Week 1 HW: Principles and Practices


Engineered bacterial cellulose to make self-pigmenting, living textiles.

Walker, K. T., Li, I. S., Keane, J., Goosens, V. J., Song, W., Lee, K.-Y., & Ellis, T. (2025). Self-pigmenting textiles grown from cellulose-producing bacteria with engineered tyrosinase expression. Nature Biotechnology, 43(3), 345–354. https://doi.org/10.1038/s41587-024-02194-3. Image from online article: https://www.nature.com/articles/s41587-024-02194-3
Describe a biological engineering application or tool you want to develop and why.
I am inspired by the bio-engineered, self- pigmenting Bacterial Cellulous developed by the Future Materials Group (FMG) at Imperial College London in collaboration with Modern Synthesis.
Their research responds to the environmental impact of textile production, largely through uncontrolled microplastic pollution from synthetic fabrics, water pollution from toxic, chemical dyes and its heavy contribution to greenhouse gas emissions. It is responsible for 10% of global carbon emissions,20% of global waste water, 35% of marine microplastic pollution and is expected to account for 25% of the global carbon budget by 2050. The question that interests me here is rather than manufacturing synthetic materials and colours, can we engineer materials to grow with the aesthetic properties we want?
Self-pigmenting bacterial cellulose offers a bio-degradable, non-toxic and grown alternative to synthetic fabrics and chemical dyeing. It builds upon the development of microbial leathers and dyes in the alternative textile field, pioneered by the likes of Suzanne Lee [Biocoutre](https://www.launch.org/innovators/suzanne-lee/, Laura Luchtman and Ilfa Siebenhaar Living Colour by adopting a synthetic biology approach. The Future Materials Group genetically engineering the bacterium Komagataeibacter rhaeticus to grow bacterial cellulose that is self pigmenting.
In carbon-rich growth media, Komagataeibacter metabolize sugars and converts them into cellulose chains, which are secreted outside the cell and self assemble into a fiber network called a pellicle. This pellicle is pure bacterial cellulose and forms the base material for the BC textile.
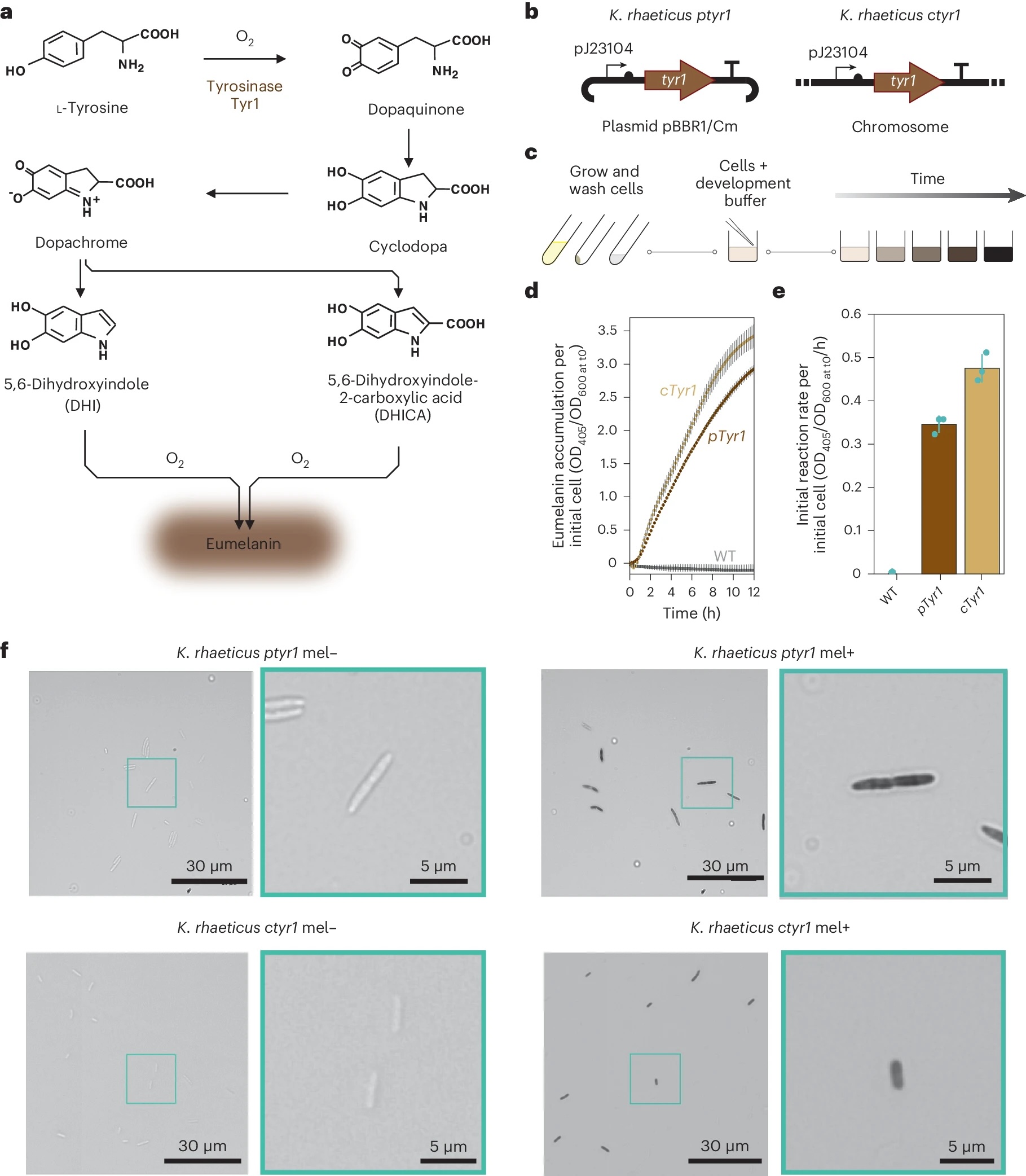
Walker, K. T., Li, I. S., Keane, J., Goosens, V. J., Song, W., Lee, K.-Y., & Ellis, T. (2025). Self-pigmenting textiles grown from cellulose-producing bacteria with engineered tyrosinase expression. Nature Biotechnology, 43(3), 345–354. Image: Eumelanin production from K. rhaeticus tyrosinase expression. From: Self-pigmenting textiles grown from cellulose-producing bacteria with engineered tyrosinase expression. Available at: https://www.nature.com/articles/s41587-024-02194-3/figures/1
