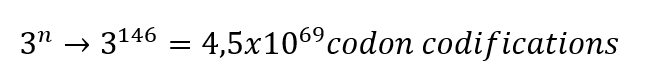Week 01 HW: Principles and Practices


This week we evaluate the principles and governance policies for our new ideas. It is important to avoid the misuse of these new developments.
1. First, describe a biological engineering application or tool you want to develop and why. This could be inspired by an idea for your HTGAA class project and/or something for which you are already doing in your research, or something you are just curious about.
Portable Biosensor for Rapid Identification of Animal Venom Toxicity Profiles in Emergency Settings
This idea came about because I am from Colombia, where in some rural areas access to health care is a challenge, especially in emergencies. In low- and middle-income countries, it is common for people to encounter dangerous and venomous animals such as snakes, scorpions, spiders, jellyfish, and venomous fish. In many cases, treating a person who has been bitten or stung is difficult because doctors and emergency teams need to know the species and characteristics of the animal to provide appropriate care.
Identifying the animal that bit or stung a person is challenging because sometimes patients are unconscious, or, in general, people do not have the knowledge to identify the animal or describe its characteristics.
In these kinds of situations, the time window is crucial because if the time between the bite and receiving proper medical attention is too long, the chances of successfully treating the person decrease. In this context, it is essential to develop a tool that allows identification of the type of toxin affecting a person, for example, neurotoxic, hemotoxic, cytotoxic, or myotoxic. The use of a portable biosensor for rapid identification of animal venom toxicity profiles will allow identification of the type of venom or toxin affecting a person among the following: neurotoxic, hemotoxic, cytotoxic, or myotoxic. This will improve positive outcomes because it will be easier to treat the symptoms and administer the appropriate antivenom.
The general idea is that the portable biosensor identifies the type of venom using lateral flow assays (LFA) with antibodies directed against families of toxins. For example, snake venom is composed of a variety of proteins and enzymes; however, there are four main groups of proteins classified according to their activity.
Three-finger toxin (3FTx) family:
- Neurotoxic effects (causing paralysis)
- Cytotoxic effects
Phospholipases (PLA2s):
- Neurotoxic effects
- Cytotoxic effects (through direct/indirect plasma membrane disruption)
Metalloproteases (SVMPs):
- Hemotoxic effects (cause severe hemorrhage, inflammation, and coagulopathy)
Serine proteases (SVSPs):
- Multifunctional enzymes
These types of proteins are present in most snake species, which provides an advantage by reducing the impact of geographical variation, because the antibodies used by this sensor will be specific to the protein family rather than to a single type of snake venom.
2. Next, describe one or more governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future, like ensuring non-malfeasance (preventing harm). Break big goals down into two or more specific sub-goals. Below is one example framework (developed in the context of synthetic genomics) you can choose to use or adapt, or you can develop your own. The example was developed to consider policy goals of ensuring safety and security, alongside other goals, like promoting constructive uses, but you could propose other goals for example, those relating to equity or autonomy.
