Week 7 HW: Genetic Circuits Part II
Part 1
What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions?
The key advantage of intracellular analog neural networks (IANNs) over traditional genetic circuits lies in the shift from discrete logic to continuous computation. Classical genetic circuits are typically engineered as Boolean systems: inputs are interpreted as “on” or “off”. This abstraction is convenient for engineering and design, but it is fundamentally misaligned with how biology actually operates, where signals exist as continuously varying concentrations and reaction rates.
IANNs, in contrast, operate on these continuous biochemical signals directly. Instead of forcing a threshold-based decision, where a small fluctuation can flip the output entirely, they produce graded responses. In practice, this means a cell can scale its response to stimulus intensity rather than switching abruptly, which is far more consistent with natural regulatory systems.
A second major advantage is robustness as biological systems are inherently noisy. Boolean circuits are fragile under these conditions because they rely on precise thresholding. IANNs mitigate this by distributing computation across multiple interacting components. Much like artificial neural networks, they integrate signals through weighted interactions, which effectively averages out noise and reduces the likelihood of incorrect responses.
Closely related to this is the issue of scalability. As traditional genetic circuits grow in complexity, the number of required components increases rapidly, often becoming impractical to implement in living cells. IANNs compress complexity into tunable parameters such as binding affinities and interaction strengths. This allows them to represent much more complex input–output relationships with fewer physical parts, making them more feasible for real biological systems.
Another important distinction is the type of functions each approach can represent. Boolean circuits are naturally suited for discrete decision boundaries, but they struggle with complex, nonlinear mappings across many inputs. IANNs, on the other hand, can approximate arbitrary nonlinear functions. This enables cells to perform tasks that resemble pattern recognition, such as distinguishing subtle combinations of molecular signals that define a disease state or environmental condition.
Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
Useful applications of an intracellular analog neural network (IANN) can be cell-based diagnostics and therapy of cancer, where a cell must interpret a complex molecular environment and make a precise decision about whether to release a therapeutic drug. This is a setting where Boolean genetic circuits struggle, because disease states are rarely defined by single markers or sharp thresholds; instead, they emerge from subtle, multidimensional patterns of signals,which are impractical to engineer with boolean logic gates.
- Concept
Engineering an immune cell that can distinguish between healthy tissue and tumor tissue by integrating multiple intracellular and extracellular signals. The cell evaluates a combination of cues, such as surface receptors, cytokine levels, metabolic indicators, and stress signals (pick peer reviewed literature for input adoption) and determines whether the overall pattern corresponds to a malignant state. If the pattern matches, the cell releases a therapeutic molecule; if not, it remains inactive. - Input
The inputs to the IANN would be continuous biochemical signals, for example:
- Expression levels of tumor-associated antigens
- Cytokine concentrations such as IL-6 or TGF-β in the microenvironment
- Indicators of hypoxia or altered metabolism
- Output
The output is also continuous but can be coupled to thresholded downstream actions. For example:- Low output function → no response (cell remains inactive)
- Intermediate output → mild response (e.g., secretion of signaling molecules to recruit immune cells)
- High output → full activation (e.g., cytotoxic killing, release of therapeutic proteins, or induction of apoptosis in the target cell)
- Advantages This application benefits directly from the properties of IANNs. The system must interpret noisy, overlapping biological signals and map them to a decision boundary that is not easily expressible as a simple logical rule. An IANN can approximate this complex mapping, remain robust to fluctuations, and avoid false positives that would arise from single-marker detection.
- Limitations Two main limitations apply. First, there is the implementation issue: encoding precise weights and nonlinearities in biochemical systems is difficult, as binding affinities and expression levels are not infinitely tunable and are subject to drift over time. Second, training and calibration remain a major challenge; unlike digital circuits, the correct parameter set must often be found through iterative screening or evolution, which is experimentally intensive.