Describe one or more governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future.
1. Ensuring biosafety and laboratory containment:
The World Health Organization defines “Biosafety” in its Laboratory Biosafety Manual as “containment principles, technologies and practices that are implemented to prevent unintentional exposure to biological agents or their inadvertent release (World Health Organization, 2004).”
Genetically engineering Komagataeibacter rhaeticus to produce self-pigmenting bacterial cellulose introduces modified microbes into research and production environments. While K. rhaeticus is non-pathogenic and requires strict growth conditions to survive, the insertion of the tyrosinase gene (Tyr1) creates new biological capabilities which carry some risks of producing unintended, harmful effects to the health of humans and the environment if released.
It is particularly important to protect laboratory personell, scientists, designers, consumers and neighbouring communities from exposure to toxic, infectious or dangerous pathogens and to prevent their accidental release outside the laboratory with strict containment protocol.
2. Preventing harm to the environment:
Self-pigmenting BC textiles offer an environmentally friendly alternative to synthetic fabrics and chemical dyes, but the long-term ecological impact of introducing genetically engineered bacteria must be considered. It is important to ensure that the development, production and onward life of the Bacterial Cellulose Textile does not negatively affect the environment.
Genetic modifications and decisions made for scalability should not compromise biodegradability. Production systems should avoid petroleum-based plastics or toxic media. End-of-life disposal, industrial cultivation and potential escape into natural ecosystems should be monitored to prevent unintended ecological consequences.
3. Ensuring equity:
Access to self-pigmenting BC textiles should extend beyond large corporations and well-funded labs to designers, artisans, and educational institutions.
Development of this technology should ensure low-cost, modular production systems to enable the equitable participation of non-traditional or underprivileged communities in sustainable material creation.
Training in biosafety and handling of engineered microbes should accompany access, ensuring that communities can safely experiment with living textiles while benefiting from environmentally responsible materials.