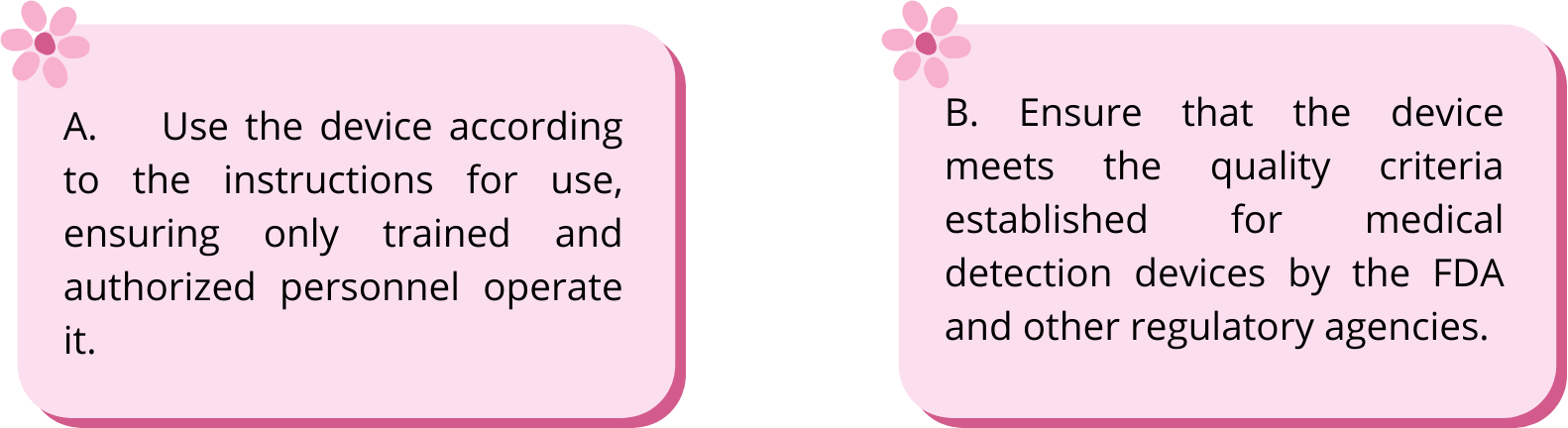
A. Use the device according to the instructions for use, ensuring only trained and authorized personnel operate it.
Specific objectives:
- Identify the secondary effects or risks associated with the use of this device
- Recommend that users comply with safety protocols and instructions.
B. Ensure that the device meets the quality criteria established for medical detection devices by the FDA and other regulatory agencies.
Specific objectives
- Identify the countries where this technology is urgently needed and therefore comply with the parameters established by their respective regulatory agency.
- Verify device accuracy and reliability
3. Next, describe at least three different potential governance “actions” by considering the four aspects below (Purpose, Design, Assumptions, Risks of Failure & “Success”). Try to outline a mix of actions (e.g. a new requirement/rule, incentive, or technical strategy) pursued by different “actors” (e.g. academic researchers, companies, federal regulators, law enforcement, etc). Draw upon your existing knowledge and a little additional