Conceptual Questions
All questions were answered using the feedback of Gemini - and ChatGPT for Question 10 - as a research starting point. Questions were used as prompts with an oriented approach (what would Shuguang Zhang answer, how would you explain a 10 year old etc.). Sources were checked and prompts refined from the larger picture into details of interest, as well as when needed if the content was unclear to me.
1. How many molecules of amino acids do you take with a piece of 500 grams of meat? (on average an amino acid is ~100 Daltons)
According to Gemini, meat generally contains approx 25% (+/- 5%) of protein by weight when cooked. Thus, for this calculation, we assume that a 500g piece of meat contains 125g of protein (5x25g). By convention, 1g = 6.022 x 10 ^ 23 Da , so 125g = 7,528 x 10 ^ 25 Da.
Thus, our piece of meat contains 7,528 x 10^23 molecules of amino acids (7,528 x 10^25 /100) : a lot of work awaits pepsin, trypsin and peptidase at the burger shop 🍔
2. Why do humans eat beef/fish but do not become a cow/fish?
Although the idiom “You are what you eat” suggests food directly becomes part of us, proteins ingested during meals are not directly incorporated into the human body: rather, they are first broken down into amino acids through the digestive process and then serve as building blocks for human proteins according to the DNA code. Proteins are species-specific and their production depends on both the genetic code and environmental factors.
3. Why are there only 20 natural amino acids?
The existence of only 20 primary natural amino acids is generally explained by an evolutionary optimization. Organisms evolve to optimize the balance between energy/resource consumption and the benefits derived from biological functions. These specific building blocks were selected over 4 billion years ago for their ability to form stable, functional, and soluble proteins. They provide enough diversity to support all necessary biological functions while remaining cost-efficient and easier to handle by the cell machinery than a larger set of building blocks.
Why twenty amino acid residue types suffice(d) to support all living systems
Teaching the principle of biological optimization
4. Can you make other non-natural amino acids? Design some new amino acids.
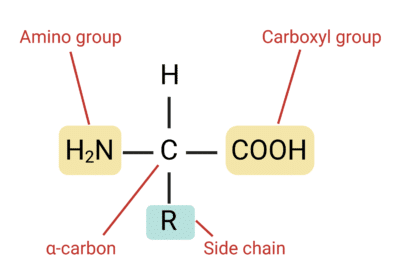
Yes, Shuguang Zhang and George Church have pushed the boundaries of molecular design by developing methods to produce non-natural amino acids. Amino acids are composed of an amino group (-NH2), a carboxyl group (-COOH) and a residue group (-R) that varies. To create new amino acids, one needs to design new residue groups. Example of new residues and how to synthesize them: SKIP for now ⏱
5. Where did amino acids come from before enzymes that make them, and before life started?
Scientists think that life originated from a “primordial soup” in deep-sea hydrothermal vents. Despite extreme heat and high pressure, these environments are teeming with life. These vents emit hot, mineral-rich fluids, creating environments where simple molecules could undergo chemical reactions to form more complex organic compounds, including amino acids. In 1952, Miller and Urey simulated early Earth conditions in an experiment and demonstrated that amino acids can form spontaneously through non-enzymatic pathways. It is also been proposed that amino acids may have an extraterrestrial origin. Amino acids have been found within meteorites that have crashed to Earth and in samples returned directly from asteroids, indicating that the chemical ingredients for life as we know it are widespread in the solar system 👽
A Short Tale of the Origin of Proteins and Ribosome Evolution
Insights into the formation and evolution of extraterrestrial amino acids from the asteroid Ryugu
6. If you make an α-helix using D-amino acids, what handedness (right or left) would you expect?
In humans, the right-handed α-helix is the most common structural arrangement in the secondary structure of proteins. These helices are composed of L-amino acids. L-amino acids and D-amino acids are stereoisomer mirror images of each other, differing in the placement of the amino group (-NH2) on the alpha-carbon (see paper given in reference below). In a Fischer projection, L-amino acids have the amino group on the left (left handed), while D-amino acids have it on the right (right handed). Thus, if building an α-helix using D-amino acids, one would expect the helix to be oriented in the opposite direction and obtain a left-handed α-helix.