4. Transparency and education:
Clear communication of the science, benefits and limitations of self-pigmenting BC is critical for stakeholder trust, informed consent and wider uptake of engineered living materials.
Designers, consumers, and regulators should understand how BC reduces reliance on petroleum based textiles and avoids toxic dyes. Transparency should include how the bacteria are engineered, the pigment synthesis process, and any potential environmental or health considerations.
Engaging the public through workshops, open-access resources and participatory design initiatives encourages adoption while supporting ethical decisions about integrating engineered living textiles into daily life.
5. Respect for life:
Self Pigmenting Bacterial cellulose is a living material produced by bacteria and ethical considerations should extend to the organisms themselves.
Scaling self-pigmenting BC production for textiles must balance functionality with respect for life, acknowledging the bacteria as active contributors to the material.
This perspective highlights a distinction between large-scale industrial production and smaller, artisanal or community-based approaches, emphasising that sustainability and innovation should be pursued without disregarding the living organisms at the core of bio-fabrication.
Describe at least three different potential governance “actions”, consider: Purpose, Design, Assumptions, Risks of Failure & Success.
I have outlined 3 categories of actors:
Researchers:
Those involved in the research and development of Self pigmenting Bacterial cellulose e.g Scientists, Universities, Research Centers, Community Bio-labs.
Producers:
Those involved in turning SPBC into actual products and for use in design and manufacturing e.g. Designers, artists, fashion designers, companies, bio-material start ups.
Regulators:
Those involved in control of safely deploying this technology on a local, national and international level e.g Local Authority Environmental Health Department, Waste Management Boards, Research Ethics Committees, Department of Environment, Food and Rural Affairs, The Health and Safety Executive.
1. Ensuring biosafety and laboratory containment
ACTION: Implement strict biosafety protocols and containment standards for research and production facilities handling engineered K. rhaeticus.
PURPOSE: Prevent accidental exposure or environmental release of genetically modified bacteria.
DESIGN:
Researchers should ensure that genetically engineered K. rhaeticus strains are safe and non-pathogenic.
Regulators should require Biosafety Level 1+ facilities, PPE, and biosafety training for all SPBC work.
Researchers should test bio-containment strategies such as kill-switches linked to the Tyr1 pigment pathway.
Producers should gain approval from institutional or local biosafety boards before scaling production.
Regulators should enforce sterilisation of spent cultures prior to disposal and include periodic risk and life-cycle assessments in SPBC workflows.
ASSUMPTION: I assume that risk to biosafety from K. rhaeticus is low and that it cannot survive in open environments. I assume that containment technologies and protocols are sufficient to prevent accidental release. I assume that all researchers and designers would adhere to training and protocol.
RISK OF FAILURE: Failure risks accidental release beyond the laboratory that could lead to unintended effect on the environment or humans.
RISK OF SUCCESS: Safe research and production is wide spread, increasing demand for DIY genetic engineering and biohacking to make SPBC, which may require more wide spread biosafety training and stricter protocol.
2. Preventing harm to the environment
ACTION: Require lifecycle assessment and contained environmental monitoring for large-scale production of self pigmenting bacterial cellulose.
PURPOSE: Ensure that self-pigmenting BC textiles remain a sustainable alternative to synthetical coloured fabrics and monitor its life cycle to ensure that use and disposal of genetic engineering materials has no unintended impact on the environment.
DESIGN:
Researchers should test large-scale cultivation and end-of-life treatment to prevent ecological pollution.
Producers should be required to submit life-cycle assessments covering carbon footprint, biodegradability, toxicity, and disposal.
Regulators should enforce compliance with local environmental and waste-management regulations for genetically engineered materials.
ASSUMPTION: I assume that engineered self-pigmented bacterial cellulose behaves consistently at different scales and with different production methods, when individual producers may need more tailored environmental restrictions. I assume that all environmental monitoring can accurately capture all possible environmental impacts long term. I assume that with scalability and commercialisation, all producers will maintain sustainability as a priority in producing SPBC.
RISK OF FAILURE: Genetic engineering and scalability effects the biodegradability of the material. Unforeseen environmental impact is created by the use and disposal of SPBC at scale.
RISK OF SUCCESS: Wide spread commercial uptake of SPBC creates challenges for scaled manufacturing and efficient cellulose yield, increasing environmental impact.
