Week 7 HW: Genetic Circuits Part II: Neuromorphic Circuits
Assignment Part 1: Intracellular Artificial Neural Networks (IANNs)
1. What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions?
Intracellular Artificial Neural Networks (IANNs) transcend the limitations of binary Boolean logic by enabling analog and graded signal processing within a cell. While traditional genetic gates are restricted to “ON/OFF” states, IANNs can integrate multiple continuous environmental inputs and assign them specific “weights,” allowing the cell to make nuanced decisions based on a threshold of combined signals. This analog capability is particularly superior for pattern recognition and processing complex biomarkers, as it mimics natural biological decision-making more closely than rigid digital circuits. Furthermore, IANNs can often achieve high levels of computational complexity with fewer genetic parts, as they leverage the inherent non-linearities of biochemical reactions as natural “activation functions,” thereby reducing the metabolic burden on the host organism compared to massive, multi-gate Boolean architectures.
2. Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
A highly useful application for an IANN is a “smart” diagnostic classifier designed to trigger programmed cell death (apoptosis) only upon detecting a specific signature of multiple microRNA (miRNA) biomarkers associated with a particular cancer subtype. In this system, the IANN inputs would be the varying concentrations of intracellular miRNAs, which are weighted based on their diagnostic significance; the output would be the expression of a pro-apoptotic protein like Bax once the weighted sum of inputs exceeds a safety threshold. However, this application faces significant limitations, such as “leaky” expression where the circuit might trigger accidentally due to molecular noise, potentially killing healthy cells. Additionally, the metabolic load required to maintain the synthetic “neurons” can stress the cell, and the current scarcity of orthogonal (non-interfering) biological parts makes it difficult to scale these networks into deep, multi-layer architectures without cross-talk.
3. Draw a diagram for an intracellular multilayer perceptron where layer 1 outputs an endoribonuclease that regulates a fluorescent protein output in layer 2.
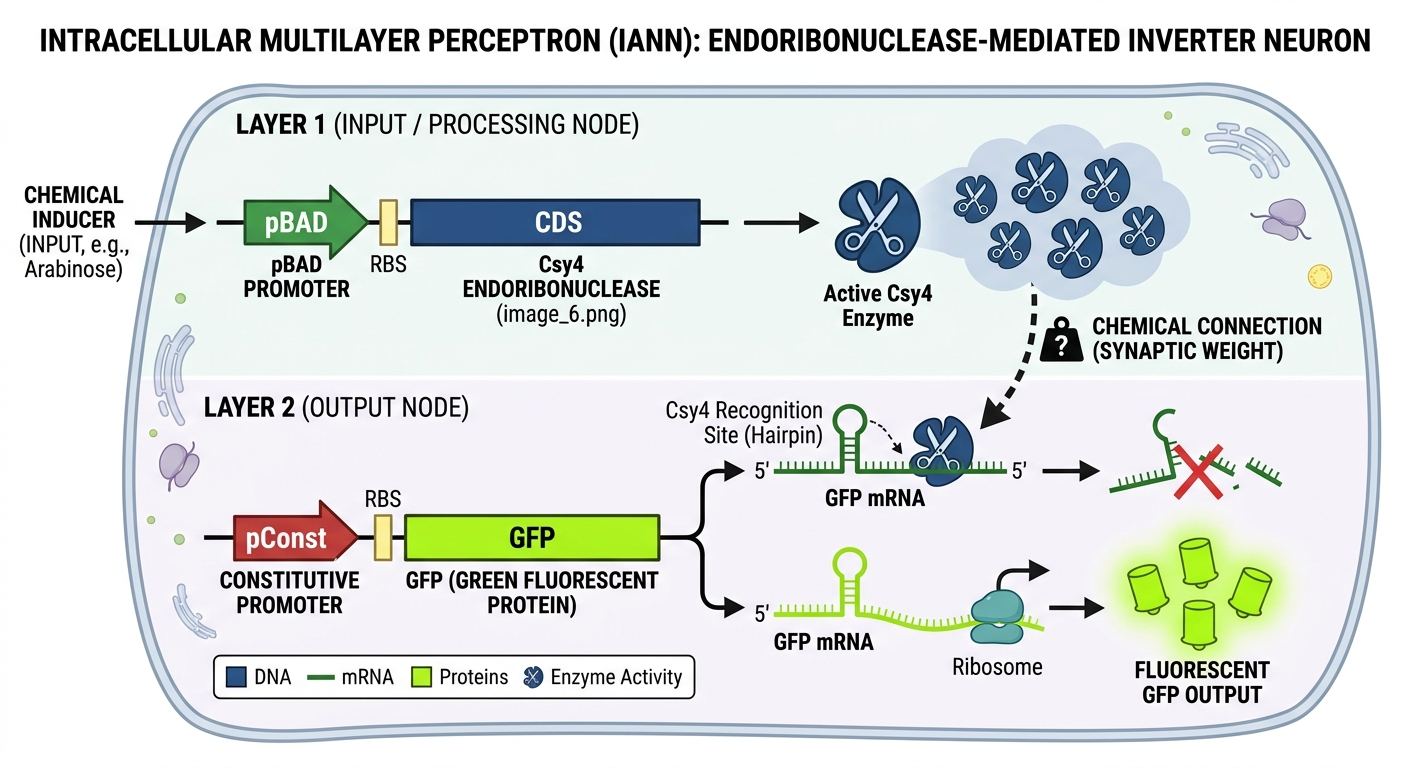
 I generated this diagram with Gemini.
