Week 1 HW: Principles and Practices
“Oncological Bacteriotherapy: Engineered Siderophore Secretion and Safety Kill-Switch via NAND Logic Gates”
My project focuses on engineering a bacterial strain capable of sensing the tumor microenvironment and responding through the synthesis of siderophores (Salmochelin), integrated with a robust safety mechanism to prevent off-target effects.
1. General Objective
To induce the death of tumor cells through iron sequestration (Salmochelin), integrating spatial control logic circuits (NAND gate) so that the bacteria act exclusively under hypoxia and ultrasound conditions, and a biocontainment system (Kill-Switch) to guarantee host safety.
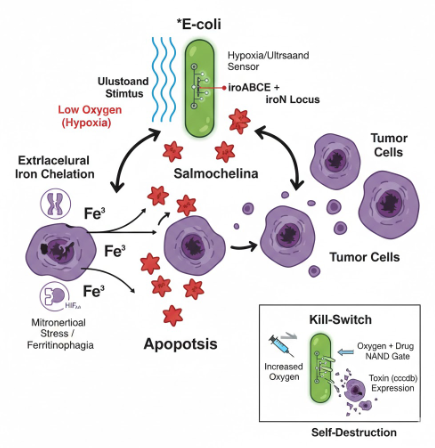

2. Experimental Design
2.1.- The effector agent: E. coli Nissle 1917 (Locus iroBCDE, iroN)
- Siderophore Selection: Release Salmochelin (glycosylated) instead of the siderophore Enterobactin (ENT), which is known to be neutralized by host Lipocalin-2 (Lcn2) (Saha et al. 2019). The use of Pyoverdine is discarded after technical analysis, as its complex biosynthetic pathway represents excessive metabolic stress for the bacteria.
- Competitive Advantage: Based on the findings of Raffatellu et al. 2010, salmochelin is a siderophore that can survive in an environment with high levels of Lcn2.
- Cytotoxic Effect (hypothetical): By depleting the labile iron pool (LIP), ferritinophagy and HIF-1α are promoted, but given the high affinity of the siderophore, mitochondrial function collapses, triggering apoptosis of cancer cells.
2.2.- Spatial Control Circuit (NAND Gate)
To avoid systemic toxicity (Pita-Grisanti et al., 2022), Salmochelin production is subject to a double de-repression. In the normal state, two repressors (LacI and TetR) block the operon. Synthesis occurs when the following conditions are met:
- Condition A (Hypoxia): The P_vgb promoter turns off → The bacteria stop producing LacI-LVA → The LacI lock degrades.
- Condition B (Ultrasound): The stimulus inactivates the P_tlpA repression system → The bacteria no longer have functional TetR-LVA → The TetR lock degrades.
- Result: The Salmochelin genes (iroBCDE, iroN) must be under a promoter with binding sites for LacI and TetR. Only when there is NO LacI (Hypoxia) AND NO TetR (Ultrasound) can the polymerase pass and produce Salmochelin and its receptor. Basal expression (leakiness) in healthy tissues is eliminated, protecting the patient’s iron homeostasis. The bacteria only release siderophores inside the tumor and when the physician decides.
2.3.- Biocontainment System (Biosafety Kill-Switch)
Implementation of a cascade activation mechanism. This design minimizes metabolic stress by not producing the drug sensor while the bacteria are colonizing the tumor.
- Oxygen Sensor (Normoxia): P_cyo promoter → araC gene. In the tumor (Hypoxia), the P_cyo promoter is turned off, so the AraC protein is not produced. In healthy tissue or the bloodstream (Normoxia), the P_cyo promoter is activated and synthesizes the AraC protein, which acts as the “key” for the drug.
- Drug Sensor: P_bad promoter → ccdB gene (Toxin). The P_bad promoter has an AND activation logic: it is only activated if the AraC protein is present (only in normoxia) AND Arabinose (the drug) is administered.
- Result: As long as the bacteria remain in the hypoxic TME, they are immune to the drug, as they lack the AraC protein. If the bacteria escape to oxygenated tissues or if the tumor is significantly reduced, the presence of oxygen allows the synthesis of AraC, making the bacteria sensitive to the drug for their total elimination.
2.4.- Note
Lipocalin-2 (Lcn2) can deplete intracellular iron in macrophages, generating an inflammatory response and promoting an attack on bacteria (Manfred Nairz et al., 2016); however, in cancer, Tumor-Associated Macrophages (TAMs) have an altered transcriptional program, where they are instead immunosuppressed. When the tumor size is significantly reduced, it is likely that macrophages will reprogram toward an M1 phenotype, assisting in the elimination of the bacteria alongside the kill-switch.
3. Governance and Policy Goals
To ensure that this bacteriotherapy contributes to an ethical and safe future, I have defined the following goals:
- Goal 1: Ensure Safety & Eficcacy
- Sub-goal 1a: Implement multi-layered biocontainment (NAND gate + Kill-switch) to prevent systemic iron depletion in the host.
- Sub-goal 1b: Prevent bacterial environmental persistence through strictly controlled clinical waste protocols (analogous to radioguided surgery) to ensure no bacterial escape into public sewage.
- Goal 2: Promote Constructive Applications & Equity
- Sub-goal 2a: Develop the platform using probiotic E. coli Nissle 1917 to keep production costs low and accessible for developing regions.