3. Ensuring equity, transparency and education
ACTION: Develop technology should also fund low-cost and accessible production available to smaller bio-production spaces. Researchers and producers would hold engagement and education initatives so that use of the sustainable material is understood and available to a wide range of stakeholders.
PURPOSE: Allow equitable access to bio-engineered, living material and ensure transparency and consent from the public.
DESIGN:
- Researchers should provide biosafety training and open educational resources for community labs and schools.
- Researchers should establish governed strain-sharing programs for qualified smaller labs.
- Researchers should partner with artists, designers and under-resourced communities to explore local applications.
- Producers should be required to provide clear labelling and marketing explaining pigment production, genetic engineering and environmental benefits.
ASSUMPTION: I assume there would be engagement from non-traditional users. I assume there would be resources and initatives in place to support community engagement and educational initatives. I assume that some smaller labs would be able to maintain proper biosafety practices to accomodate the use of genetically engineered bacterial cellulose.
RISK OF FAILURE: Risk the misuse of genetically engineered material. Risk that underfunding may limit accessibility to use of the material and its development remains expensive and highly centralised. Miscommunication could cause fear or misinformation and create an aversion to the uptake of the material.
RISK OF SUCCESS: Success expands access to sustainable textiles and gentic engineering but poses complexities with a higher demand for DIY biohacking and public access to genetic engineering.
4. Respect for life
ACTION: Establish and enforce ethical guidelines for scaling SPBC production.
PURPOSE: To recognise bacteria as living contributors and discourage unnecessary cultivation, waste or harmful treatment.
DESIGN:
- Regulators should create industry standards for responsible growth volumes (avoiding over-production of Komagataeibacter rhaeticus).
- Open forum for discussion between researchers and producers to establish conduct for most humane termination and sterilisation of cultures after use.
ASSUMPTION: I assume stakeholders value ethical treatment of bacteria. I assume that ethics will be prioritised over industrial production. I assume ethical guidelines influence industrial practices.
RISK OF FAILURE: Industrial production may prioritise efficiency over respect for living organisms.
RISK OF SUCCESS: The inherent conflict of the use of other living beings still exists.
Score (from 1-3 with, 1 as the best) each of your governance actions against your rubric of policy goals.
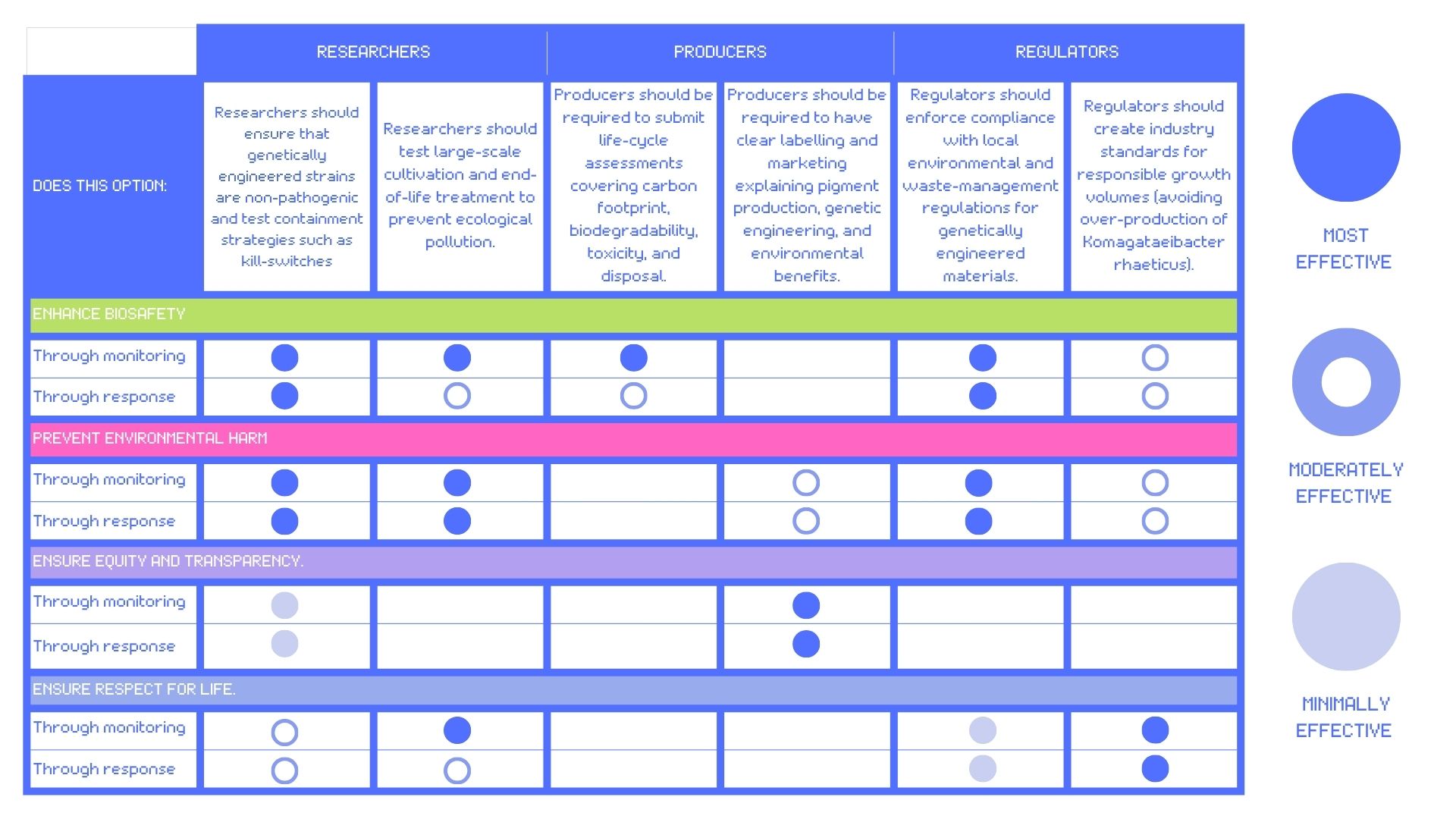

Rubric by Isobel Jo Leonard
Describe which governance option or combination of options, you would prioritise, and why.
Priorities
