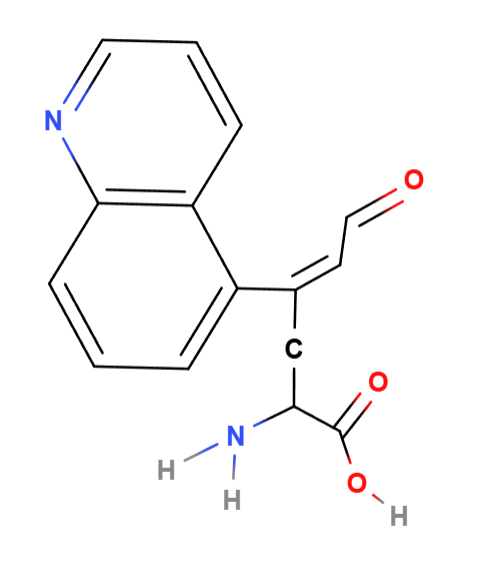
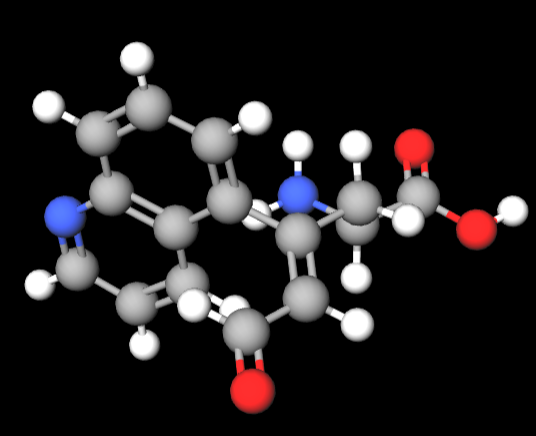
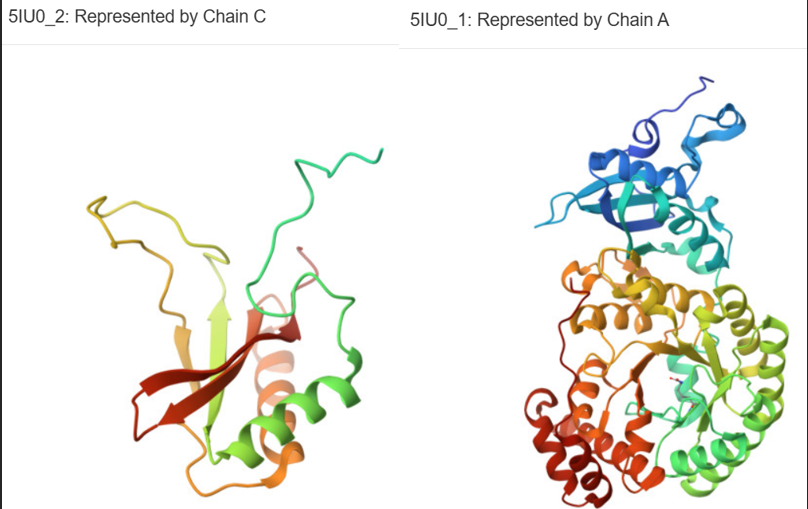
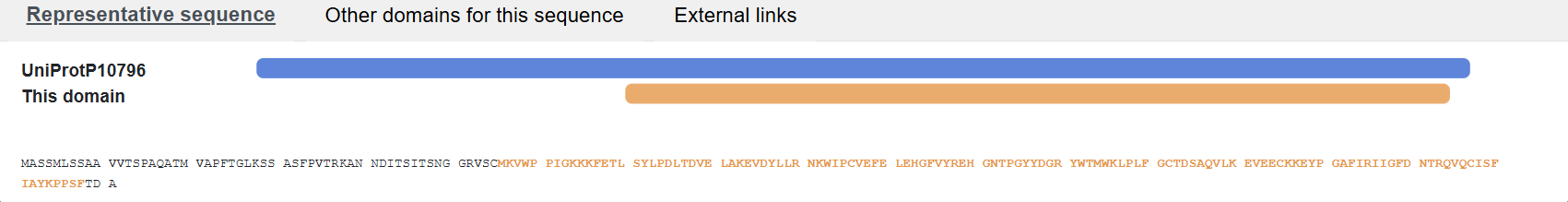
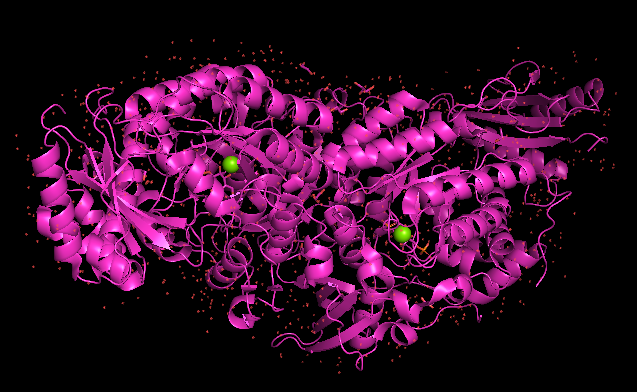
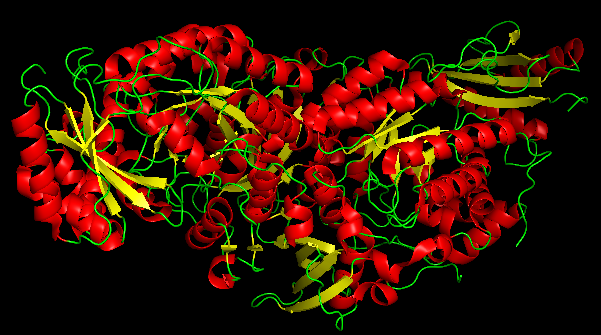
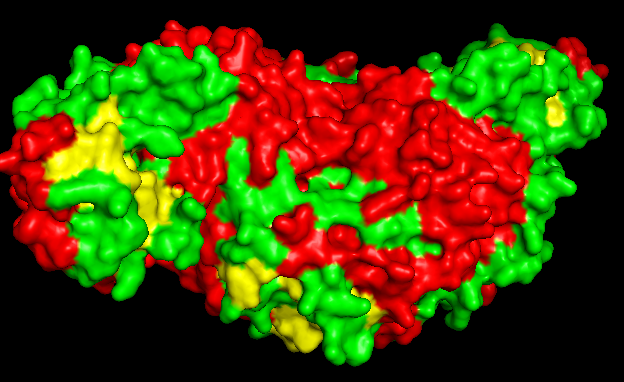
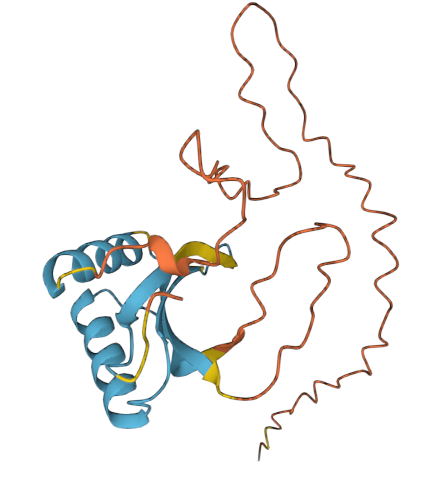