4. Governance Actions Matrix
To manage the specific risks of genetic drift and metabolic stress associated with the AraC/CcdB cascade design (Kill-Switch only), I propose the following interconnected actions:
| Aspect | Action 1: Technical (Biocontainment) | Action 2: Academic (Transparency) | Action 3: Regulatory (Standards) |
|---|---|---|---|
| Purpose | Implementation of a genetic circuit that prevents activation of the kill-switch when the synthetic bacteria is administered and at the same time minimizes metabolic stress in a hypoxic environment (TEM). | Establishment of a shared database focusing on the leakiness and metabolic burden of hypoxia-responsive sensors like P_cyo. | Development of standardized certification to ensure clinical reliability. |
| Design | Implementation of a cascade activation mechanism, where P_cyo promoter acts as a gatekeeper for AraC synthesis. P_bad then requires both AraC AND Arabinose to express the CcdB toxin. | Peer-reviewed publication of the “Stress-Safety Curve” of the AraC/CcdB cascade to define at what point mutation frequency increases. | Technical standards (e.g., ISO/TC 276) that define the Mean Time To Failure (MTTF) of the cascade before plasmid loss. |
| Assumptions | Assumes the AraC and ccdB genes remains functional. Risk of plasmid loss is acknowledged. Assumes the P_cyo promoter remains tightly repressed in hypoxia to prevent metabolic burden. | Assumes that labs will transparently share data when the P_cyo sensor leaks and kills the bacteria prematurely. | Assumes regulatory bodies (like ISP/FDA) have the expertise to audit compliance with established rules. |
| Risks | Genetic Drift: Loss-of-function mutations in araC or ccdB it generates bacteria that are immune to the drug. The overexpression of araC due to promoter mutations it generates Arabinose-sensitive bacteria even within the tumor. | Dual-Use: Detailed performance maps of the P_cyo sensor could be exploited to design oxygen-evading pathogens. | Innovation Lag: Excessive bureaucracy in certifying may delay new targeted gene therapies. |
Note on Scope: While my policy goals include the NAND gate for spatial control, the following Governance Matrix focuses specifically on the Kill-switch (AraC/CcdB cascade). I have prioritized this component because it represents the highest risk for environmental escape and is the “weakest link” in terms of biocontainment due to potential genetic drift.
5. Scoring and Prioritization
I have scored my proposed governance actions against my specific Policy Goals (1=Best, 3=Worst):
| Policy Goal | Action 1: Technical (Cascade Stability) | Action 2: Academic (Transparency) | Action 3: Regulatory (Waste & Standards) |
|---|---|---|---|
| Goal 1: Safety (Non-malfeasance) | 2 | 2 | 2 |
| Goal 2: Equity (Low-cost Access) | 2 | 1 | 3 |
| Feasibility (Implementation) | 1 | 2 | 2 |
Technical Note: In this assessment, Action 1 is scored based on the current plasmid-based design. However, to minimize the probability of genetic drift, I propose that the final implementation should transition to genomic integration of the AraC/CcdB cascade. This would ensure that the safety circuits are permanently embedded in the bacterial DNA, significantly reducing the risk of mutants compared to episomal (plasmid) expression.
6. Final Recommendation and Prioritization
Based on the scoring, I prioritize a combination of Action 1 (Technical) and Action 3 (Regulatory).
Priority and Audience: My recommendation is directed to the authorities responsible for verifying the safety and efficacy of new gene therapies. The technical design alone is insufficient without a clear regulatory framework.
Trade-offs: I have chosen to prioritize these over Action 2 (Academic Transparency) to mitigate the Dual-Use risk. While sharing detailed performance data of the P_cyo and P_bad sensors would promote global equity, the risk of this information being exploited to design pathogens that evade oxygen-based immune barriers is a trade-off I consider necessary for public safety.
Assumptions and Uncertainties: One assumption and uncertainty regarding what is being proposed is that the safety and efficacy criteria defined by the respective authorities could have undetected safety biases due to a lack of evidence.
7. Ethical Reflection
The most significant ethical concern that arose for me is the Dual-Use Dilemma in the context of biocontainment. I realized that the very mechanisms I am designing to ensure a therapy is safe (like high-precision oxygen sensors) are the same tools that could be used to engineer biological threats that are harder to detect or neutralize.
8. References
- Saha P., et al. (2019). “Enterobactin, an iron chelating bacterial siderophore, arrests cancer cell proliferation” Biochemical Pharmacology.
- Raffatellu M., et al. (2010). “Lipocalin-2 resistance of Salmonella enterica serotype Typhimurium confers an advantage during life in the inflamed intestine” Cell Host & Microbe.
- Pita-Grisanti V., et al. (2022). “Understanding the Potential and Risk of Bacterial Siderophores in Cancer” Frontiers in Oncology.
- Nairz M., et al. (2015). “Lipocalin-2 ensures host defense against Salmonella Typhimurium by controlling macrophage iron homeostasis and immune response” Journal of Immunology.
9. AI Prompts
In compliance with HTGAA 2026 guidelines, I certify that this homework was developed with the assistance of Gemini (Google AI).
- Image Generation: “Used Nano Banana to generate image “Abstract_Draw.png” from the detailed description of my project.”
- Assessment: “Create a table in markdown format that allows me to compare the design of my logic circuit with a standard design.”
- Troubleshooting: “Technical troubleshooting for personal profile configuration in the repository and helps to transfer the project in markdown format”