From the analysis in my table, the governance goals I would prioritise for the ethical development of self-pigmenting bacterial cellulose would be bio-security, prevnting environmental harm and transparency. This would require a hybrid strategy that triangulates researcher responsibility, producer accountability and high regulatory standards on a local, national and international level.
First, I prioritise actions that embed biosafety at the research stage. Researchers engineering Komagataeibacter rhaeticus should be responsible for ensuring non-pathogenic behaviour, testing containment strategies such as Tyr1-linked kill-switches, evaluating large scale cultivation strategies and end-of-life sterilisation. Designing safety into self-pigmenting bacterial cellulose production early is the most effective way of reducing harm when the materials enter production or circulation.
Second, environmental regulation becomes critical as SPBC scales beyond the lab. Producer-level actions such as life-cycle assessment, biodegradability standards and regulated waste management ensure that genetic modification and increased scale and access do not undermine the sustainability goals of replacing petroleum-based textiles, nor does SPBC’s use or disposal disrupting natural ecosystems.
Finally, these technical controls must be supported by transparency and public engagement. Clear labelling and education initiatives surrounding how genetic engineering operate in SPBC allow designers, consumers, and regulators to make informed decisions. Transparency builds trust and supports adoption, ensuring that living, engineered biomaterials can be widely adopted safely.
Trade offs
The rubric highlights a clear trade-off between biosecurity and equity. The stricter regulatory measures for researchers and producers increase the costs and barriers to working with these materials. While it is important that DIY bio-fabrication spaces and small-scale industries maintain access, this access cannot compromise biosafety regulations or the requirement for ethical research training.
Another inherent trade-off involves respect for life. Efforts to expand the development, accessibility and scalability of this technology raise ethical questions regarding the extractavist use of more than human life in the service of human ends.
Assumptions and uncertainties
I think an assumption underpinning my analysis is that producers and regulators will prioritise sustainability over the efficiency and profit of scaling this highly functional material. Although the fundamental process of producing self-pigmenting bacterial cellulose is sustainable, I am uncertain whether its development for applications in industries such as fashion and design, would create pressures for increasing yield, durability and aesthetic that compromise its sustainability. For this reason, my proposed actions may have over stated the influence and enthusiasm of regulatory bodies to prevent harm.
