Week 7 HW: Genetic Circuits Part II

Important
PART 1: Intracellular Artificial Neural Networks (IANNs)
What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions?
Non-linear computing:
Boolean genetic circuits can only compute linearly separable functions. As Halužan Vasle and Moškon state, a single-layer perceptron “can solely learn to classify linearly separable classes” meaning XOR and more complex classifications are unachievable without exponentially more logic gates which rapidly becoming unscalable (Britto Bisso et al. 2025). The multilayer architecture of IANNs instead allows hierarchical processing across layers, where the output of one layer becomes a weighted regulatory signal for the next. This gives IANNS the advantage of being able to encode sophisticated behaviours with far fewer biological parts.
Analogue computation better suited to biological signals:
Boolean circuits treat gene expression as on or off, forcing continuous biological signals into discrete, binary categories. IANNs can speak the same language as biologically regulated networks with intermediate levels, analogue values, multi-input systems and non-monotonic behaviours (Gago et al., 2010). E.g. Britto Bisso et al. identify four ubiquitous chemical reaction networks: molecular sequestration, catalytic degradation, competitive binding, and activation/deactivation cycles, that all produce continuous threshold-like input-output curves resembling ReLU or sigmoidal activation functions. Since real intracellular signals are rarely binary, IANNs are architecturally better suited to these naturally graded, multi-dimensional inputs whereas Boolean circuits must artificially threshold them, introducing error and information loss.
Tuneable weights:
In Boolean circuits the logic function is hardwired into their topology and changing behaviour requires redesigning the circuit entirely. In IANNs, weights are encoded in production rates e.g promoter strengths, ribosome binding site efficiencies or enzyme catalytic rates, meaning the same circuit architecture can be reprogrammed to implement different functions simply by tuning these parameters (Britto Bisso et al. 2025) which is a flexibility Boolean circuits fundamentally cannot achieve.
Limitations:
Current IANNs predominantly rely on in silico pre-training with weights hardwired biologically afterwards, online learning within a living cell remains unsolved (Halužan Vasle & Moškon, 2024). Furthermore, the number of perceptrons stably deliverable into a cell is limited by current DNA delivery methods which constrains the circuit complexity achieveable in practice (Britto Bisso et al. 2025).
Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
A useful future application for IAAN is as a intracellular diagnostic biosensor. These could continuously monitoring multiple molecular biomarkers to distinguish early-stage oncogenic signalling from normal cellular behaviour. Ultimately, the IAAN could control a targeted response e.g trigger apoptosis or a therpeutic pathway when a cancerous state is confirmed.
While this has not yet been experimentally realised, Britto Bisso et al. (2025) demonstrate in silico that biomolecular neural networks can classify healthy versus cancerous cells across 19 tissue types using non-linear decision boundaries impossible, Rizik et al. (2022) proved that genuine multilayer perceptron architectures can be physically implemented in living cells using the perceptgene framework and Senn et al. (2026) demonstrate that synthetic gene circuits sensing multiple RAS-driven oncogenic inputs can selectively kill cancer cells in vivo (using Boolean AND-gate logic). An future IANN biosensor would provide a multilayer, continuously weighted circuit capable of detecting and responding to cancerous cells.
