Week 1 HW: Principles and Practices

Biological engineering application or tool you want to develop and why.
Functional thyroid hormone biosensing
Thyroid hormones play a central role in human physiology. The thyroid gland produces thyroxine (T4; 3,3′,5,5′-tetraiodo-L-thyronine) and triiodothyronine (T3; 3,3′,5-triiodo-L-thyronine), of which T3 is the biologically active form. T3 regulates energy homeostasis, metabolism, growth, and normal development by controlling transcriptional programs across multiple tissues. Disruptions in thyroid hormone signaling therefore can cause severe consequences. For people with thyroid-related disorders is neccesary then to monitore their hormone levels frequently and “play” with the doses until they obtain a precise amount that doesnt trigger any negative effects.
Specifically, I focused on this problem because I personally know some people that have thyroid-related disorders and have change form hiper to hypothyrodism due to unprecise doses and long waiting time before their next medical appointment. This is very dangerous overall for people in vulnerable communities that have even less opprotunities to go to medical appointments due to financial or time problems.
To address this challenge, I propose the development of an at-home biosensing platform capable of frequently monitoring thyroid hormone activity.
The envisioned system would allow individuals to collect small capillary blood samples at home and transmit hormone-related data to a mobile application. These data collected daily or weekly can create a personalized database of their hormone levels over time and relating this with their mood and other health symptoms, it will be able with time to predicted efficiently the change in their doses and alert when they have an abnormal level and need urgent in person medical attention.
To do this, rather than creating just an immunodiagnostic assay that measures free circulating T3, my approach centers on capturing functional thyroid hormone signaling. Specifically, the platform would rely on a sealed, single-use biosensing cartridge containing engineered eukaryotic cells, specifically modified yeast, designed to act as living biosensors. I decided to go with this approach because free circulating T3 in blood is not always a synonim of active hormones, because sometimes T3 has problems or delays in passing the cell membrane and then binding to the thyroid hormone receptor (TR) to regulate trasncription. With the yeast in the biosensor I can test both conditions to be fulfilled.
As a result, at the circuit level, the engineered yeast would constitutively express a synthetic TR. In another module, it will be a thyroid-response-element (TRE)–regulated promoter next to a reporter gene. In this way the synthetic TR will be the one regulating the expression of the reporter gene and only with the action of the T3 this signal will change. The signal could be colorimetric and can reflect different levels from low, normal to high fucnitonal T3 siganling. The succesful biosensor will be an example of precise and personalized medicine thanks to synthetic biology.
Governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future
a. Biosecurity and biosafety (preventing harm)
Ensure the engineered yeast biosensor does not pose risks to the operator, surrounding community, or environment.
Sub-goals:
- Containment and non-proliferation
- Environmentally safe disposal
b. Accessibility, equity, and health justice
Ensure the technology is accessible to everyone, especially the underrepresented communities for whom this biosensor is primarily intended. Guarantee these communities are treated with respect and can use the technology effectively and safely.
Sub-goals:
- Affordability and scalability
- Health literacy and inclusive communication
c. Ethical data governance and responsible AI use
Protect patient privacy and ensure that machine learning does not completely replace clinical judgment, which could lead to harmful consequences.
Sub-goals:
- User consent and data protection
- Responsible decision support
Describe at least three different potential governance “actions”
Healthcare professionals
Action 1: Clinical validation and contextualization of biosensor outputs
Healthcare professionals should validate the patient’s biosensor results by integrating them with patient-reported symptoms such as energy levels and then approve any dose changes. Also they should always be aware to refer patients to in-person care when abnormal patterns are detected.
- Purpose: Ensure remote monitoring supports good patient care and ML models do not produce medical risks.
- Design: Biosensor outputs are reviewed alongside symptom reports, with predefined thresholds that trigger clinical alerts.
- Assumptions: Clinicians have the time to review the patients’ results and will take them seriously in any part of the planet. Patients report symptoms honestly and consistently.
- Risks Failure: Incorrect interpretation or delayed referral could lead to inappropriate dose adjustments or other complications. “Success”: Over-reliance on remote monitoring that make patients disminih their routine clinical evaluations for other health problems.
Action 2: Patient training and informed consent
Obtain and maintain informed consent from all the patients by clearly explaining how the technology works, its limitations, and how medical data will be used and protected.
- Purpose: Empower patients to safely use the technology while ensuring autonomy and ethical data use.
