Week 7 HW: Genetic Circuits Part 2
Part 1: Intracellular Artificial Neural Networks (IANNs)
What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions?
There is currently a need for non-binary biological computing, where an input for example is analog, or we want an output to be graded, this is where Intracellular Artificial Neural Networks are useful. This means that outputs can be low, medium, high. We can manupulate the inputs/outputs as if they were signals (using low-pass, high-pass, band-pass, etc filters) and with mathematical functions, rather than strictly Boolean ON/OFF outputs. IANNs combine many inputs simultaneously, and each input has a different “weight”. They can also detect patterns that are nonlinear and create thresholds. IANNs also help with scalability, as we can include many layers (multi-layers) while dealing with boolean circuits, and gates can get very messy.
Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
A useful application of an intracellular artificial neural network (IANN) is in early cancer detection and targeted therapy. In this system, multiple intracellular biomarkers such as oncogenic mRNA levels, microRNA expression, and hypoxia signals serve as inputs. Each input produces regulatory molecules, such as endoribonucleases, which act on engineered mRNA transcripts with specific cleavage sites. These interactions allow the system to integrate signals in a weighted and nonlinear manner, producing a graded response.
The output can be either a diagnostic signal, such as fluorescence, or a therapeutic response, such as expression of a pro-apoptotic protein. The system is designed so that only a strong and specific combination of cancer-related signals produces a high output, enabling selective targeting of cancer cells while minimizing effects on healthy cells.
However, several limitations must be considered. Biological noise in gene expression can reduce reliability, while precise tuning of regulatory weights is difficult to achieve. Off-target effects of endoribonucleases may disrupt normal cellular processes, and delivering the system efficiently into target cells remains a major challenge. Additionally, the metabolic burden of maintaining complex circuits and the risk of mutations affecting stability over time further complicate implementation. Despite these challenges, IANNs offer a powerful framework for implementing complex decision-making in living cells.
Draw a diagram for an intracellular multilayer perceptron where layer 1 outputs an endoribonuclease that regulates a fluorescent protein output in layer 2.
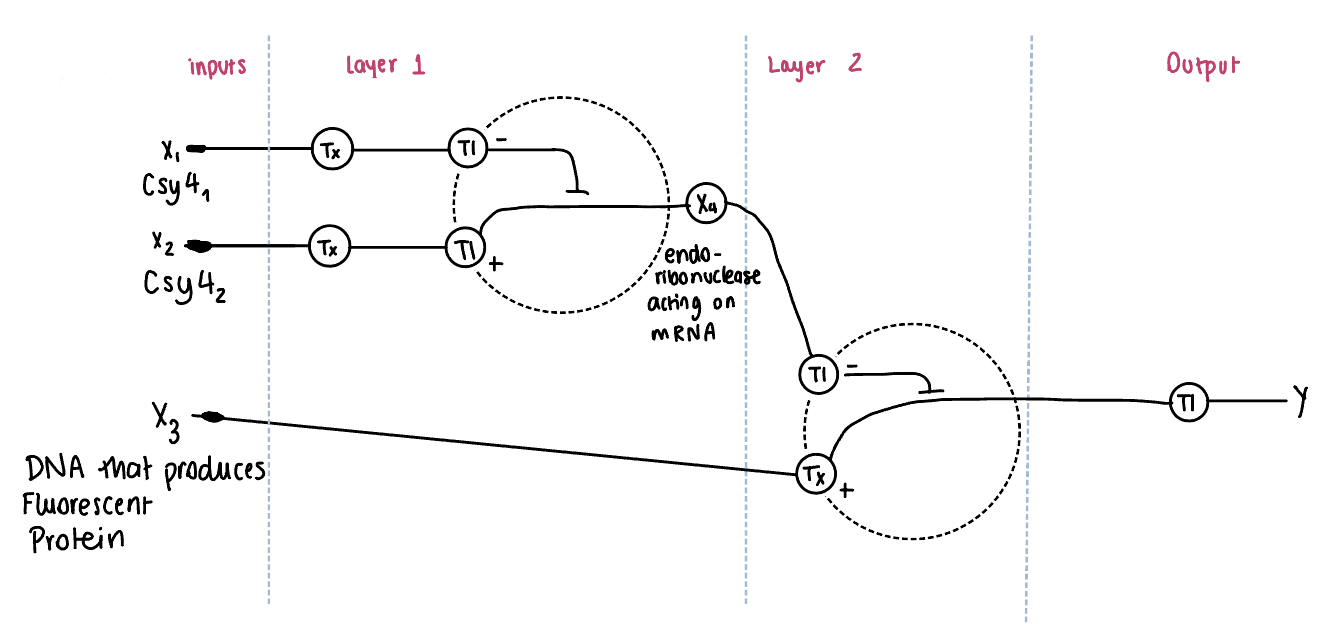

In this diagram, an intracellular multilayer perceptron composed of two regulatory layers is represented. In Layer 1, inputs X1 and X2 encode for endoribonucleases (Csy4 variants), which are produced through transcription (Tx) and translation (Tl). These endoribonucleases act on mRNA and generate an intermediate regulatory signal X4, representing the hidden layer. We need both inputs in layer one to downregulate and control the system from the difference or the sum of the inputs. This would be regulated right after the promoter and before the CDS.
In Layer 2, the intermediate regulator X4, controls the expression of a downstream gene X3, which encodes a fluorescent protein. The regulation occurs at the mRNA level. Finally, the fluorescent protein is produced through translation, resulting in the output Y.
Part 2: Fungal Materials
What are some examples of existing fungal materials and what are they used for? What are their advantages and disadvantages over traditional counterparts?
An example of existing fungal materials are Mycelium-based materials used for packaging, leather-like materials or other textiles for fashion, and bricks for construction.
Some advantages are that it is biodegradable, sustainable, customizable, and it has a low carbon footprint. In some cases, it is fire-resistant. However, some disadvantages are that they often have lower mechanical strength, are sensitive to moisture, and can be difficult to scale consistently.
What might you want to genetically engineer fungi to do and why? What are the advantages of doing synthetic biology in fungi as opposed to bacteria?
One potential application of synthetic biology in fungi is the development of self-healing, environmentally responsive building materials. In this system, fungal mycelium would be engineered to form structural materials such as bricks or insulation panels, while also incorporating genetic circuits that enable sensing and response to environmental conditions.
Inputs to the system could include the presence of environmental toxins (such as heavy metals or pollutants) or physical damage to the material. These signals would activate engineered biosensing pathways that regulate gene expression. The outputs could include the production of fluorescent proteins to signal contamination or damage, as well as the activation of growth pathways that promote self-repair of the material.
This system would allow the material to both detect and respond to changes in its environment, making it more adaptive and functional than traditional materials. However, limitations include challenges in controlling fungal growth, maintaining long-term stability, and ensuring safety when deploying genetically engineered organisms in real-world environments. Additionally, precise tuning of gene expression and scalability remain significant challenges.
Fungi are particularly well-suited for this application because they naturally form interconnected three-dimensional networks and can grow into large structures, unlike bacteria. This makes them ideal for material-based applications where both structure and function are required.