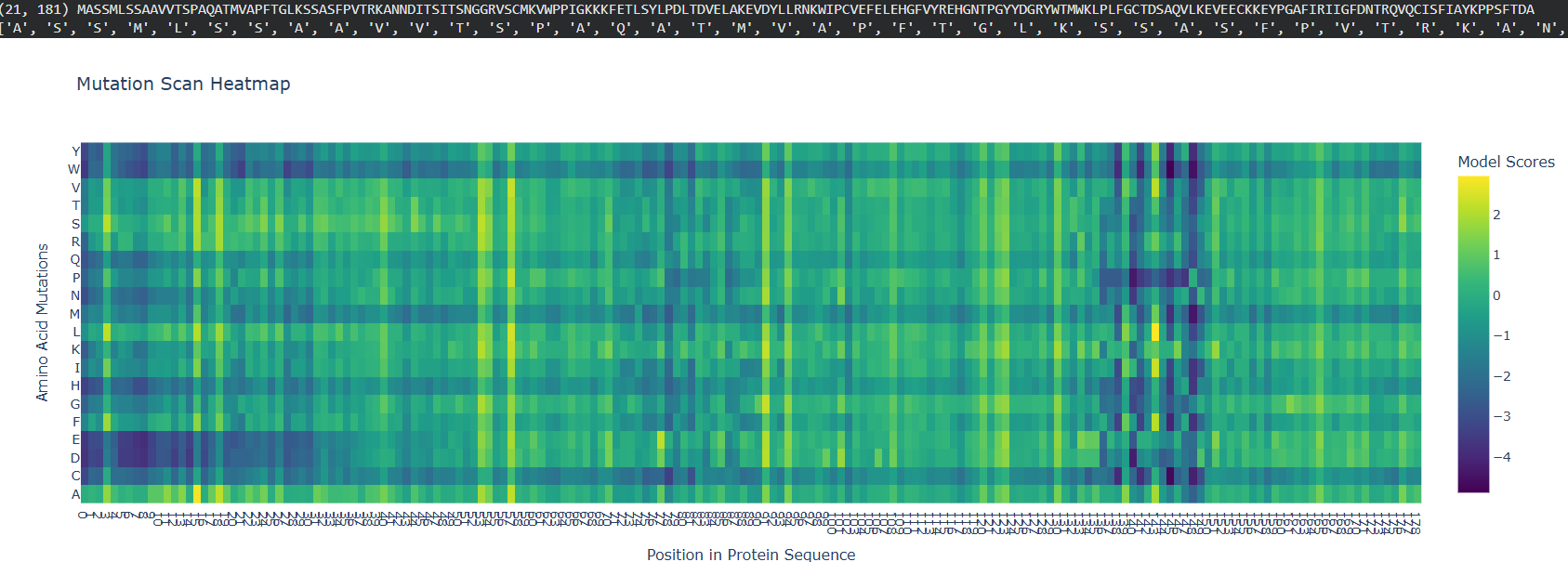
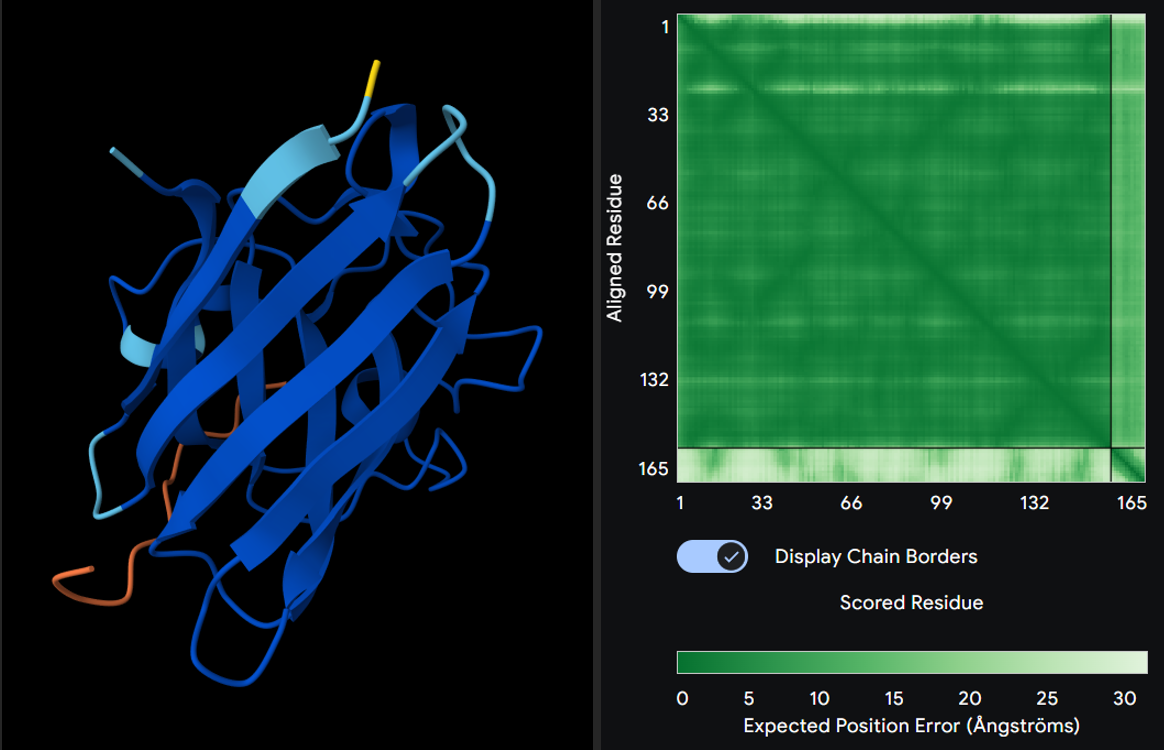
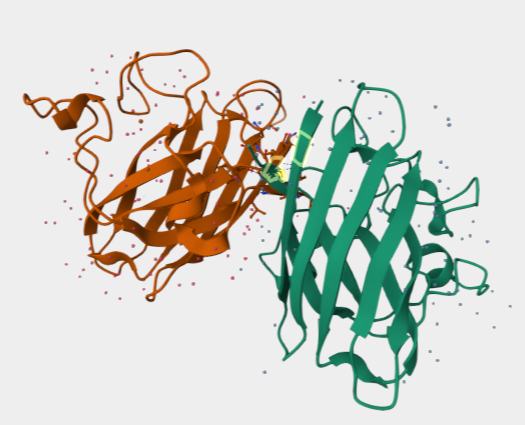
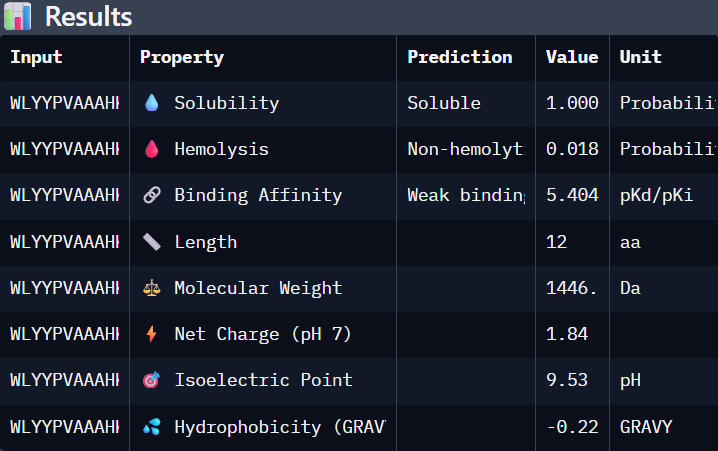
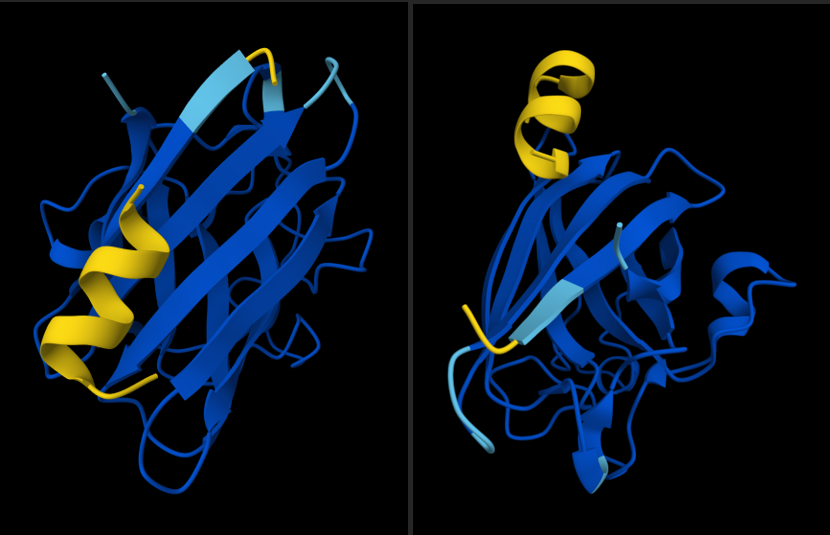
Week 5 Protein Design part II; Superoxide dismutase 1 (SOD1) – cytosolic antioxidant enzyme that converts superoxide radicals (O2-) into H2O2 and O2. In its native state, it forms a stable homodimer and binds Co and Zn. Mutations in SOD1 cause familial Amyotrophic Sclerosis (ALS). A4V leads to most aggressive forms of disease, as it destabilizes the N-terminus, perturbs folding energetics and promotes toxic aggregation.
