Projects
Final projects:
- Nature’s Latent Image is a project which explores the possibility of using chlorophyll and light-harvesting proteins a novel photographic system to substitute toxic silver in current analog photography media.




Analog photography has been experiencing a growing revival and with it a growing ecological concern, especially regarding the impacts of its “magical” component — silver halides. Much of the movement of trying to address the environmental impact of analogue film has fallen on individual artists and researchers, by trying to mitigate the consequences of silver. However, despite the efforts of exploring plant-based developers, and darkroom procedures to prevent damaging disposal of silver contaminated solutions, (extremely toxic for the environment affecting primarily microbial life) we are still left with the need to use this toxic metal in lack of any other option for analog camera photography.
Nature’s Latent Image is a project which explores the possibility of using chlorophyll and light-harvesting proteins a novel photographic system to substitute toxic silver in current analog photography media, with the ultimate objective of developing working photographic emulsions which can be applied to film rolls, paper, etc.
This hypothesis is based on personal experiments that show that chlorophyll, when exposed to light, rapidly degrades into derivatives— pheophytin/pheophorbide that have a porphyrin type ring which (without the central magnesium ion) has the capacity of chelating iron ions. Therefore, chlorophyll can be used to create positive latent images (acting as light-sensitive agent) which can be developed with iron that forms the final negative image (acting as density builder).
Chlorophyll is highly prone to degradation, particularly through photooxidation, where light excitation leads to the formation of reactive oxygen species (ROS) such as singlet oxygen. These ROS can oxidize chlorophyll and nearby molecules, resulting in localized propagation of degradation. In addition, chlorophyll is also sensitive to air oxidation and environmental conditions, especially when not stabilized within a protein complex. And so, the main focus of this research project is understanding the influence of chlorophyll-binding proteins in order to maximize this pigment’s properties and stabilize it so it doesn’t degrade under unwanted situations.
Chlorophyll-binding proteins are here explored as a way to aggregate chlorophyll molecules and amplify their potential as a novel photographic substance, by creating matrixes of organized chlorophyll light-sensitive domains which should improve light absorption and allow for a controlled degradation in order to obtain photographic exposure speeds, stable latent images with good contrast and resolution, which can be later developed.
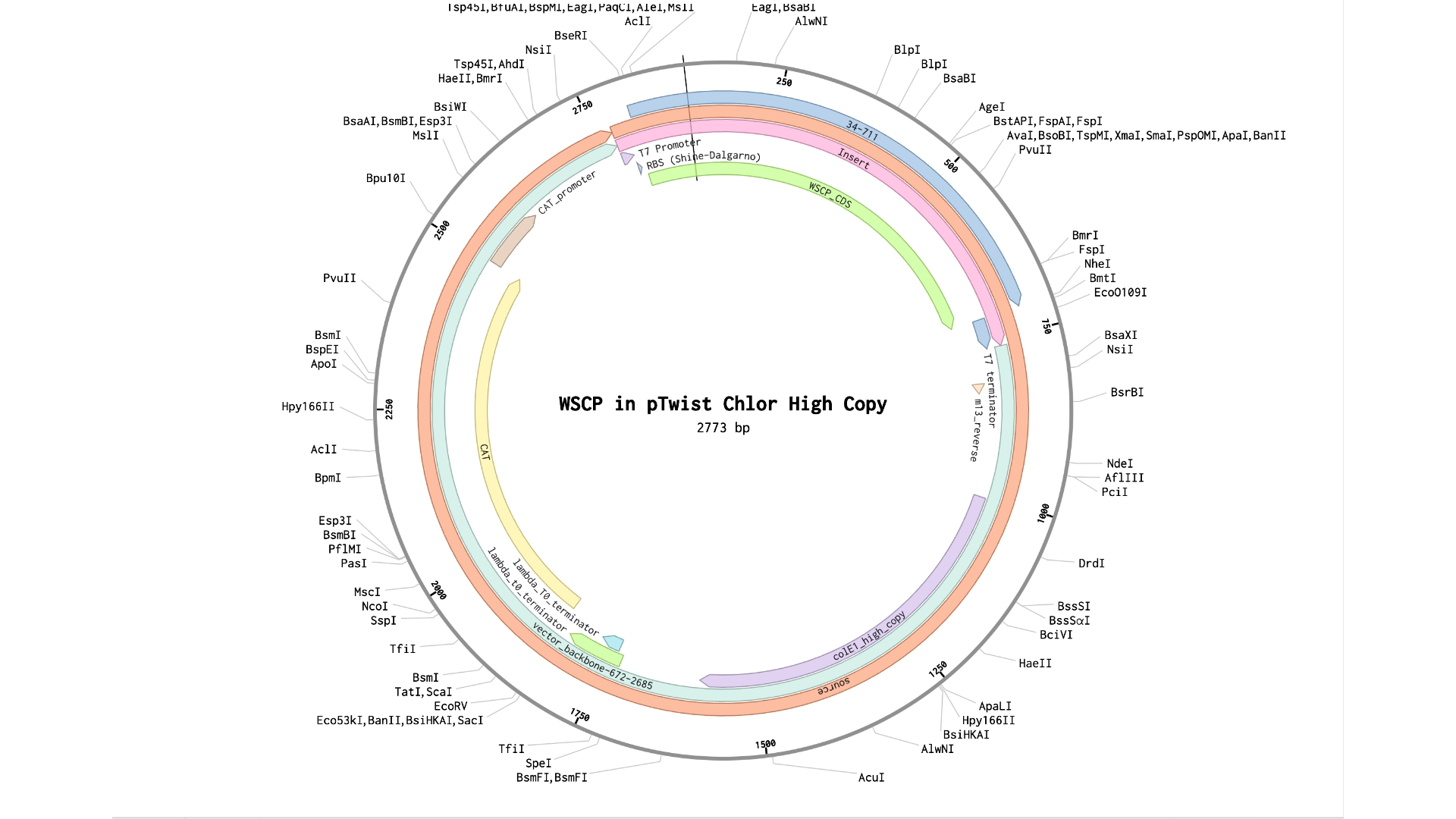