digging, and feel free to use analogies to other domains (e.g. 3D printing, drones, financial systems, etc.). Purpose: What is done now and what changes are you proposing? Design: What is needed to make it “work”? (including the actor(s) involved - who must opt-in, fund, approve, or implement, etc) Assumptions: What could you have wrong (incorrect assumptions, uncertainties)? Risks of Failure & “Success”: How might this fail, including any unintended consequences of the “success” of your proposed actions?
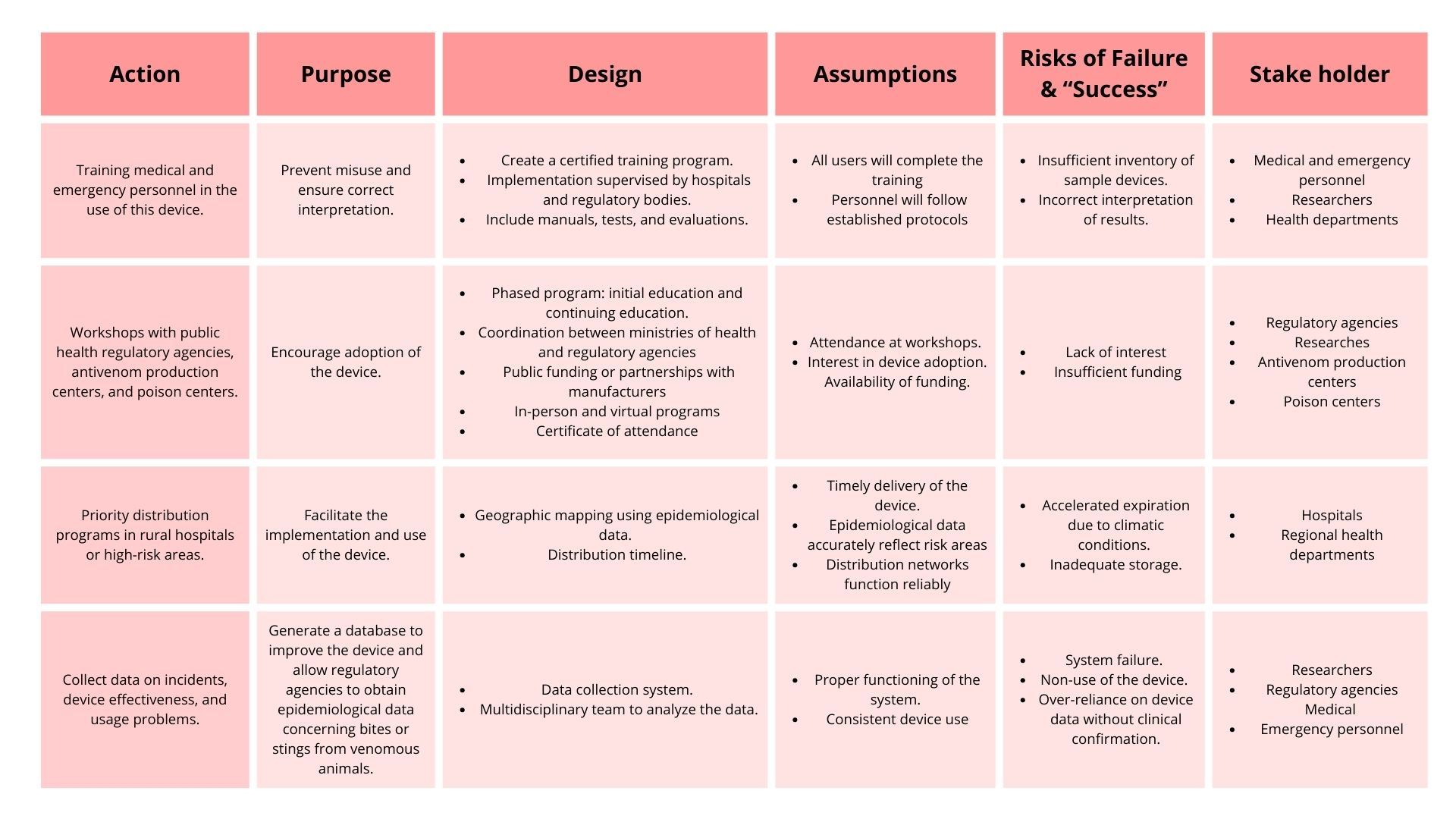

4. Next, score (from 1-3 with, 1 as the best, or n/a) each of your governance actions against your rubric of policy goals.
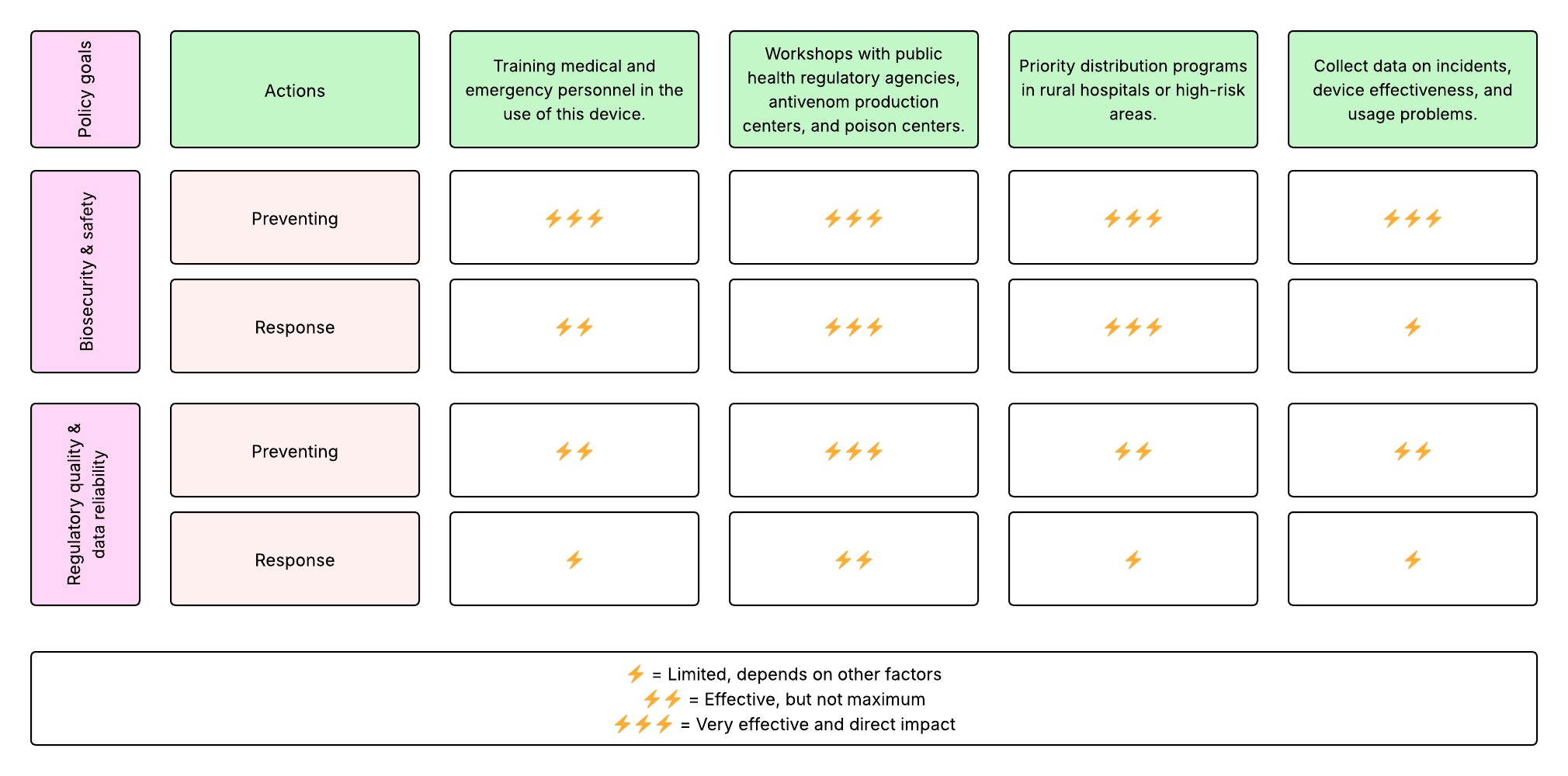

5. Last, drawing upon this scoring, describe which governance option, or combination of options, you would prioritize, and why. Outline any trade-offs you considered as well as assumptions and uncertainties. For this, you can choose one or more relevant audiences for your recommendation, which could range from the very local (e.g. to MIT leadership or Cambridge Mayoral Office) to the national (e.g. to President Biden or the head of a Federal Agency) to the international (e.g. to the United Nations Office of the Secretary-General, or the leadership of a multinational firm or industry consortia). These could also be one of the “actor” groups in your matrix.
Based on the scoring of each governance action, these options ranked highest. Consequently, they would be prioritized:
- Workshops with public health regulatory agencies, antivenom production centers, and poison centers.
- Priority distribution programs in rural hospitals or high-risk areas.
These actions have a direct influence on the two governance policies:

The priority distribution programs ensure that populations in high-risk areas have access to this device first, as they are the people who need it most. However, it is important to recognize that these priority programs require funding from regulatory agencies, hospitals, and health departments in order to function properly and avoid, for example, delays because of weather conditions or areas of difficult access, especially communities in middle- and low-income countries where there are some paramilitary groups.