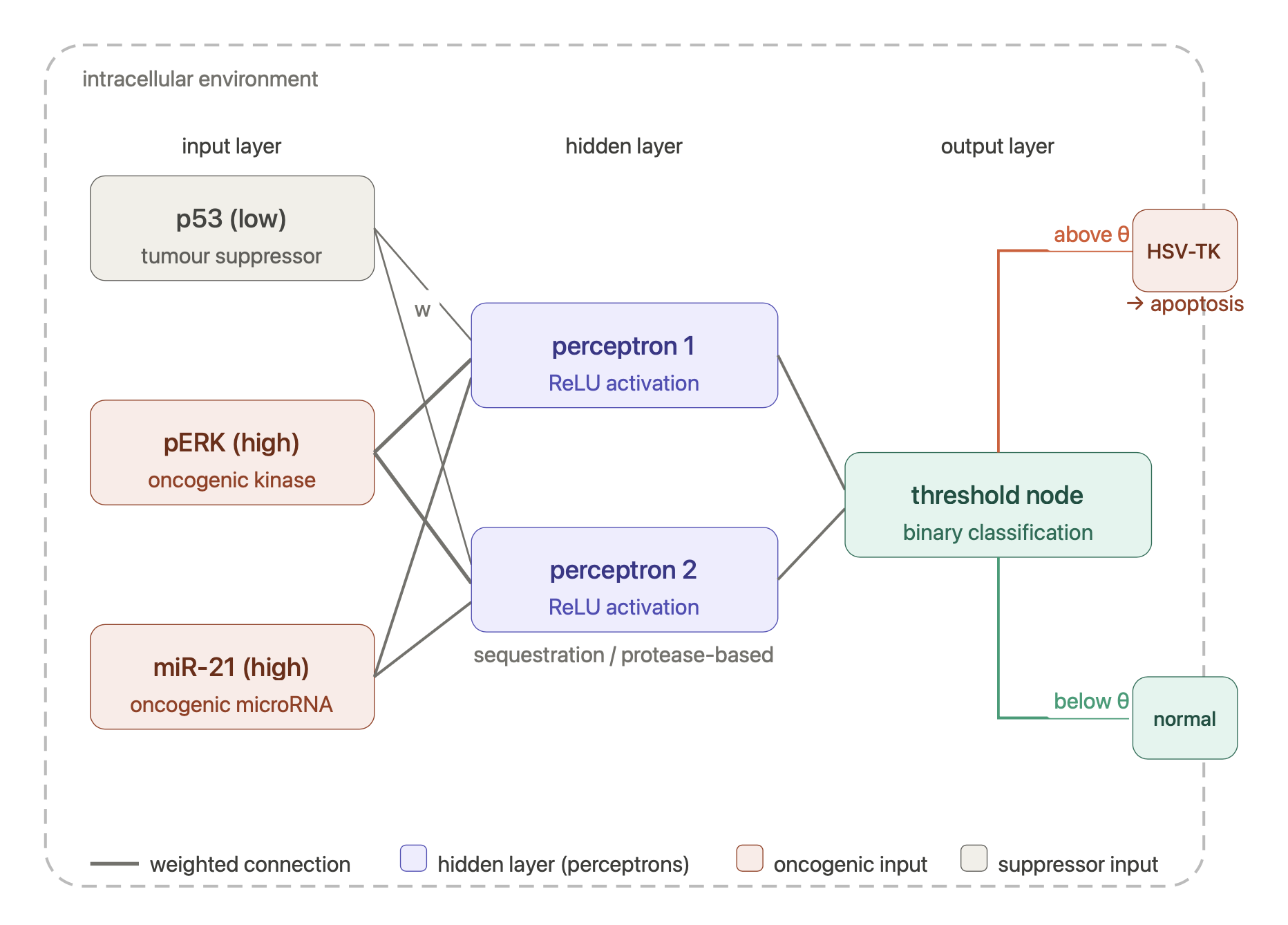

Schematic made with Claude
Input/Output Behaviour:
The IANN uses molecular concentrations as inputs, processes them through a bio-molecular circuit (protein-protein interactions, DNA strand displacement or RNA interference) and produces a molecular output.
Several biomarkers are simultaneously monitored and their combined weighted pattern identifies the cancerous state (encoding weights in binding affinities, Hill coefficients and promoter strengths) reflecting the reality that healthy and cancerous cells differ in degree of expression rather than binary presence or absence(Rizik et al. 2022).
Inputs could include biomarkers such as:
- Low concentration of a tumor suppressor protein (e.g p53)
- High concentration of an oncogenic signaling kinase (e.g phosphorylated ERK)
- High expression of a specific microRNA associated with cancer progression (e.g miR-21)
These inputs would be processed through a hidden layer of sequestration-based or protease-based perceptrons implementing ReLU-like activation functions.
The output layer then produces a binary classification e.g below threshold the cell is deemed healthy and no action is taken but above threshold a therapeutic effector such as the HSV-TK suicide gene demonstrated by Senn et al. (2026) is expressed and selectively trigger cancer cell death. Critically, because the decision boundary is non-linear and continuously tuneable, the IANN could discern overlapping expression profiles that would cause a Boolean circuit to misfire.