In order to achieve this project’s objectives, water soluble chlorophyll-binding proteins (WSCPs), CauWSCPs from Brassica oleracea, will be expressed to understand their impact on chlorophyll’s stability, light-sensitive capacity, degradation rate and ability to chelate iron. Another challenge rests on finding the right polymer in which to embed the protein-chlorophyll emulsion as to be able to achieve good experimental results. In further experiments, optimized chlorophyll-binding proteins could be design, if natural occurring ones do not meet photographic needs. Thinking in terms of scalability it would be interesting to synthesize these chlorophyll light-sensitive domains with cyanobacteria with the objective of having a renewable source to produce these emulsions. Finally, the ultimate step would be to develop a fully working photographic emulsion which could be applied to film rolls, paper, etc.
The first experimental approaches (as to achieve Aim 1 of this project) will start with the extraction and purification of chlorophyll from a plant source Urtica dioica (Nettles), as they are abundant and have high concentrations of Chlorophyll. E.coli plasmids for the expression of WSCPs will be design and expressed using an E.coli cell-free system where the purified chlorophyll will be used as co-folding factor. The purified chlorophyll-protein will be embedded into a polymer either chitosan based or algae based like agar to allow for visual tests of the formation of latent images and reaction with iron.


Aim 1 is based on further understanding chlorophyll-binding proteins and their effect on chlorophyll from a photographic point of view. These proteins seem to be the biological way to organize and make the most use of this photosensitive pigment.
Some challenges that are comprised in this aim are: understanding to what extent the organization of the chlorophyll through WSCPs is favorable for photographic purposes; understand if iron will still react with chlorophyll if it is bonded to WSCPs; and stopping chlorophyll degradation by air oxidation which might be aided by being bond to proteins and by being embedded into the right polymer;
To achieve these goals
Use Twist clonal genes tool
Ginkgo Cell Free Protein Expression Validation automation workflow — Gel imaging of protein expression
This will allow for a range of photostability and controlled degradation, since the presence of the pythol tail in WSCP chlorophyll-protein complexes drastically increases the photostability of chlorophyll according to Agostini et al., 2017
Analyse absorprion spectrum of proteins + chlorophyll vs pure chlorophyll
Embed both proteins + chlorophyll and pure chlorophyll in polymer to and test one against another in light sensitivity and reaction with iron
Designing an optimized version of WSCPs so it better suits photographic objectives, if needed. These adjustments are yet to be determined and rest on the experimental data from Aim 1.
After having understood the mechanisms of chlorophyll and WSCPs, develop a system where cyanobacteria, which already have both the mechanisms to produce chlorophyll and the proteins in question, could be used, instead of always having to resort to cell-free synthesis, as they would be a renewable source for scalability
The visionary aim of this project would be to develop fully working emulsions that could be applied to the fabrication of film rolls and paper, as well as creating Open-source low-tech protocols to produce chlorophyll-based emulsions so that the experimental analog photography community could expand on the findings of this research.
