Week 5 HW: protein desing part 2
Homework — DUE BY START OF MAR 10 LECTURE
Part A: SOD1 Binder Peptide Design (From Pranam)
Introduction
Superoxide dismutase 1 (SOD1) is a cytosolic antioxidant enzyme that converts superoxide radicals into hydrogen peroxide and oxygen. In its native state, it forms a stable homodimer and binds copper and zinc.
Mutations in SOD1 cause familial Amyotrophic Lateral Sclerosis (ALS). Among them, the A4V mutation (Alanine → Valine at residue 4) leads to one of the most aggressive forms of the disease. The mutation subtly destabilizes the N-terminus, perturbs folding energetics, and promotes toxic aggregation.
Part 1: Generate Binders with PepMLM
Begin by retrieving the human SOD1 sequence from UniProt (P00441) and introducing the A4V mutation.
Using the PepMLM Colab linked from the HuggingFace PepMLM-650M template card:
Generate four 12-amino-acid peptides conditioned to the mutant SOD1 sequence.
Add the known SOD1-binding peptide, FLYRWLPSRRGG, to the generated list for comparison.
Record the puzzle scores, which indicate PepMLM’s confidence in the ligands.

sp|P00441|SODC_HUMAN Superoxide dismutase [Cu-Zn] OS=Homo sapiens OX=9606 GN=SOD1 PE=1 SV=2
MATKAVCVLKGDGPVQGIINFEQKESNGPVKVWGSIKGLTEGLHGFHVHE
FGDNTAGCTSAGPHFNPLSRKHGGPKDEERHVGDLGNVTADKDGVADVSI
EDSVISLSGDHCIIGRTLVVHEKADDLGKGGNEESTKTGNAGSRLACGVI
GIAQ
Part 2: Evaluate Binders with AlphaFold3
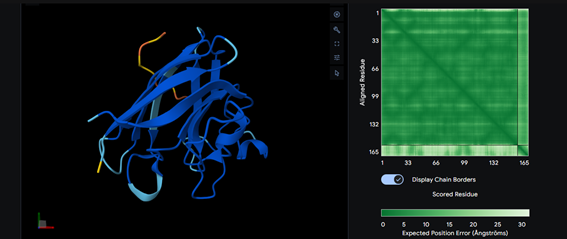
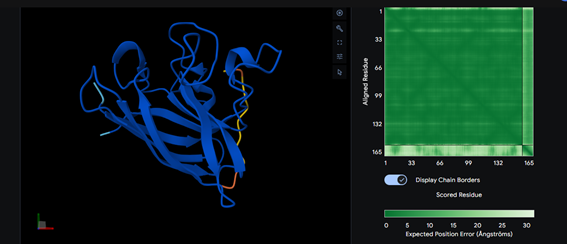
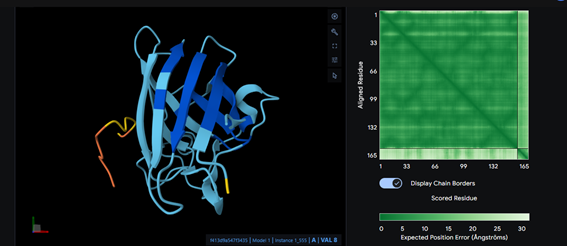
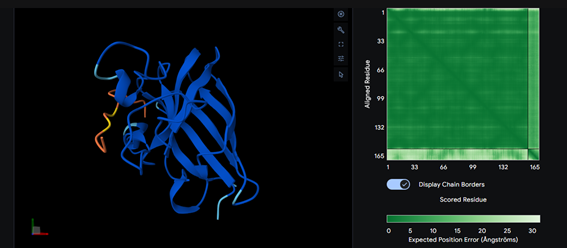
Part 3: Evaluate Properties of Generated Peptides in the PeptiVerse
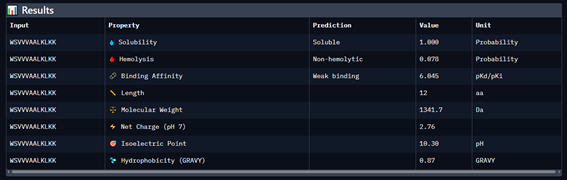
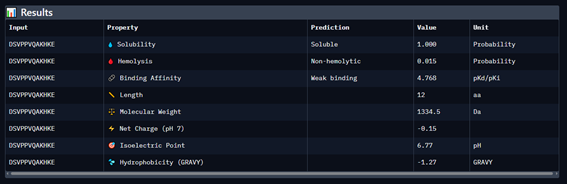
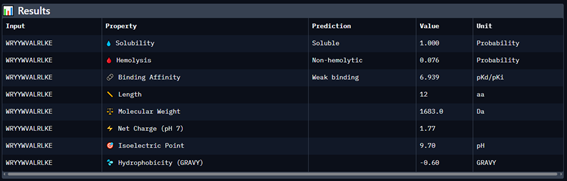
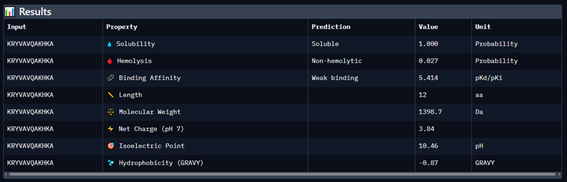
Part 4: Generate Optimized Peptides with moPPIt
Part B: BRD4 Drug Discovery Platform Tutorial (Gabriele)

Part C: Final Project: L-Protein Mutants