As well, this is relevant for the medical care team in order to learn how to access, use, and interpret the results of the device. Regarding this idea, the workshops will require interest from researchers, the population, health care teams, emergency teams, and paramilitary groups. Additionally, they will require the availability of time to develop the workshops and meetings to monitor the continuing evaluation of the device and its use in the community.
The prioritization of these two actions assumes that all resources will be available consistently, which is uncertain, especially in remote areas. This is why it is important to consider these actions as a whole and not as separate actions. If there is no interest in promoting the device from regulatory agencies, antivenom production centers, and poison centers, it will be difficult for populations in remote areas to begin using the device. These two combined ideas will promote the safe use of the device and save lives in emergencies because it allows the identification of the type of toxin affecting a person after a bite or sting. Moreover, the medical team will provide adequate medical attention.
The target population for this recommendation is national health ministries, regulatory agencies, hospitals, and emergency teams responsible for device distribution and training.


Warm-up Questions for Week 2
In preparation for Week 2’s lecture on “DNA Read, Write, and Edit," please review these materials:
Lecture 2 slides as posted below. The associated papers that are referenced in those slides. In addition, answer these questions in each faculty member’s section:
Homework Questions from Professor Jacobson:
1. Nature’s machinery for copying DNA is called polymerase. What is the error rate of polymerase? How does this compare to the length of the human genome? How does biology deal with that discrepancy?
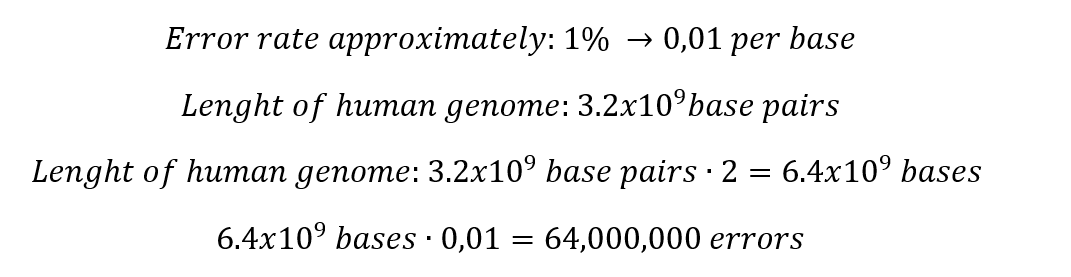
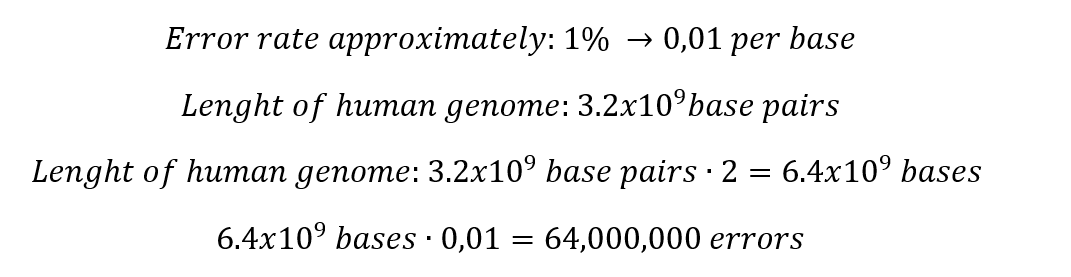
Which means that in the total length of the human genome, the polymerase fails in only 64 million bases. Indeed, this situation generates some discrepancy, but it is important to consider that 99% of the human genome is copied successfully. When some mismatches occur, the polymerase has several ways to correct these mistakes.
- 3'-exonuclease activity
- Mismatch repair:Fixes mistakes in DNA after replication or transcription, a repair mechanism for endogenous damage
- Base excision: This method works by excising a single damaged base or nucleotide
- Nucleotide excision:Use the mechanism Cut and patch
2. How many different ways are there to code (DNA nucleotide code) for an average human protein? In practice what are some of the reasons that all of these different codes don’t work to code for the protein of interest?