- Design: In-person or video-based training on finger blood sampling, cartridge use, safe disposal, and data-sharing practices, with follow-up verification of understanding like tests.
- Assumptions: Clinicians will give clear and easy explanations without discrimination and always verifying multiple times that the patients understood.
- Risks: Failure: Patients doesnt understand and refuse to use the device due to fear or complexity. “Success”: They understand how to follow these practices but in their homes they do not have all the facilities to achieve it, such as safe disposal.
Ecuadorian regulatory bodies
Action 1: Biosafety, disposal, and data protection standards
Establish and enforce regulations addressing biosafety, transport, disposal of GMO-based cartridges, and data protection specific to ML-driven biosensors.
- Purpose: Update existing regulatory frameworks to responsibly enable new biotechnologies and AI technologies.
- Design: National guidelines and protocols to use, transport and dispose this biosensors. Anonymization standards and limits on third-party data sharing.
- Assumptions: Regulations will be supported and enforced consistently across all the Ecuadorian regions.
- Risks: Failure: Lack of regulatory adaptation may force illegal use of the technology. This is dangerous without regulations. “Success”: Overly rigid regulations could delay implementation or limit scalability.
Action 2: Equity-focused subsidy and access programs
Implement subsidies and distribution programs to ensure affordability and prevent exclusion of low-income or rural populations.
- Purpose: Prevent the technology from becoming accessible only to high-income users.
- Design: Price controls, public subsidies, and partnerships with rural clinics.
- Assumptions: Government will have the budget to give these subsidies and there will not be corruption.
- Risks: Failure: Without subsidies, access remains limited to middle and high socioeconomic groups. “Success”: Cost reduction pressures could compromise safety or quality controls. Also subsidies can create fake technology.
Developers / researchers
Action 1: Biologically contained yeast biosensor design
Design yeast biosensors that are non-reproducible and biologically contained, aligned with international GMO biosafety guidelines.
- Purpose: Enable safe and inocuous at-home use without risking environmental release and negative effects.
- Design: Intrinsic safeguards in the engineered yeast such as kill switches. Dedicated disposal containers that fully inactivate the cells.
- Assumptions: Containment mechanisms remain stable and do not interfere with sensing accuracy.
- Risks: Failure: Environmental release could occur, the biomass released can interfere with food chains. “Success”: Excessive containment may reduce biosensor reliability.
Action 2: Accessible user interfaces and deployment design
Develop interfaces and devices easy to use and in different languages. Be useful for people that can not read.
- Purpose: Ensure effective use across diverse cultural and educational contexts.
- Design: Pictographic instructions, community workers’ guidance, multilingual support.
