Week 10 HW: Advanced Imaging & Measurement Technology
Final Project
For my final project I would like to measure a few things:
- whether the biosensor can detect carbon monoxide (CO) in a reliable and quantifiable way. The main readout will be the signal generated after CO binding, such as fluorescence, or a visible color change, depending on the final design. By exposing the system to known CO concentrations, I can build a calibration curve and evaluate sensitivity, detection range, and response time.
- also if the protein sensor itself is being produced correctly and remains functional. For this, I would use SDS-PAGE to confirm protein size and purity, and UV-visible spectroscopy to verify heme incorporation and observe spectral changes upon CO binding. To test specificity, I would compare the response to CO with other gases or environmental factors such as CO2 and humidity.
Waters Part I — Molecular Weight
Question 1
Using the online calculator, the calculated molecular weight (unmodified) is 28006.60


Question 2
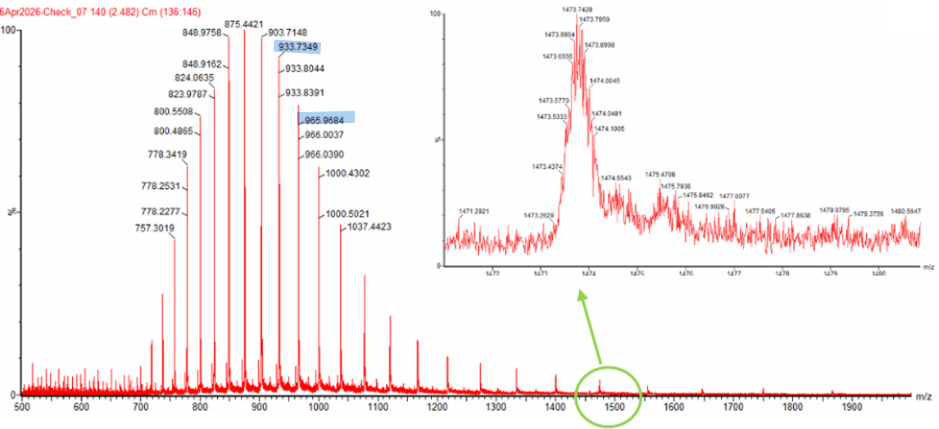
The two adjacent peaks I selected from the LC-MS spectrum at m/z 933.7349 and 965.9684.
 To determine $z$, we use
To determine $z$, we use
[ z = \frac{m/z_{n+1}}{(m/z_n - m/z_{n+1})} ]
Using the two adjacent peaks at $m/z = 933.7349$ and $m/z = 965.9684$, we assign $m/z_{n+1} = 933.7349$ and $m/z_n = 965.9684$, so
[ z = \frac{933.7349}{965.9684 - 933.7349} = \frac{933.7349}{32.2335} \approx 28.97 \approx 29 ]
Thus, the charge state of the peak at $m/z = 965.9684$ is $29+$, and the adjacent peak at $m/z = 933.7349$ corresponds to $30+$.
To determine the MW of the protein we can use the relationship between $\frac{m}{z_n}$, $MW$, and $z$:
[ \frac{m}{z} = \frac{MW + z}{z} ]
we rearrange to solve for $MW$:
[ MW = z\left(\frac{m}{z}\right) - z ]
Using $z = 29$ and the peak at $m/z = 965.9684$:
[ MW = 29(965.9684) - 29 ]
[ MW = 27984.08\ \text{Da} ]
Therefore, the molecular weight of the protein is 27984 Da
We can calculate the accuracy of the measurement using both this number 27984 Da, the predicted MW of 28006.60 Da and the formula: $$ \text{Accuracy} = \frac{|MW_{\text{experiment}} - MW_{\text{theory}}|}{MW_{\text{theory}}} $$ $$ \text{Accuracy} = \frac{|27984 - 28006.6|}{28006.6} = 8 \times 10^{-4}$$
Question 3
Yes, I can.
Waters Part I — Molecular Weight
There are
- Lysine (K): 20 residues
- Arginine (R): 6 residues
- Total K + R: 26.
MVSKGEELFTG VVPILVELDG DVNGHKFSVS GEGEGDATYG KLTLKFICTT GKLPVPWPTL VTTLTYGVQC FSRYPDHMKQ HDFFKSAMPE GYVQERTIFF KDDGNYKTRA EVKFEGDTLV NRIELKGIDF KEDGNILGHK LEYNYNSHNV YIMADKQKNG IKVNFKIRHN IEDGSVQLAD HYQQNTPIGD GPVLLPDNHY LSTQSALSKD PNEKRDHMVL LEFVTAAGIT LGMDELYKLE HHHHHH