Structure and Function of Proteins
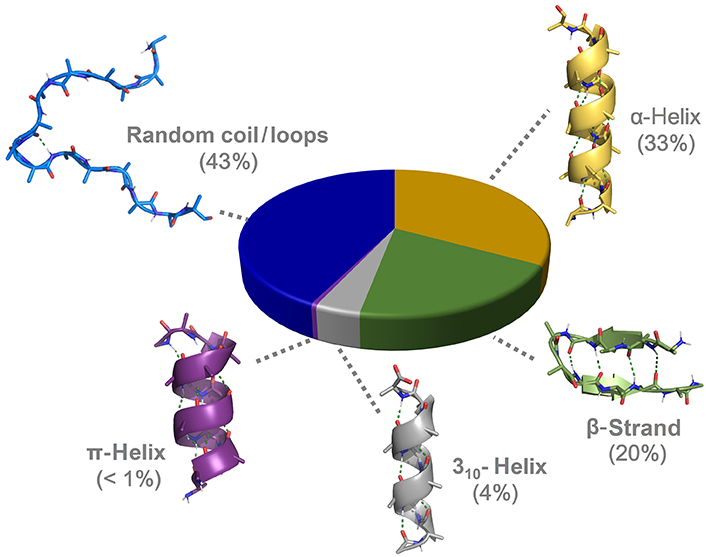
7. Can you discover additional helices in proteins?
After decades of intensive focus on the study of the α-helices, other helicoidal structures have been and keep being discovered, or rather reclassified after slowly regaining a certain relevance in protein science:
- 310 helices are typically observed as extensions of α-helices. They have been proposed to be intermediates in the folding/unfolding of the alpha helices and possibly involved in the initiation of the folding.
- π helices are evolutionarily derived by the insertion of a single residue into an α-helix and typically found near functional sites of proteins.
- Polyproline helices are mainly composed of proline polymers but can also form in sequences rich in other residues such as glycine, lysine, glutamate, and aspartate. They exist in two forms, either left-handed (PPII) or right-handed (PPI, rarer). Because of the rigidity and the restricted conformational space of the proline, the PPII has no internal hydrogen bonding and is relatively open as compared with the other helices. The PPII structure has been demonstrated to be essential to biological activities such as signal transduction, transcription, cell motility, and immune response.
- Collagen helices are formed by 3 PPII helices that twist together into a right-handed supercoil, also known as tropocollagen. They are the primary structural component of connective tissues (skin, bone, tendons, cartilage) providing high tensile strength.
- β-helices are formed by the supramolecular association of β-sheets in a helical pattern.
- γ-helices have been predicted but not observed yet in natural proteins.
8. Why are most molecular helices right-handed?
During evolution, L-amino acids were preferred for protein synthesis and main metabolism: ribosomes possess a remarkable ability for chiral selectivity and are designed to use L-amino acids for protein synthesis. This explains why most molecular helices found in nature are right-handed (see Question 6 for reference).
To go a bit further: In the early stage of amino acids discovery, scientists actually believed that L-amino acids were solely found in nature and D-amino acids are artificial products. However, with the development of analytical methods in the past decades, D-amino acids have been found in a wide variety of living organisms both in their free form and as isomeric residues in many proteins. Their various biological functions are closely relevant to human physiology and diseases, including cancer. Although not typically formed by the translation machinery, left-handed helices can still be found in nature and are typically built through post-translational modification, non-ribosomal peptide synthetases (NRPS), and the incorporation of glycine and achiral residues.
Natural Occurrence, Biological Functions, and Analysis of D-Amino Acids
d‐amino acids: new functional insights
9. Why do β-sheets tend to aggregate? What is the driving force for β-sheet aggregation?
The aggregation of β-sheets is a phenomenon of molecular self-assembly that is due to hydrophobicity and structural complementation. A β-sheet consists of β-strands that are connected laterally by at least two or three backbone hydrogen bonds, forming a generally twisted, pleated sheet. Because the structure of their edges is complementary (unpaired amino and carboxyl groups just waiting to find their match 💘), adjacent β-sheets naturally bond via their “sticky edges”. But what drives adjacent β-sheets to pile up like pancakes is linked to the amphiphilic nature of the β-sheets. Indeed, one of their sides is hydrophilic and the other is hydrophobic. In the same way that oil droplets gather together when added to water, β-sheets tend to aggregate in order to “hide” their hydrophobic sides from hydrophilic groups or aqueous environment (nucleoplasm, cytoplasm and ground substance).