- Assumptions: Users have basic access to smartphones to use the app.
- Risks: Failure: People doesnt understand the device so communities decide to not use it. “Success”: Extensive customization increases costs and limits scalability.
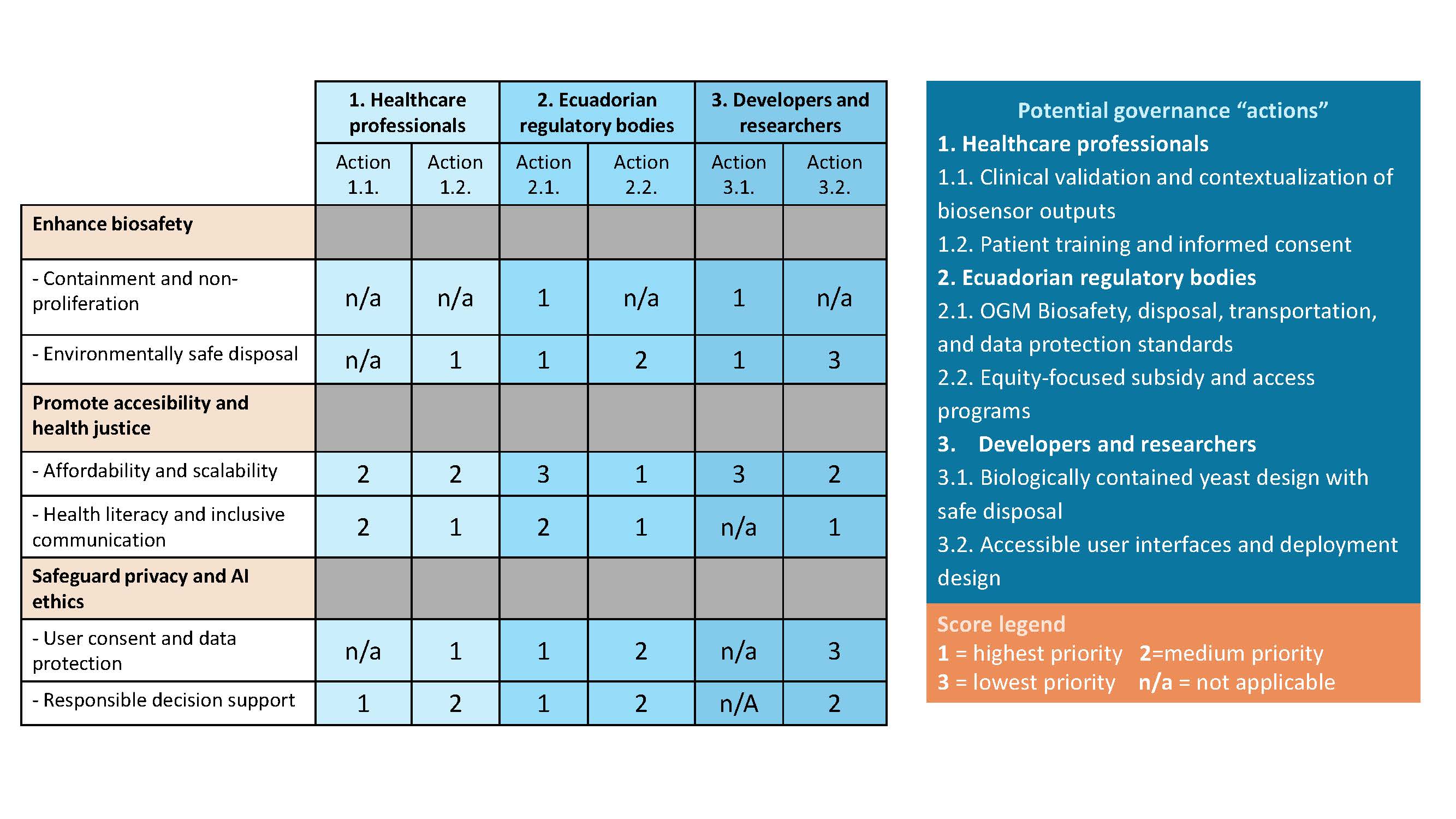
Score (from 1-3 with, 1 as the best, or n/a) each of your governance actions against your rubric of policy goals.

Describe which governance option, or combination of options, you would prioritize, and why.
Based on the scoring matrix, I would prioritize Action 2.1 (Ecuadorian biosafety, disposal, and data protection standards), Action 3.1 (biologically contained yeast design), and Action 1.2 (patient training and informed consent). Together, these actions have the highest direct impact on biosafety, accessability, and responsible data use, which are particularly important due to
- Ecuador’s biodiversity
- Biosensor’s focus on underserved communities
Actions 2.1 and 3.1 reduce biosafety risks prioritizing the user and environment protections thanks to clear regulations and smart design. Additionally, action 1.2 is essential because as this device is supposed to be used at-home, is important to make sure understand how to proper use it and always get informed consent.
Prioritizing this combination however may result in higher costs, this trade-off is justified to prevent environmental harm, maintain public trust, ensure safe and effective use by marginalized populations.
Audience: Ecuadorian regulatory bodies, local research institutions, and community health centers.
Reflection:
Based on what I learned in class, together with what I researched for this assignment about governance, a new ethical concern for me was the successful application of AI ethics to all these emerging technologies. I realized that these new developments are not going to stop, as was clearly stated in class, and that all we can do is “trust”. However for countries like Ecuador where our own data is not secured, as the constitution doesnt have clear guidelines and punishments for data protection, it is more than necessary to change and improve the laws so communities and their data is safe.
Week 2 Lecture Prep
Homework Questions from Professor Jacobson:
1. Nature’s machinery for copying DNA is called polymerase. What is the error rate of polymerase? How does this compare to the length of the human genome. How does biology deal with that discrepancy?
Error Rate of Polymerase = 1:10^6
Length of the human genome = approximately 3.2 billion base pairs
Comparison:
If the human genome were a book with 3.2 billion letters, and the polymerase made 1 error per million letters, you would find approximately 3,200 typos per replication scattered throughout the entire book.
Biological strategy to deal with this:
- Built-in error correction mechanisms:
3’-5’ proofreading exonuclease
5’-3’ error-correcting exonuclease
- Other error correction mechanisms:
MutS Repair System - Employed by bacteria
2. How many different ways are there to code (DNA nucleotide code) for an average human protein? In practice what are some of the reasons that all of these different codes don’t work to code for the protein of interest?