References
Ellis, T. (n.d.) Engineering bacteria to grow a leather that dyes itself black. Available at: https://www.tomellislab.com/post/engineering-bacteria-to-grow-a-leather-that-dyes-itself-black
Florea, M., Hagemann, H., Santosa, G., Abbott, J., Micklem, C.N., Spencer-Milnes, X., de Arroyo Garcia, L., Paschou, D., Lazenbatt, C., Kong, D., Chughtai, H., Jensen, K., Freemont, P.S., Kitney, R., Reeve, B. and Ellis, T. (2016) ‘Engineering control of bacterial cellulose production using a genetic toolkit and a new cellulose-producing strain’, _Proceedings of the National Academy of Sciences of the United States of America. https://doi.org/10.1073/pnas.1522985113
Goosens, V.J., Coussement, P., De Paepe, B., De Maeseneire, S., De Mey, M. and Soetaert, W. (2021) ‘Komagataeibacter tool kit (KTK): a modular cloning system for multigene constructs and programmed protein secretion from cellulose producing bacteria’, ACS Synthetic Biology. https://doi.org/10.1021/acssynbio.1c00365
Huang, Y., Zhu, C., Yang, J., Nie, Y., Chen, C. and Sun, D. (2014) ‘Recent advances in bacterial cellulose’, Cellulose. https://doi.org/10.1007/s10570-013-0088-z
Hunckler, M.D. and Levine, A.D. (2022) ‘Navigating ethical challenges in the development and translation of biomaterials research’, Frontiers in Bioengineering and Biotechnology. https://doi.org/10.3389/fbioe.2022.899428
Malcı, K., Li, I.S., Kisseroudis, N. and Ellis, T. (2024) ‘Modulating microbial materials: Engineering bacterial cellulose with synthetic biology’, ACS Synthetic Biology. https://doi.org/10.1021/acssynbio.4c00615
Next Nature Network (2019) Bio-textiles. Available at: https://nextnature.org/en/magazine/story/2019/bio-textiles
Ou, Y. and Guo, S. (2023) ‘Safety risks and ethical governance of biomedical applications of synthetic biology’, Frontiers in Bioengineering and Biotechnology. https://doi.org/10.3389/fbioe.2023.1292029
Quijano, L., Fischer, D., Ferrero-Regis, T. et al. (2025) ‘Exploring bacterial cellulose as an engineered living and programmable biomaterial across disciplines through qualitative thematic analysis’, Scientific Reports. https://doi.org/10.1038/s41598-025-01931-1
Schiros, T.N. et al. (2022) ‘Microbial nanocellulose biotextiles for a circular materials economy’, Environmental Science: Advances. https://doi.org/10.1039/D1VA00044E
Shuster, V. and Fishman, A. (2009) ‘Isolation, cloning and characterization of a tyrosinase with improved activity in organic solvents from Bacillus megaterium’, Journal of Molecular Microbiology and Biotechnology. https://doi.org/10.1159/000243640
Singhania, R.R., Patel, A.K., Tsai, M.L., Chen, C.W. and Dong, C.D. (2021) ‘Genetic modification for enhancing bacterial cellulose production and its applications’, Bioengineered. https://doi.org/10.1080/21655979.2021.1968989
Tkaczyk, A., Mitrowska, K. and Posyniak, A. (2020) ‘Synthetic organic dyes as contaminants of the aquatic environment and their implications for ecosystems’, Science of the Total Environment. https://doi.org/10.1016/j.scitotenv.2020.137222
Hub for Biotechnology in the built environment. Human–bacteria interfaces. Available at: http://bbe.ac.uk/human-bacteria-interfaces/
Week 2: DNA: Read, Write, Edit Homework Questions
Homework Questions from Professor Jacobson:
Nature’s machinery for copying DNA is called polymerase. What is the error rate of polymerase? How does this compare to the length of the human genome. How does biology deal with that discrepancy?