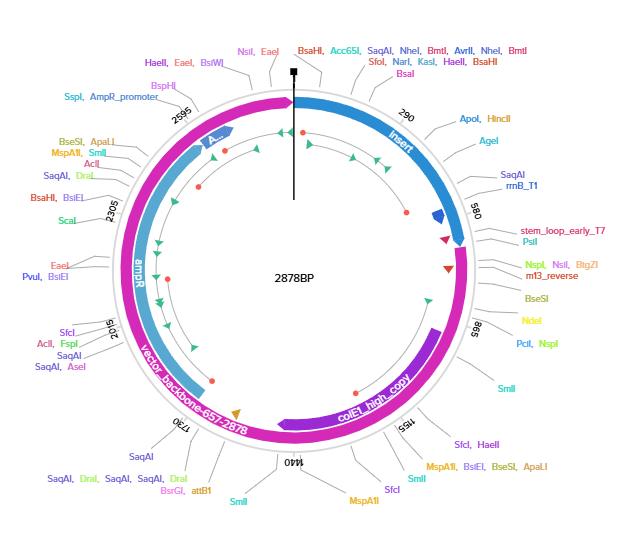
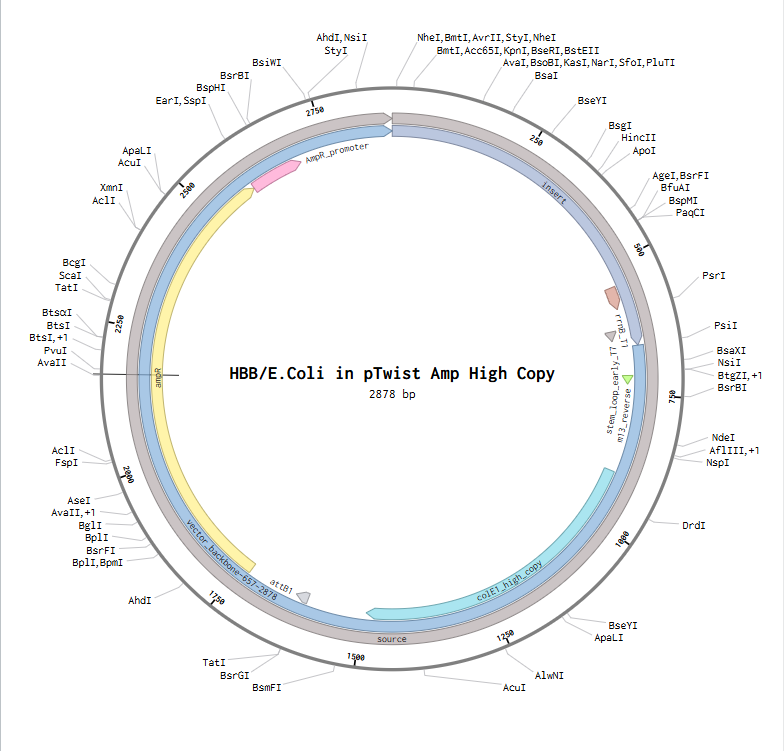
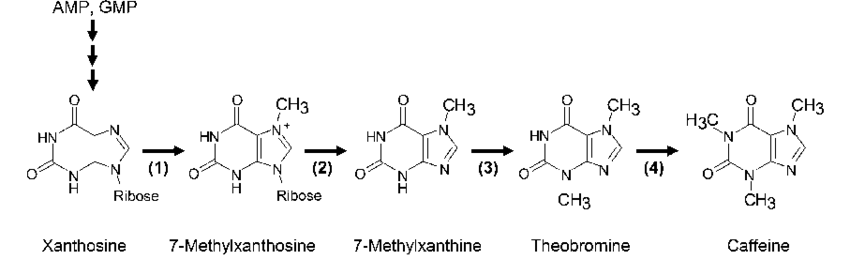
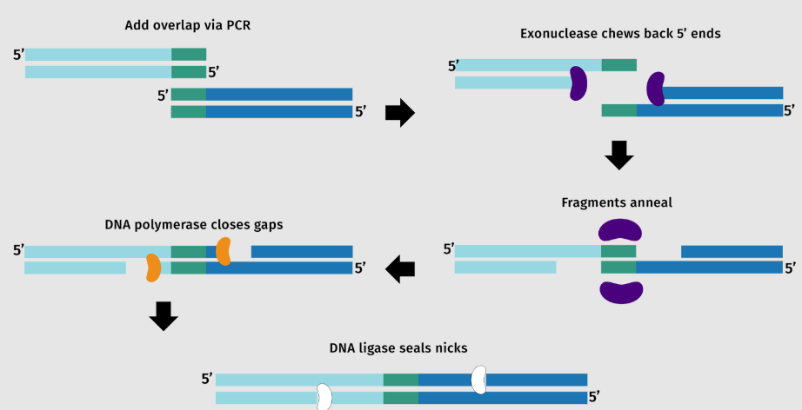
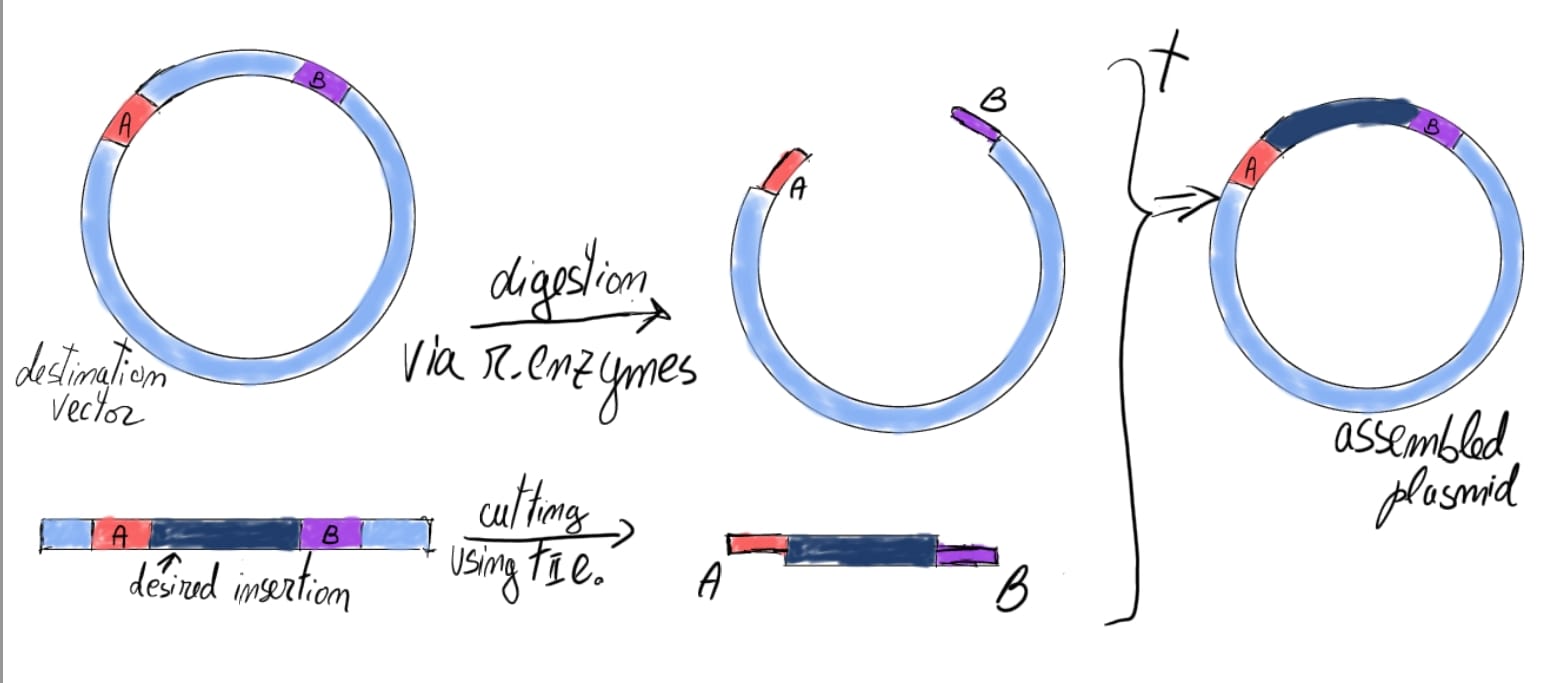
Week 6 Genetic circuits part I: Assembly Technologies; Abstract: Using different techonolgies, DNA can be synthesized via PCR (polymerase chain reaction); and genes can be added to vectors that later get expressed in cell dependent systems. Lab protocol questions: linked docs 1. What are some components in the Phusion High-Fidelity PCR Master Mix and what is their purpose?
Week 7 HW: Genetic circuits II
Week 7 Genetic circuits part II: Neuromorphic Circuits; Abstract: . Part 1) 1. What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions? IANNs allow for designing circuits that can process multiple types of input signals at once to execute sophisticated, non-linear tasks (logic pathways); traditional genetic circuits operate on linear Boolean logic (yes/no, 0/1), and are inefficient for mapping complex goals or biological relationships. Furthermore, IANNs adapt and learn: (or at least have the potential for it) they enable the execution of complex decision boundaries by approximating any continuous function on a bounded domain.