Limitations:
- Adaption:
Current IANNs rely on in silico pre-training with fixed weights , the network cannot adapt to tumour evolution or emerging resistance mutations once deployed (Halužan Vasle & Moškon, 2024). Even Senn et al. (2026), working with a simpler Boolean architecture, acknowledge that heterogeneous cancer cell lines required circuit adaptation for each target cell type, highlighting how demanding this problem is.
- Metabolic Burden:
Any synthetic gene circuit introduces exogenous components (DNA/RNA/proteins) that consume the host cell’s resources such as ATP, amino acids. This creates a metabolic burden that can weaken the cell, alter its normal function or lead to cell death before the IANN completes its task (Halužan Vasle & Moškon, 2024). This is an even greater challenge for multilayer IANNs, where each additional layer further adds to this burden.
- Scalability:
Britto Bisso et al. (2025) note there is an upper limit to the number of perceptrons stably deliverable into primary therapeutic cell lines, constraining classification accuracy. Senn et al. (2026) acknowledge, multi-component delivery into solid tumours remains a fundamental barrier and integrating the additional complexity of a multilayer IANN architecture onto a deliverable vector is even more challenging.
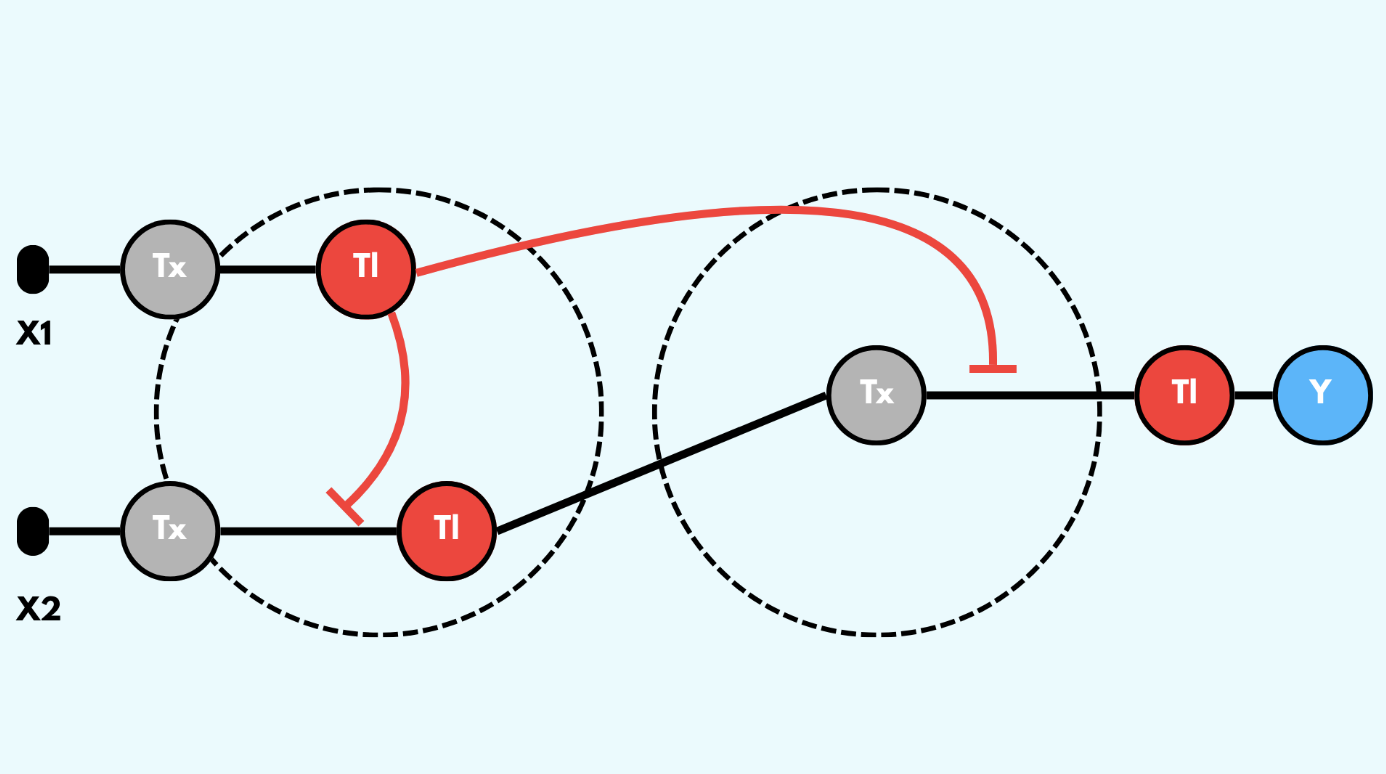
Draw a diagram for an intracellular multilayer perceptron where layer 1 outputs an endoribonuclease that regulates a fluorescent protein output in layer 2.