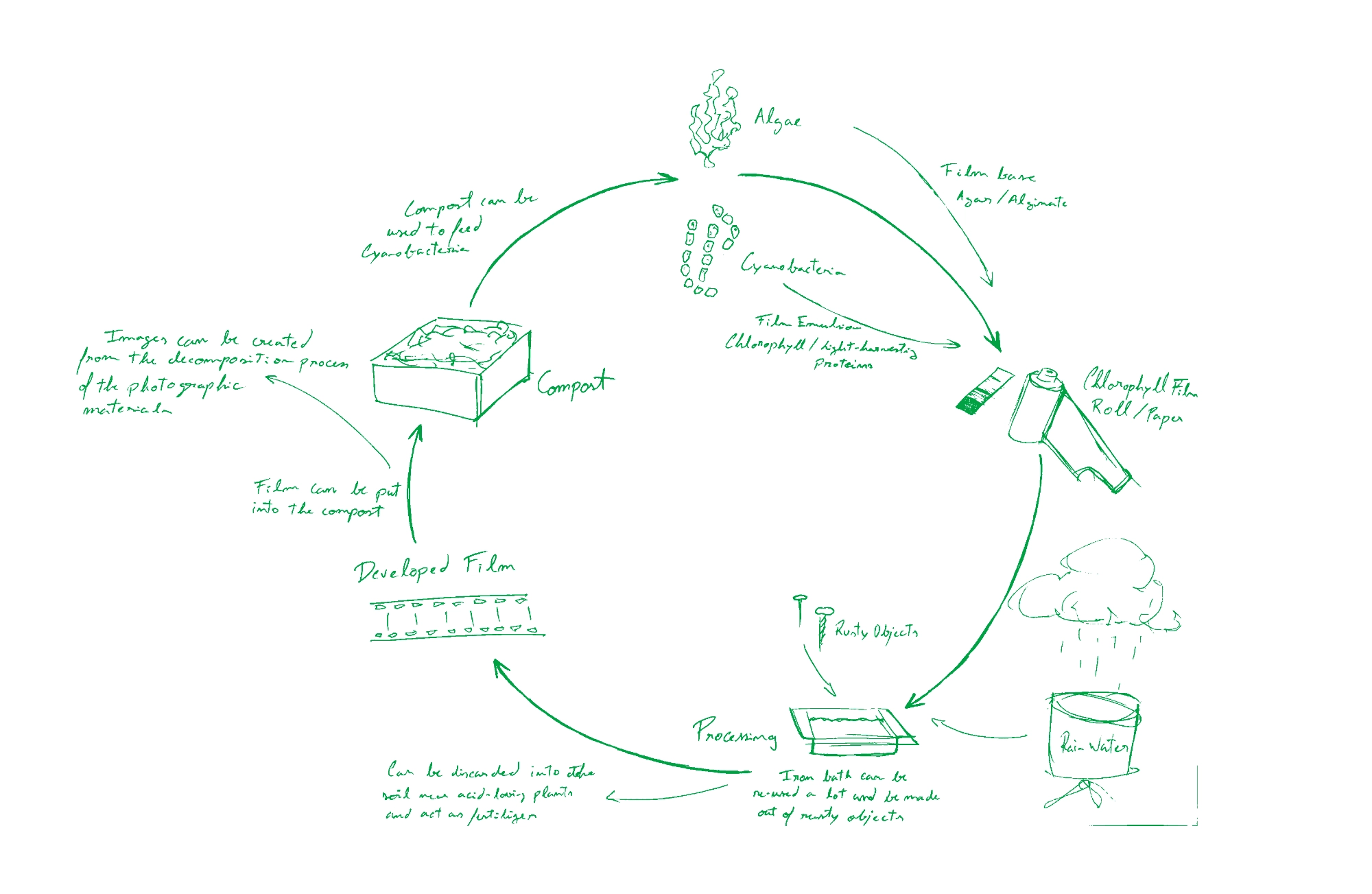

In the diagram above I drew a conceptual cycle in which this new media could participate in. It would open the possibility of a photographic practice completely compatible with the environment and new ways of producing images could be born, for example, through the partial decomposition of the photographic materials themselves, allowing for a direct intervention of other-than-human beings like bacteria, fungi and plants on the photographic medium.
This would enable an ecologically involved photographic practice that doesn’t rest on an active degradation of the ecosystems from the extraction of silver and its processing to the use of film rolls and other photographic materials and the darkroom waste they produce and their hazardous disposal.