Number of different DNA codes:
• Average human protein: 1,036 bp (~345 amino acids)
• Due to the degeneracy of the genetic code (most amino acids are encoded by 2-6 different codons), there are an a lot of different DNA sequences that could encode the same protein
For a 345 amino acid protein, if we assume an average of ~3 synonymous codons per amino acid, there would be roughly 3³⁴⁵ ≈ 10¹⁶⁵ different possible DNA sequences encoding the same protein sequence.
Why all these codes don’t work in practice:
a. Secondary structure formation (GC content issues)
GC content affects secondary structure
b. RNA cleavage sites
Certain sequence motifs make the mRNA unstable
c. Codon usage bias
Different organisms prefer different synonymous codons
d. Repeats and homopolymers
Sequences like “TTTTTTTTTT” or “GGGGGGGGGG” (shown in assembly examples) cause problems
e. Base pairing energies
Homework Questions from Dr. LeProust:
1. What’s the most commonly used method for oligo synthesis currently?
The phosphoramidite method is the most commonly used approach. This was developed by Caruthers in 1981.
2. Why is it difficult to make oligos longer than 200nt via direct synthesis?
Concerns about “truncation products” and maintaining “full-length” material. Although, Twist has pushed boundaries by demonstrating direct synthesis of 700-mers, which they describe as a breakthrough.
3. Why can’t you make a 2000bp gene via direct oligo synthesis?
Because the stepwise synthesis chemistry becomes too error-prone at such lengths. Instead, genes are made through assembly of shorter oligonucleotides (Ilumina).
Homework Question from George Church
1. What are the 10 essential amino acids in all animals and how does this affect your view of the “Lysine Contingency”?
The 10 essential amino acids are: Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan, Valine, and Arginine.
This make me understand that the “Lysine contingency” stablished in Jurassic Park is not a good containment method. Why the “Lysine Contingency” fails for me
Carnivorous dinosaurs would get lysine from eating other animals (all animal tissues contain lysine)
Herbivorous dinosaurs could get lysine from legumes and other lysine-rich plants
Lysine is already in the food chain everywhere
Lysine is not the only aminoacid we cannot create.
So there’s no real contingency. The dinosaurs would just eat normally and get lysine like every other animal does.
For real biocontainment, you need dependency on something that doesn’t exist in nature at all.
2. What code would you suggest for AA:AA interactions?
Existing codes:
• NA:NA → Watson-Crick base pairing (A-T, G-C)
• NA:AA → Genetic code (codons → amino acids)
• AA:NA → TALE code (slide shows 2 AA recognize 1 DNA bp)
For AA:AA, the simplest code would be:
- Charge complementarity: (+) ↔ (−)
Positive (K, R, H) pairs with Negative (D, E)
- Hydrophobic complementarity: Nonpolar ↔ Nonpolar
(L, I, V, F, W, A) cluster together
- Polar complementarity: H-bond donors ↔ H-bond acceptors (S, T, N, Q, Y) form H-bond networks
3. Given the one paragraph abstracts for these real 2026 grant programs sketch a response to one of them or device one of your own.
Sketch Response to BioStabilization Systems (BoSS) Program
The BoSS program seeks to eliminate the cold chain for biologics by achieving room-temperature stability.
I propose engineering biologic-producing cells with protective genes from extremophile organisms that naturally survive extreme heat. Specifically, inserting heat-shock proteins from Thermus aquaticus (hot springs bacteria), trehalose biosynthesis pathways from tardigrades, and compatible solutes from halophilic bacteria would protect therapeutic cells and proteins from heat-induced degradation.
References
Baluta, S., Romaniec, M., Halicka-Stępień, K., Alicka, M., Pieła, A., Pala, K., & Cabaj, J. (2023). A novel strategy for selective thyroid hormone determination based on an electrochemical biosensor with graphene nanocomposite. Sensors, 23(2), 602.
Eskandar, K. (2026). Artificial intelligence and synthetic biology: biosecurity risks, dual-use concerns, and governance pathways. AI and Ethics, 6(1), 66.
Shahid, M. A., Ashraf, M. A., & Sharma, S. (2018). Physiology, thyroid hormone.
Note on AI assistance: Claude.ai was used to improve the clarity and organization of English writing based on ideas and concepts already developed by the student.