Part 2: Fungal Materials
What are some examples of existing fungal materials and what are they used for? What are their advantages and disadvantages over traditional counterparts?
Fungal Materials, Applications and Analysis by Isobel LeonardWhat might you want to genetically engineer fungi to do and why?


Maca Barrera 2024, Melanin-spiked bodysuits provide sun protection in Melwear concept :https://www.dezeen.com/2024/06/10/melanin-melwear-maca-barrera-design-technology/
I am interested in the genetic engineering of the pigmentation pathways of fungi to create an engineered living material for textiles and design. Fungi naturally produce a diverse palette of pigments including carotenoids, flavins, melanins, quinones and violacein. Genetically engineering the over-expression or selective activation of these pathways could eliminate the need for post-processing dyeing entirely in the textile industry which accounts for approximately 20% of global industrial water pollution. This concept has already been demonstrated in bacterial cellulose, where recombinant tyrosinase expression achieved dark black melanin coloration robust to material use, with the potential for optogenetic patterning of gene expression to create spatially controlled pigment patterns (Walker et al .2025).
Beyond static colour, engineering responsive pigmentation in fungal materials could have interesting applications in bio-sensing or environmentally responsive wearables that could function as a reactive fungal second skin. For example, by placing melanin-producing enzymes under the control of environmentally sensitive promoters, a fungal materials could be designed to change colour in response to mechanical stress, humidity, UV exposure or temperature and provide real-time feedback on the body or environmental conditions of the wearer without the addition of electronics.
What are the advantages of doing synthetic biology in fungi as opposed to bacteria?
Eukaryotic:
Fungi are eukaryotes, meaning that unlike bacteria they possess specialised organelles like the Endoplasmic Reticulum (ER) and Golgi apparatus which enable post-translational modifications e.g. glycosylation, chaperone assisted folding to help complex proteins into the correct 3D strcuture and disufide bond formation, necessary to stabilise the structure of secreted proteins, such as antibodies. These are fundamentally eukaryotic properties and cannot be replicated in bacterial systems. They is essential for engineering structurally complex proteins such as hydrophobins or silk-like fibres require proper folding to function mechanically.
Substrate extension:
Fungi extend their mycelium to reach organic food sources and incorporate these particles into their hyphal network, with glucan and chitin rich hyphae binding themselves to low-cost and renewable substrates that are rich in cellulose and lignin together. Bacteria generally cannot efficiently degrade lignocellulosic agricultural waste. This means fungal synthetic biology can be deployed directly on cheap, abundant waste substrates without expensive pre-treatment, making it more economical at scale.
Metabolic and genetic diversity:
Fungi have extensive secondary metabolic pathways encoded by Biosynthetic Gene Clusters, this means they produce diverse compounds such as terpenoids and polyketides e.g.statins, antibiotics. Fungi can generate four times as many secondary metabolites as bacteria.
Secretory Pathways:
Fungi are high-volume producers of extracellular enzymes and are capable of secreting large amounts of proteins directly into the culture medium. This makes downstream purification much easier when compared to bacteria.