In the present day, we recognize approximately 500 amino acids; however, only 20 are considered in the Protein synthesis. Some scientists believe that this occurs due to a matter of evolution; they explain that evolution prioritized modifications that provided advantages to survive.
In the beginning of the process, there are 4 nucleotides in DNA: A, T, C, and G, A, U, C, and G in RNA, these for nucleotides form codons of three nucleotides each, which means that:
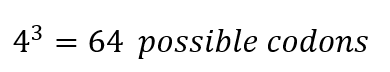
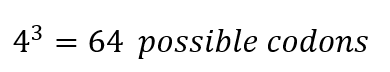
From these 64 codons, 61 correspond to amino acids for protein synthesis, and 3 are used as stop signals in the process. Which means that initially we have 20 elements to correspond to 61 options, consequently there are several codons which codify for 1 amino acid. Indeed, to identify how many ways there might be to code 1 protein, it is necessary to know the number of amino acids in the protein.
For example, the Hemoglobin has 146 amino acids in one chain; basically, the calculation would be done like this.
This is the theory of degeneracy in the genetic code. This theory suggests that the unequal distribution of 64 codons would be caused by nature to maintain stable C : H ratios. Although there are several ways to code a protein, not all of them work properly; this is because, during the protein synthesis process, there are a variety of codons more commonly used than others.
This might cause a problem when the process tries to use codons used rarely, as it will be difficult for the ribosomes decode these types of codons. This theory is known as Codon usage bias.
Homework Questions from Dr. LeProust:
1. What’s the most commonly used method for oligo synthesis currently?
- Solid-phase phosphoramidite synthesis
It occurs on a solid support held between filters, in columns that allow all reagents and solvents to pass through freely.
Principal component: Solid supports
These are insoluble particles of 50-200 μm in diameter; the oligonucleotide makes bonds with these supports during the synthesis process.
The two most common materials used in solid supports are Controlled-pore glass (CPG) and Polystyrene (PS). Both methods work perfectly when synthesizing oligonucleotides up to 150 bases in length. After that limit, the efficiency rate decreases. One nucleotide is added per synthesis cycle; the whole process includes approximately 14 steps. This is because the growing oligonucleotide blocks the pores, which means that the diffusion of reagents will decrease. Additionally, in the case of PS with oligonucleotides longer than 40 bases, the efficiency rate will decrease because long chains generate steric hindrance.
Advantages:
- Occurs in columns that allow reagents and solvents to pass through freely
- Quick completion
- No purification is required
- The whole process might be automated
2. Why is it difficult to make oligos longer than 200nt via direct synthesis?
After reaching the limit, we can identify the principal disadvantage of the process, which is that the growing oligonucleotide blocks the pores, and long chains generate steric hindrance, which means that the diffusion of reagents will decrease.
The efficiency of the process is approximately 95.6% per 10 base chain length, which means that if you have a chain of 100nt, and you try to add, for example, 10 more bases, approximately 4 bases of the chain might fail, leaving the chain with 96nt. Imagine that you want to continue the process: you try to add 10 more bases, but again, probably 4 bases will be stuck, which leaves you with a chain of approximately 92 bases.
This is what scientists call coupling efficiency. At the end of the process, a chain of 200nt will have an efficiency rate of 36.9 %, which is less than half.
3. Why can’t you make a 2000bp gene via direct oligo synthesis?
A gene of 2000 bp has more than 200nt, which means that if the efficiency rate is less than half with just 200nt, then the efficiency rate of a gene of that length will be close to 0%.
Homework Question from George Church: Choose ONE of the following three questions to answer; and please cite AI prompts or paper citations used, if any.
1. [Using Google & Prof. Church’s slide #4] What are the 10 essential amino acids in all animals and how does this affect your view of the “Lysine Contingency”?