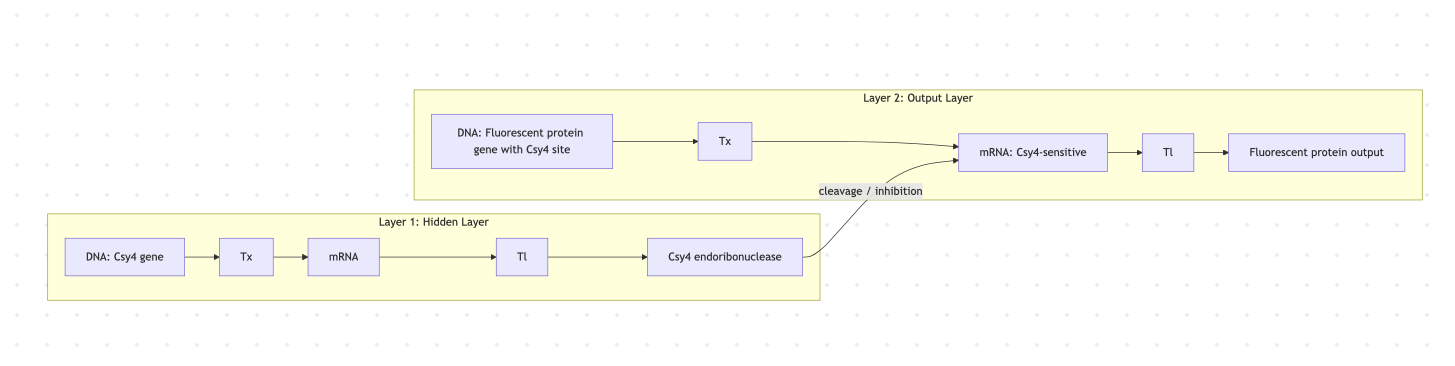
Below is a diagram depicting an intracellular single-layer perceptron where the X1 input is DNA encoding for the Csy4 endoribonuclease and the X2 input is DNA encoding for a fluorescent protein output whose mRNA is regulated by Csy4. Tx: transcription; Tl: translation.Draw a diagram for an intracellular multilayer perceptron where layer 1 outputs an endoribonuclease that regulates a fluorescent protein output in layer 2.

Part 2
What are some examples of existing fungal materials and what are they used for? What are their advantages and disadvantages over traditional counterparts?
Fungal materials usually based on mycelium (the filamentous network of fungi) are increasingly used as sustainable alternatives to plastics, foams, leather, and some construction materials.
- Mycelium packaging materials These are grown by letting mycelium bind loose biomass into a foam-like structure and are used for protective packaging (e.g., replacing Styrofoam). Their main advantage is that they are fully biodegradable, low-energy to produce, and utilize waste streams as feedstock. However, they suffer from lower mechanical consistency, reduced moisture resistance, and currently higher costs compared to petrochemical foams.
- Mycelium-based construction materials These include insulation panels, acoustic materials, and experimental bricks. They offer a very low carbon footprint, good thermal and sound insulation, and natural fire resistance. In contrast, they have much lower compressive strength than concrete or engineered wood, are sensitive to moisture, and face significant regulatory hurdles before widespread adoption in construction.
- Mycelium leather and emerging fungal textiles These materials are derived from fungal structures grown into dense mats or processed into fiber-like forms for use in fashion, upholstery, and potentially broader textile applications. They offer clear advantages, including avoiding animal use, significantly reducing environmental impact, and enabling tunable material properties during growth. Additionally, they hold promise as biodegradable alternatives to synthetic fibers produced through low-energy processes. However, they currently face several limitations: lower durability compared to high-quality leather, reliance on coatings to achieve desired performance characteristics, insufficient mechanical strength for many applications, and ongoing challenges in processing and scaling production with consistent quality.
What might you want to genetically engineer fungi to do and why? What are the advantages of doing synthetic biology in fungi as opposed to bacteria?
Fungi represent an underexploited chassis for synthetic biology. Unlike bacteria, they are eukaryotic, naturally adapted to degrade complex substrates, and already widely used in industry. This makes them particularly attractive for engineering applications that require secretion, material formation, or complex metabolic processing.
- Enhancing material properties for fungal biomaterials One compelling direction is to engineer fungi to produce mycelium-based materials with improved mechanical strength, water resistance, or functionality. This could involve modifying cell wall composition (e.g., chitin, glucans), introducing crosslinking proteins, or enabling secretion of hydrophobic compounds.
- Programming fungi for advanced biomanufacturing Fungi could be engineered to produce high-value compounds such as pharmaceuticals, enzymes, or specialty chemicals, especially those requiring post-translational modifications. Compared to bacteria, fungi (as eukaryotes) can correctly fold complex proteins, perform glycosylation, and secrete products efficiently into the medium.
- Improving degradation of complex waste streams Fungi are naturally capable of breaking down lignocellulose, plastics, and other recalcitrant materials. This enables applications in waste valorization and circular bioeconomy systems.
- Developing functional living materials Fungi could be programmed to create living materials that respond to environmental stimuli, such as self-healing construction materials or responsive insulation. This might involve integrating sensing circuits with growth or material production pathways.