This diagram illustrates a two-layer biological computer where a cell “calculates” a response to its environment. In Layer 1, the cell senses an external input (like a chemical inducer), which acts as a switch to produce Csy4 endoribonuclease enzymes (the “scissors”). These enzymes represent the processed signal traveling to the next stage. The efficiency of this enzyme’s action acts as a synaptic weight, determining how strongly the signal is passed forward. In Layer 2, the cell is constantly trying to produce a Green Fluorescent Protein (GFP), but the “scissors” from the first layer recognize and cut the GFP’s instruction manual (the mRNA). This results in an inversion logic: if the input is high, the scissors destroy the output, and the cell stays dark; if the input is low, the scissors aren’t made, and the cell glows bright green.
I generated this diagram with Gemini.
This diagram illustrates a two-layer biological computer where a cell “calculates” a response to its environment. In Layer 1, the cell senses an external input (like a chemical inducer), which acts as a switch to produce Csy4 endoribonuclease enzymes (the “scissors”). These enzymes represent the processed signal traveling to the next stage. The efficiency of this enzyme’s action acts as a synaptic weight, determining how strongly the signal is passed forward. In Layer 2, the cell is constantly trying to produce a Green Fluorescent Protein (GFP), but the “scissors” from the first layer recognize and cut the GFP’s instruction manual (the mRNA). This results in an inversion logic: if the input is high, the scissors destroy the output, and the cell stays dark; if the input is low, the scissors aren’t made, and the cell glows bright green.
Assignment Part 2: Fungal Materials
1. What are some examples of existing fungal materials and what are they used for? What are their advantages and disadvantages over traditional counterparts?
Fungal materials, primarily mycelium composites, are grown by inoculating agricultural waste (like hemp or wood chips) with fungal spores. The mycelium acts as a natural biological glue, binding the substrate into solid shapes. Key examples include biodegradable packaging (an alternative to Styrofoam), myco-bricks for sustainable construction insulation, and fungal leather (like Mylo) used in the fashion industry as a vegan alternative to animal hides. The primary advantage of fungal materials is their sustainability; they are carbon-negative, fire-resistant, and fully compostable, whereas traditional plastics and leathers rely on petrochemicals or high-energy livestock farming. However, they face disadvantages in structural consistency and water resistance. While a plastic brick has standardized strength, a grown myco-brick can vary based on the “diet” of the fungi, and many fungal materials tend to absorb moisture, which can lead to premature degradation if not properly sealed.
2. What might you want to genetically engineer fungi to do and why? What are the advantages of doing synthetic biology in fungi as opposed to bacteria?
We can genetically engineer fungi to create “living materials” with functional properties. For example, engineering a fungal brick to be self-healing would allow the material to remain dormant until a crack appears, at which point moisture triggers the fungi to grow and “fill” the gap. Other goals include bioluminescent architecture for zero-electricity lighting or “sensing” materials that change color when they detect toxic pollutants in the air through embedded genetic biosensors. Fungi offer several biological advantages over bacteria for material science. As multicellular eukaryotes, they grow in long, branching filaments called hyphae, allowing them to create massive, physically interconnected 3D structures that single-celled bacteria cannot form. Additionally, fungi are “professional secretors”; they are naturally evolved to pump huge amounts of enzymes into their environment, making them superior to bacteria for industrial-scale production of complex proteins and the breakdown of tough environmental waste.