The proposed mutations in the Bacteriophage MS2 lysis protein are generally biochemically consistent and plausible for improving stability without compromising function. The Q→E and S→T changes are conservative substitutions between polar residues that could favor electrostatic interactions or slightly increase local stability. The hydrophobic L→I and V→I mutations are particularly reasonable within the transmembrane domain, as they maintain the hydrophobicity necessary for membrane insertion and may improve helix packing. The F→Y substitution preserves aromaticity but introduces a hydroxyl group that could facilitate interactions at the membrane-water interface. Overall, these mutations do not significantly alter the length or overall hydrophobicity of the transmembrane segment, which is important because the L protein exerts its function by interfering with the MurJ lipid flippase II in Escherichia coli; therefore, the proposed changes are compatible with maintaining activity while potentially improving folding or structural stability.
Variants with more “aggressive” mutations (increasing hydrophobicity or propensity for helix formation in the transmembrane region):
- S -> L (increased hydrophobicity within the TM domain)
METRFPQQSQQTPASTNRRRPFKHEDYPCRRQQRSLTLYVLIFLAIFLSKFTNQLLLSLLEAVIRTVTTLQQLLT - T -> L (reinforces the hydrophobic character of the propeller)
METRFPQQSQQTPASTNRRRPFKHEDYPCRRQQRSSTLYVLIFLAIFLSKFTNQLLLSLLEAVIRLVTTLQQLLT - A -> L (greater transmembrane helix stability)
METRFPQQSQQTPASTNRRRPFKHEDYPCRRQQRSSTLYVLIFLLIFLSKFTNQLLLSLLEAVIRTVTTLQQLLT - L -> F (introduces aromatic residue into TM)
METRFPQQSQQTPASTNRRRPFKHEDYPCRRQQRSSTLYVLIFLAIFFSKFTNQLLLSLLEAVIRTVTTLQQLLT
These variants introduce less conservative changes that increase hydrophobicity, or the propensity to form α-helices, within the transmembrane domain of the Bacteriophage MS2 L protein. By replacing polar or small residues with more hydrophobic amino acids such as leucine or phenylalanine, more stable insertion into the bacterial membrane and more efficient packing of the transmembrane helix can be favored. This potentially increases the protein’s interaction with the lipid flippase II MurJ in Escherichia coli, the functional target of this lysis protein, which could intensify the inhibition of peptidoglycan precursor transport and accelerate the cell lysis process. Overall, mutations of this type aim to increase the structural stability and functional efficiency of the lysis mechanism.