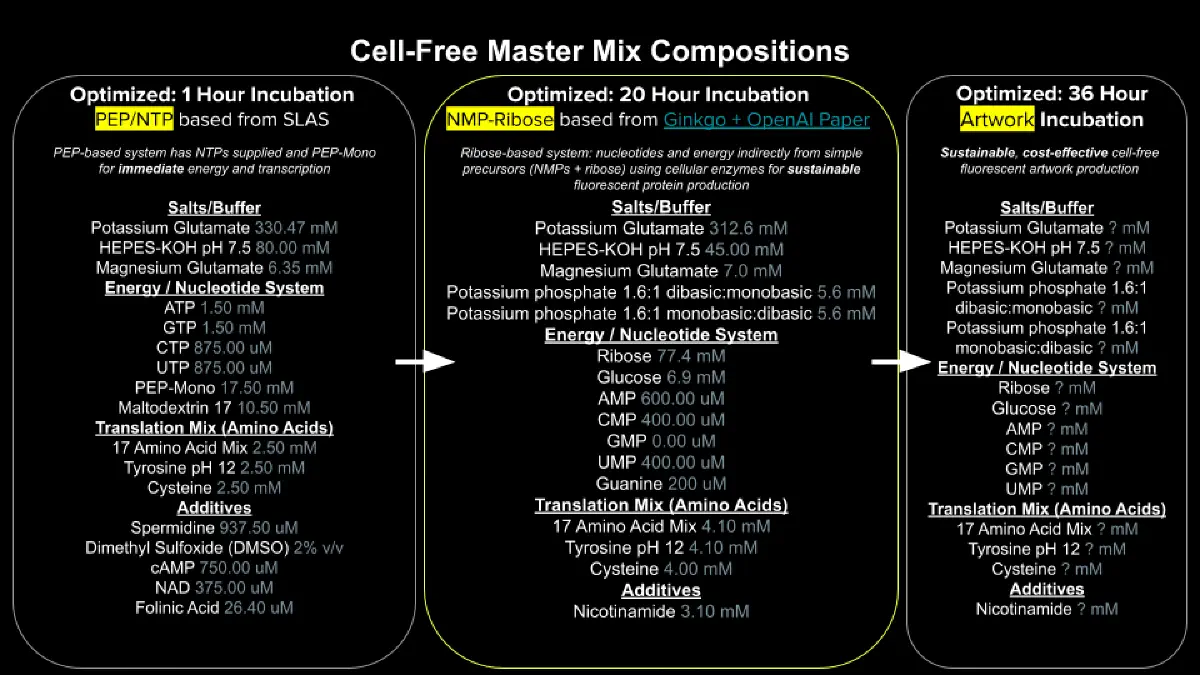
Week 9 Cell-Free Systems; Abstract: Cell free (protein synthesis) systems are crude cell independent synthesis technologies that, by using lysate substrate can perform biochemical reaction such as prot synthesis outside of living cells. General homework questions 1. Explain the main advantages of cell-free protein synthesis over traditional in vivo methods, specifically in terms of flexibility and control over experimental variables. Name at least two cases where cell-free expression is more beneficial than cell production.