The Supramolecular Chemistry of β-Sheets
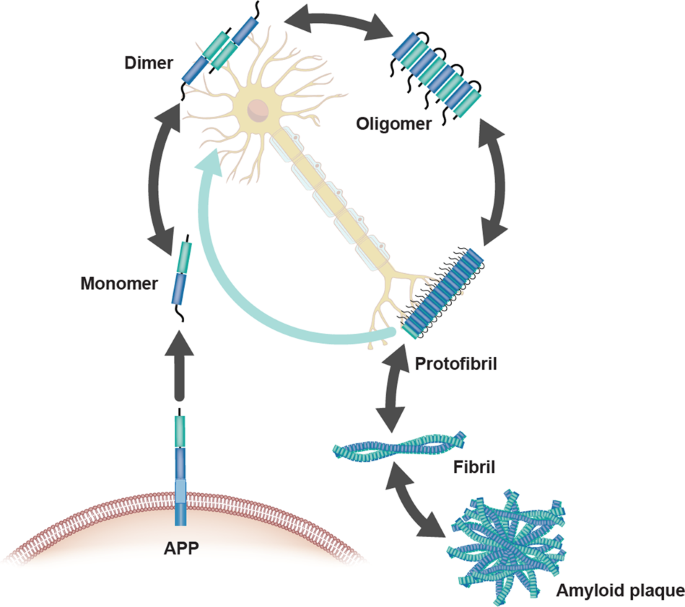
10. Why do many amyloid diseases form β-sheets? Can you use amyloid β-sheets as materials?
Amyloids have been linked to the development of various neurodegenerative diseases such as Alzheimer’s and Parkinson’s. Pathogenic amyloids form when previously healthy proteins misfold: these proteins lose their normal structure and physiological functions, and start forming fibrous deposits within and around cells that cause the progressive disruption of brain functions. Amyloid fibrils form from different proteins, each associated with a particular disease, but they all contain a distinctive dysfunctional β-sheet pattern known as cross-β spine. When the peptides misfold, they align next to each other forming extended β-sheets that present highly stable and ordered structures. Once a small cluster of misfolded peptides is formed, a nucleation effect causes the misfolding and aggregation of other adjacent peptides, leading to an accelerated amplification of the self-propagating aggregation process. Furthermore, the aggregation of cross-β-sheets leads to the creation of steric zippers that makes the amyloids resistant to enzyme degradation (proteolysis) allowing them to accumulate in tissues and organs over time.
.
The Amyloid-β Pathway in Alzheimer’s Disease
.
One man’s loss is another man’s gain… The nanocrystal properties of cross-β sheet aggregates make them suitable for the engineering of biomaterials that can better withstand thermal stress and chemical denaturation such as:
- Hydrogels and Scaffold Engineering: Due to their ability to form nanofibrils, cross-beta peptides are used to create supramolecular hydrogels for tissue engineering and regenerative medicine.
- High-Strength Biomaterials: Synthetic polymeric amyloid fibers, containing cross-beta nanocrystals, can be engineered to exhibit exceptional mechanical strength, surpassing some natural spider silk fibers with high tensile strength and toughness.
- Nanowires and Conductive Materials: Fibrils can serve as templates for creating metallic nanowires for applications in nano-electronics, such as biosensors, actuators, and memory devices.
- Nanoporous Matrix Formation: Cross-beta aggregates are used to construct highly stable, porous, and rigid materials for specialized applications.
.

Amyloid-induced mineralization: From biological systems to biomimetic materials
.
11. Design a β-sheet motif that forms a well-ordered structure.
SKIP for now ⏱