AI prompts: ChatGPT accessed 2026: “Can you explain Error Correcting Polymerase to me in the stages of extension, extension error and proofreading? “What would the error rate of polymerase be copying the human genome, error rate of polymerase is 1:10⁶”
The Error Rate of polymerase is 1:10⁶. Human genome length is ~3 × 10⁹ bp. Without proofreading, the error rate for copying the human genome would be large (approx. error rate is ~3,000 mistakes per replication.)
Biology deal with this discrepency with Error Correcting Polymerase which works in stages:
Extension: The polymerase adds nucleotides (nucleotide triphosphates) one by one to the growing DNA strand.
Extension error: Occasionally, the polymerase incorporates an incorrect, non-complementary nucleotide and stalls.
Proofreading: The polymerase detects and removes misincorporated nucleotides via 3′→5′ exonuclease activity, drastically reducing errors.
This all allows for accurate replication of the large human genome despite the high error rate of the polymerase.
How many different ways are there to code (DNA nucleotide code) for an average human protein? In practice what are some of the reasons that all of these different codes don’t work to code for the protein of interest?
Due to codon degeneracy, an average human protein (~345 amino acids) can be coded by an astronomically large number of different DNA sequences.
Even though they code for the same amino acids, many sequences are unusable due to:
- Codon bias: different organisms prefer different codons due to tRNA abundance. If an organism uses a rare codons it can slow or stall translation because trna is scarce causing the protien to misfold.
- mRNA stability and folding: some DNA nucleotide sequences produce mRNA that folds into secondary structures, which can prevent efficient translation even though the amino acid sequence is correct.
Homework Questions from Dr. LeProust:
What’s the most commonly used method for oligo synthesis currently?
Solid-phase phosphoramidite synthesis.
Why is it difficult to make oligos longer than 200nt via direct synthesis?
AI prompts: ChatGPT accessed 2026: Explain this section to me simply: “A key challenge in DNA synthesis is the generation of >300- nucleotide DNA, which is limited by the elongation cycle efficiency, that is, the efficiency with which each nucleotide is incorporated in the sequence. For example, with the elongation cycle efficiency of 99%, the theoretical yield for an oligonucleotide comprising 120 nucleotides is ~30%(0.99 120 × 100%). However,for a 200 bp polymer/ oligonucleotide,this is reducedtojust 13%. Attempts to over come this issue have focused on improving the accuracy and speed of DNA assembly processes.”
It is difficult to make oligonucleotides longer than 200 nt via direct chemical synthesis because of the elongation cycle efficiency (the efficiency with which each nucleotide is added during synthesis). Even if the per-step efficiency is high (e.g. 99%), the overall yield of full-length oligos drops exponentially with length. For example, a 120 nt oligo would have ~30% full-length product, while a 200-nt oligo drops to ~13%. This cumulative effect of imperfect nucleotide addition makes direct synthesis of long oligos impractical.
Why can’t you make a 2000bp gene via direct oligo synthesis?
AI prompts: “What would be the difference to synthesise a 2000bp gene via direct synthesis?”
Essentially for the same reason. Oligos are synthesised with solid phase phosphormidite one nucleotide at a time. A 2000bp gene is double stranded DNA meaning 1bp is 2 nucleotides, that is 4000 nt. The yield of full-length product declines rapidly as the length exceeds 150-200 nucleotides. 200 nt is already low (~13% full-length at 99% efficiency per step). For a 2000 bp, the theoretical yield of the correct molecule becomes negligibly small, often less than 1%.
Homework Question from George Church:
What are the 10 essential amino acids in all animals and how does this affect your view of the “Lysine Contingency”?
Histidine (His),Isoleucine (Ile), Leucine (Leu),Lysine (Lys), Methionine (Met), Phenylalanine (Phe), Threonine (Thr), Tryptophan (Trp), Valine (Val), Arginine (Arg).
By “Lysine Contingency” I assume this is refering to Jurassic Park, where scientists genetically engineer the Dinosaurs DNA so they cannot synthesise lysine. This creates a dependency on the lysine supplements provided by the park staff. Given that lysine is an essential amino acid for metabolising proteins and the dinasaurs would not be able to survive without it, the “Lysine Contingency” is effectively a kill switch, a clever containment strategy which prevents the survival of the dinosaurs if they escaped the island and ensuring they could not harm the outside world or global ecosystems.