Photography field references:
Biotechnology references:
1. Briefly summarize two peer-reviewed research citations relevant to your research
Experimental photography has increasingly foregrounded the material and ecological implications of its processes. In Blue Mud: Entangled Geologies and Lives of Photographic Silver, Alice Cazenave reframes photography as an extractive practice, entangled with mining, toxicity, and planetary infrastructures. Similarly, The Ecology of Grain exposes the environmental and animal-derived dependencies of gelatin-based film, while Andrés Pardo’s Back to Basics Vol. 1 and 2 explore alternative and low-toxicity processes. However, while these works critically examine photographic materials and propose process-based alternatives, they do not replace the reliance on silver as the core photosensitive element.
In parallel, research in biotechnology offers insight into chlorophyll as a light-sensitive molecule. Studies such as An unusual role for the phytyl chains in the photoprotection of the chlorophylls bound to Water-Soluble Chlorophyll-binding Proteins demonstrate that chlorophyll-binding proteins (WSCPs) can stabilize chlorophyll and protect it from degradation. Additionally, Fine tuning of chlorophyll spectra by protein-induced ring deformation shows that protein environments can modulate chlorophyll’s optical properties, suggesting that its behavior as a photosensitive material is not fixed but can be engineered.
The gap this project addresses lies at the intersection of these fields. While experimental photography has critically engaged with the ecological cost of its materials, it has yet to fully develop a non-toxic, non-silver-based photosensitive system. Conversely, scientific research on chlorophyll and WSCPs has not explored their potential within image-forming processes. This project proposes to bridge this gap by investigating chlorophyll as an alternative photosensitive agent, combined with plant-based polymers as an emulsion matrix, aiming to develop a biologically derived photographic process that replaces both silver and animal-based materials.
2. Explain how your project is novel or innovative. (Minimum 3 sentences.)
Analog photography has always stood in a paradox with ecology, while being the media many use through artistic practices to document and explore the problem, it actively contributes to it through its materiality based on toxic silver and also gelatine components. This project could bring about a change in the way analog photography relates itself with the natural environment, and allow for practices that participate in ecological cycles
3. Explain why your project matters and what impact it could have
The significance of this work lies in the absence of viable, non-toxic alternatives to silver-based camera photography that retain both functional and artistic potential. By exploring biologically derived materials as active components in image formation, the project seeks to expand the possibilities of experimental photography while reducing its environmental impact.
Beyond its technical aims, the project contributes to a broader rethinking of how artistic practices can engage with ecological systems—not as subjects to be represented, but as processes to work within. It also opens a critical dialogue within the experimental ecological community on whether synthetic biology should be considered a viable tool to address environmental challenges, even within artistic contexts. If successful, this research could shift experimental photography toward more sustainable, biologically integrated methods, contributing for the artistic practice as a site for both ecological awareness and material innovation.
4. Describe the ethical implications associated with your project and identify relevant ethical principles (e.g., non-maleficence, beneficence, justice, or responsibility). (Minimum 2 paragraphs.)
This project raises ethical considerations primarily related to the use of biotechnology, material sustainability, and the environmental impact of artistic practices. By proposing chlorophyll and plant-based polymers as alternatives to toxic silver-based photographic materials, the project aligns with the principle of non-maleficence, aiming to reduce harm to ecosystems and human health. At the same time, it engages with responsibility in the use of synthetic biology, particularly in the potential development of genetically modified cyanobacteria as a production system. While the final photographic materials would not contain viable genetically modified organisms, the use of such systems during production raises questions about containment, ecological risk, and the broader normalization of biotechnology within artistic contexts. The project also seeks to contribute to more sustainable material practices, and opens a critical discussion on whether synthetic biology can be ethically integrated into ecological and artistic frameworks.
To ensure the project remains ethical, several measures should be implemented. The initial use of cell-free expression systems minimizes risk by avoiding living genetically modified organisms, while any later use of genetically modified cyanobacteria would require strict laboratory containment, controlled handling, and responsible disposal protocols. A key action is to ensure that no viable GM organisms are present in the final materials, clearly separating production from outcome.
Potential unintended consequences include the accidental release of modified organisms, or the broader risk of legitimizing biotechnological interventions without sufficient critical reflection.
Additionally, one possible incorrect assumption in this project is that chlorophyll is the most suitable photosensitive pigment for these purposes; although it has shown strong conceptual and experimental promise so far, alternative pigments or systems may prove more effective or sustainable. To address this uncertainty, the project remains open to alternative approaches and iterative testing.
Written with use of HTGAA AI Tutor