10 Essential amino acids
- Histidine (His)
- Isoleucine (Ile)
- Leucine (Leu)
- Lysine (Lys)
- Methionine (Met)
- Phenylalanine (Phe)
- Threonine (Thr)
- Tryptophan (Trp)
- Valine (Arg)
Lysine Contingency:
Lysine is the first limiting amino acid, which means that it is the amino acid present in food in the lowest quantity, compared to the amount needed by the body. In short, the quantity of lysine present in foods is less than the quantity required. However, we talk about lysine contingency even though there are nine other essential amino acids, because lysine has a fundamental role in the proper functioning of the body.
- Acts as a substrate for post-translational modifications and the synthesis of a variety of substances used and produced by the body to work properly
- Fundamental to adequate muscle performance
- Possible Herpes Simplex Infections Therapy
- Modulation of Immune and Cardiovascular Functions
- Cancer Therapy
- Elimination of Harmful Substances
- Osteoporosis Therapy
- Wound Healing
When we detail all these uses of lysine, it is possible to notice that since this amino acid is extremely relevant, its availability in the diet is low. Therefore, it makes sense to establish it as a principal amino acid and consider its importance as an advantage in science, for example, developing an organism that depends mostly on lysine; this might be a way of controlling the activity of this organism. Other applications could be developing new therapies to treat diseases or even indicators of diseases.
References
- Tasoulis, T., & Isbister, G. (2017). A review and database of snake venom proteomes. Toxins, 9(9), 290. https://doi.org/10.3390/toxins9090290 Alonso, L. L., Slagboom, J., Casewell, N. R., Samanipour, S., & Kool, J. (2025). Categorization and Characterization of Snake Venom Variability through Intact Toxin Analysis by Mass Spectrometry. Journal of Proteome Research, 24(3), 1329–1341. https://doi.org/10.1021/acs.jproteome.4c00923
- Miller, E. (n.d.). GeNotes. GeNotes. https://www.genomicseducation.hee.nhs.uk/genotes/knowledge-hub/genome/
- Marteijn, J. A., Lans, H., Vermeulen, W., & Hoeijmakers, J. H. J. (2014). Understanding nucleotide excision repair and its roles in cancer and ageing. Nature Reviews Molecular Cell Biology, 15(7), 465–481. https://doi.org/10.1038/nrm3822
- Behura, S. K., & Severson, D. W. (2012). Codon usage bias: causative factors, quantification methods and genome‐wide patterns: with emphasis on insect genomes. Biological Reviews/Biological Reviews of the Cambridge Philosophical Society, 88(1), 49–61. https://doi.org/10.1111/j.1469-185x.2012.00242.x
- On the origin of degeneracy in the genetic code. (2019, October). The Royal Society Publishing. https://royalsocietypublishing.org/rsfs/article/9/6/20190038/35070/On-the-origin-of-degeneracy-in-the-genetic
- Ye, S., & Lehmann, J. (2022). Genetic code degeneracy is established by the decoding center of the ribosome. Nucleic Acids Research, 50(7), 4113–4126. https://doi.org/10.1093/nar/gkac171
- Qian, Y., Zhang, R., Jiang, X., & Wu, G. (2021). The constraints between amino acids influence the unequal distribution of codons and protein sequence evolution. Royal Society Open Science, 8(6), 201852. https://doi.org/10.1098/rsos.201852
- ATDBio - Nucleic Acids Book - Chapter 5: Solid-phase oligonucleotide synthesis. (n.d.). ATDBio - Nucleic Acids Book. https://atdbio.com/nucleic-acids-book/Solid-phase-oligonucleotide-synthesis
- Holeček, M. (2025). Lysine: sources, metabolism, physiological importance, and use as a supplement. International Journal of Molecular Sciences, 26(18), 8791. https://doi.org/10.3390/ijms26188791
- Lopez, M. J., & Mohiuddin, S. S. (2024, April 30). Biochemistry, essential amino acids. StatPearls - NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK557845/
- MMDB. (n.d.). 2H35: Solution structure of Human normal adult hemoglobin. https://www.ncbi.nlm.nih.gov/Structure/pdb/2H35