References
Britto Bisso, F., Shree, D., Zhu, Y., & Cuba Samaniego, C. (2025). Design principles of neuromorphic computing using genetic circuits. bioRxiv. https://doi.org/10.64898/2025.12.01.691482
Gago, J., Landín, M., & Gallego, P. P. (2010). Strengths of artificial neural networks in modelling complex plant processes. Plant Signaling & Behavior, 5(6), 743–745. https://doi.org/10.4161/psb.5.6.11711
Halužan Vasle, A., & Moškon, M. (2024). Synthetic biological neural networks: From current implementations to future perspectives. BioSystems, 238, 105164. https://doi.org/10.1016/j.biosystems.2024.105164
Rizik, L., Danial, L., Habib, M., Weiss, R., & Daniel, R. (2022). Synthetic neuromorphic computing in living cells. Nature Communications, 13, 5602. https://doi.org/10.1038/s41467-022-33288-8 Senn, G. V., Nissen, L., & Benenson, Y. (2026). Synthetic gene circuits that selectively target RAS-driven cancers. eLife. https://doi.org/10.7554/eLife.104320.3
Smole, A., Lainšček, D., Bezeljak, U., Horvat, S., & Jerala, R. (2017). A synthetic mammalian therapeutic gene circuit for sensing and suppressing inflammation. Molecular Therapy, 25(1), 102–119. https://doi.org/10.1016/j.ymthe.2016.10.005
Gago, J., Landín, M., & Gallego, P. P. (2010). Strengths of artificial neural networks in modelling complex plant processes. Plant Signaling & Behavior, 5(6), 743–745. https://doi.org/10.4161/psb.5.6.11711
Alaneme, K. K., Anaele, J. U., Oke, T. M., Kareem, S. A., Adediran, M., Ajibuwa, O. A., & Anabaranze, Y. O. (2023). Mycelium-based composites: A review of their processing, properties and applications. Alexandria Engineering Journal, 83, 234–250.
Appels, F. V. W., Camere, S., Montalti, M., Karana, E., Jansen, K. M. B., Dijksterhuis, J., Krijgsheld, P., & Wösten, H. A. B. (2019). Fabrication factors influencing mechanical, moisture- and water-related properties of mycelium-based composites. Materials & Design, 161, 64–71. https://doi.org/10.1016/j.matdes.2018.11.027
Haneef, M., Ceseracciu, L., Canale, C., Bayer, I. S., Heredia-Guerrero, J. A., & Athanassiou, A. (2017). Advanced materials from fungal mycelium: Fabrication and tuning of physical properties. Scientific Reports, 7, 41292. https://doi.org/10.1038/srep41292
IDTechEx. (2024). Emerging leather alternatives — mushrooms, microbial and lab grown. https://www.idtechex.com/en/research-article/emerging-leather-alternatives-mushrooms-microbial-and-lab-grown/31706
Jones, M., Bhat, T., Kandare, E., Thomas, A., Joseph, P., Dekiwadia, C., Yuen, R., John, S., Ma, J., & Wang, C. H. (2018). Thermal degradation and fire properties of fungal mycelium and mycelium-biomass composite materials. Scientific Reports, 8, 17583. https://doi.org/10.1038/s41598-018-36032-9
Pelletier, M. G., Holt, G. A., Wanjura, J. D., Bayer, E., & McIntyre, G. (2013). An agroecological approach to the creation of a new building material. Industrial Crops and Products, 43, 612–616. https://doi.org/10.1016/j.indcrop.2012.07.047
Vandelook, S., Elsacker, E., Van Wylick, A., De Laet, L., & Peeters, E. (2021). Current state and future prospects of pure mycelium materials. Fungal Biology and Biotechnology, 8, 20. https://doi.org/10.1186/s40694-021-00128-7
Elsacker, E., Martin, J. S., Sangosanya, A., Verstuyft, A., Van Wylick, A., & Peeters, E. (2025). Gradients of aliveness and engineering: A taxonomy of fungal engineered living materials. Advanced Materials. https://doi.org/10.1002/adma.202502728
Walker, K. T., Li, I. S., Keane, J., Goosens, V. J., Song, W., Lee, K.-Y., & Ellis, T. (2025). Self-pigmenting textiles grown from cellulose-producing bacteria with engineered tyrosinase expression. Nature Biotechnology, 43(3), 345–354. Available at: https://www.nature.com/articles/s41587-024-02194-3/figures/1_