Week 10 HW: Measurement Technology
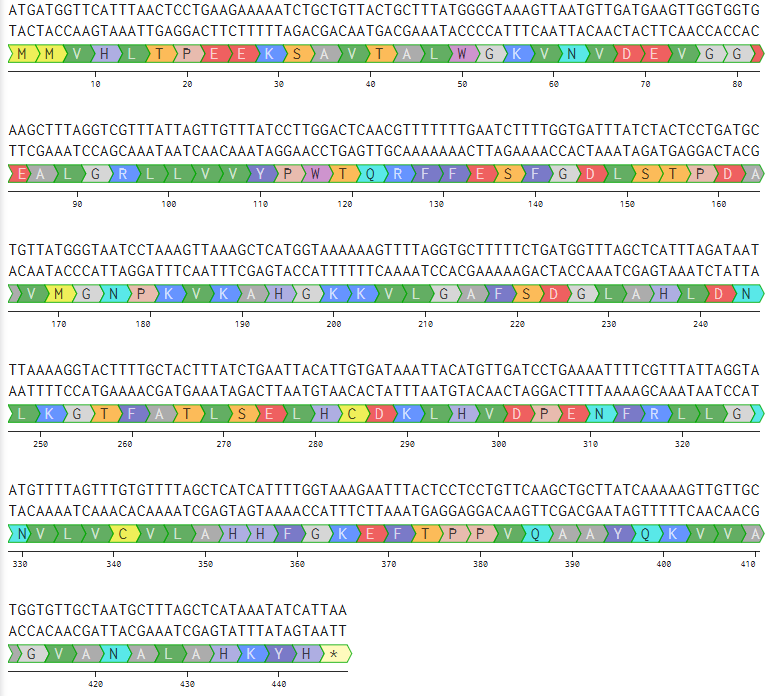
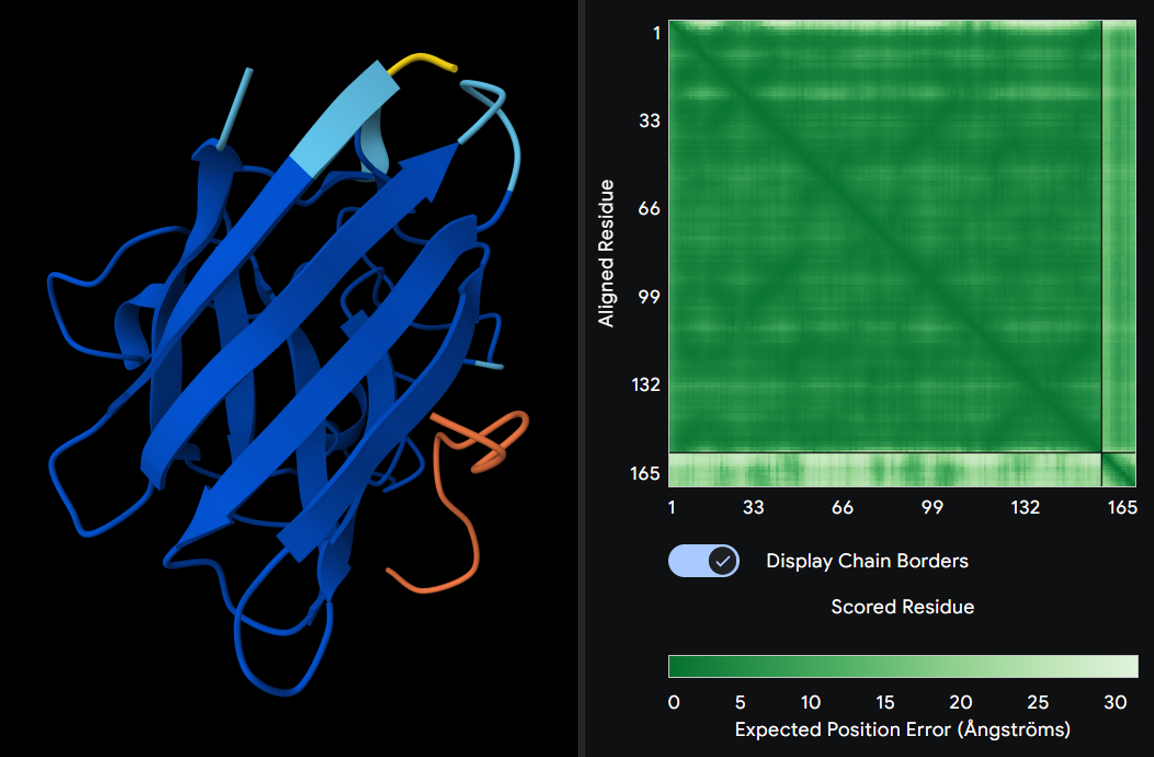
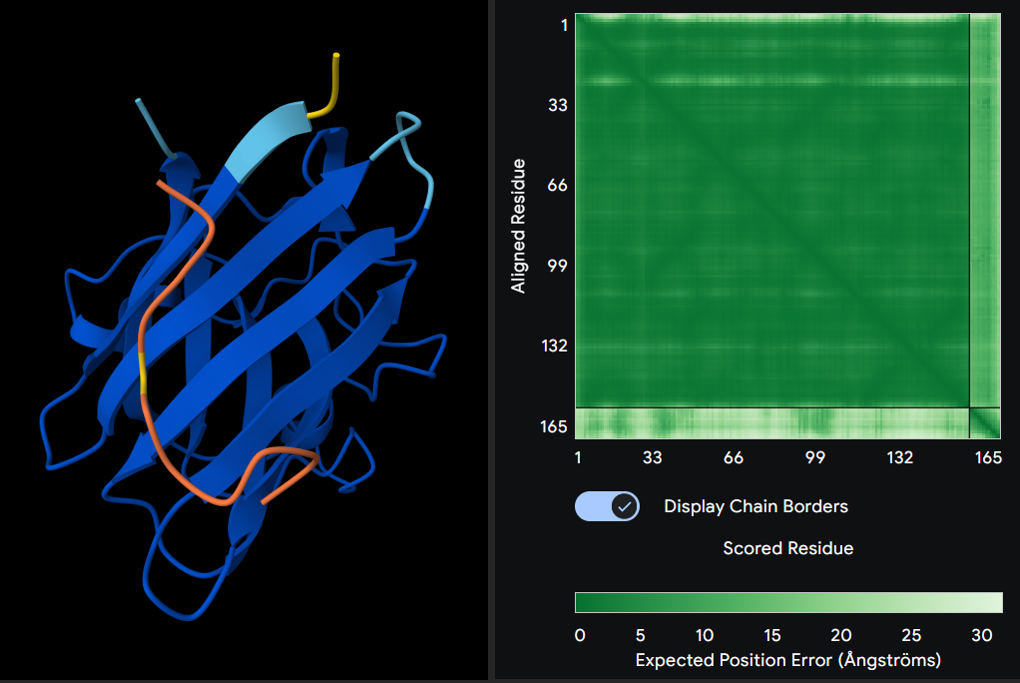
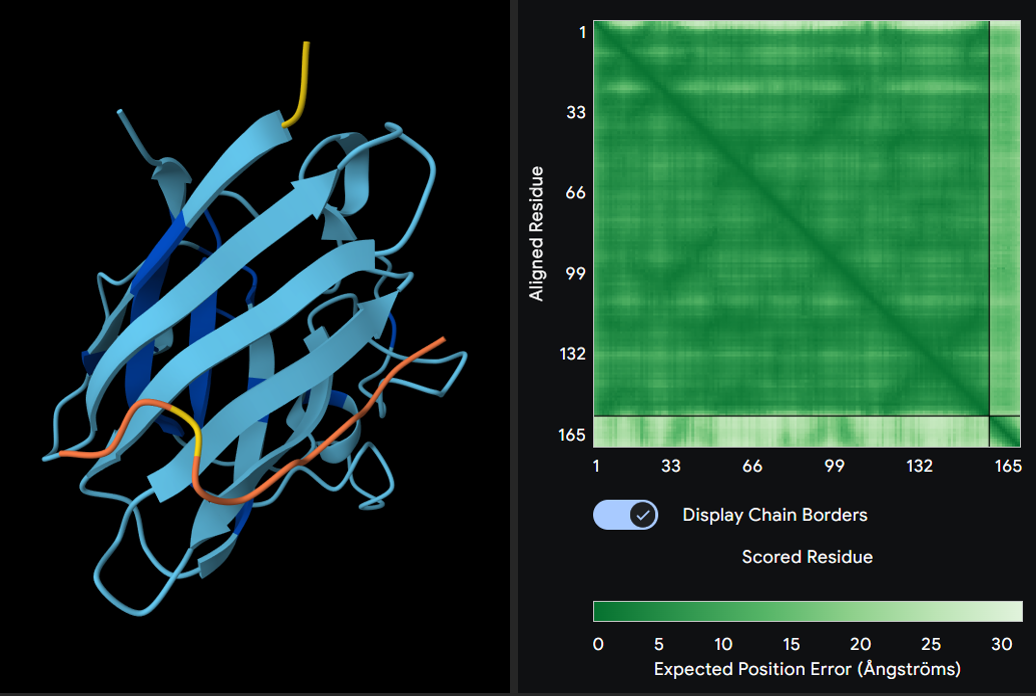
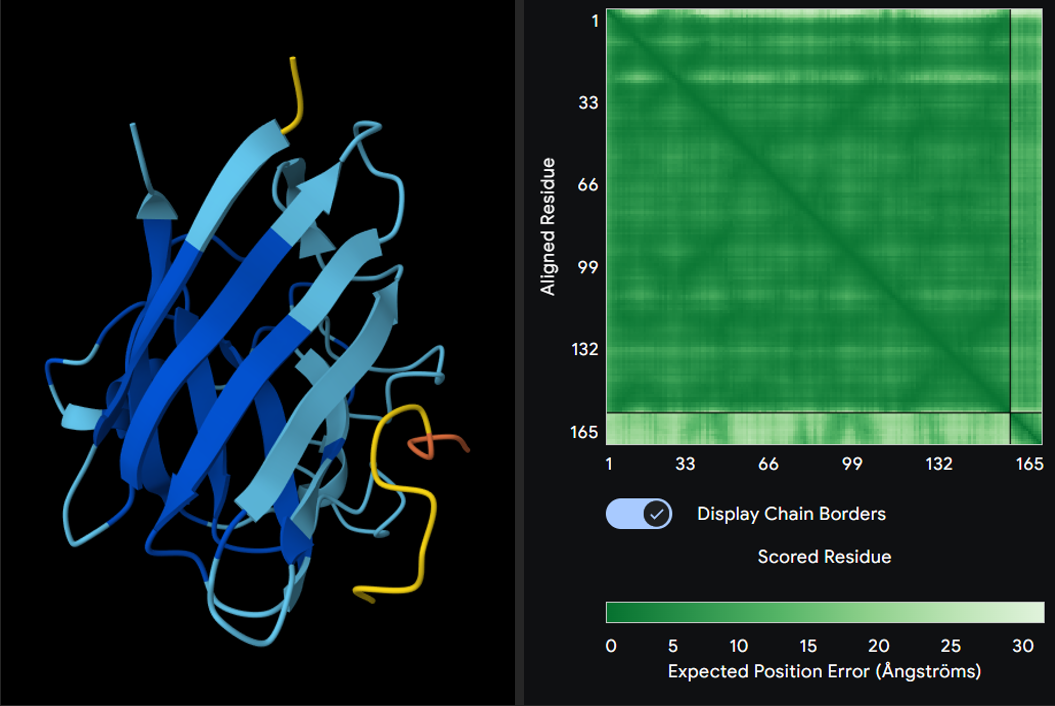
Week 10 Advanced Imaging & Measurement Technology; Abstract: . Waters Part 1 — Molecular Weight eGFP Sequence: MVSKGEELFTG VVPILVELDG DVNGHKFSVS GEGEGDATYG KLTLKFICTT GKLPVPWPTL VTTLTYGVQC FSRYPDHMKQ HDFFKSAMPE GYVQERTIFF KDDGNYKTRA EVKFEGDTLV NRIELKGIDF KEDGNILGHK LEYNYNSHNV YIMADKQKNG IKVNFKIRHN IEDGSVQLAD HYQQNTPIGD GPVLLPDNHY LSTQSALSKD PNEKRDHMVL LEFVTAAGIT LGMDELYKLE HHHHHH Note: This contains a His-purification tag (HHHHHH) and a linker (the LE before it).
Week 11 HW: Bioproduction & Cloud Labs
Week 11 Bioproduction, cloud-autonomous laboratories; Abstract: . A: The 1,536 Pixel Artwork Canvas | Collective Artwork global community project: *I guess I’m more the destructive type as I had more fun deleting pixels than I had painting the canvas (only after the experiment ended tho!!) ;)