Week 1 Lab: Pipetting

Contents
Practice
Dilution practice 1
The stock concentration of a mystery substance (MS) is 5 M. Calculate how to dilute to 100 µM (0.1 mM).
$$ C_1 V_1 = C_2 V_2 $$
$$ (5,000,000 \mu M)* V_1 = (10,000 \mu M)* (500 \mu L) $$
$$ (5,000,000 \mu M)* V_1 = 5,000,000 \mu M * \mu L $$
$$ V_1 = 1 \mu L $$
$$ V_{diluant} = V_2 - V_1 $$
$$ V_{water} = 500 \mu L - 1 \mu L $$
$$ V_{water} = 499 \mu L $$
$$ C_2 V_{2B} = C_3 V_3 $$
$$ (10,000 \mu M)* V_{2B} = (100 \mu M) * (100 \mu L) $$
$$ (10,000 \mu M)* V_{2B} = 10,000 \mu M * \mu L $$
$$ V_{2B} = 1 \mu L $$
$$ V_{diluant2} = V_3 - V_{2B} $$ $$ V_{water2} = 100 - 1 \mu L $$ $$ V_{water2} = 99 \mu L $$
For the first step, I would use 1 µL of the stock solution diluted into 499 µL of water to make 500 µL of a 10,000 µM solution. Then for the next step, I would use 1 µL of the 10,000 µM dilution, diluted into 99 µL of water to make 100 µL of a 100 µM solution.
Dilution practice 2
The stock concentration of a mystery substance (MS) is 5 M. If the molar mass of MS is 532 g/mol, what’s the concentration of the stock concentration in g/mL? $$ 5 M = 5 \frac{mol}{L} $$ $$ 5 \frac{mol}{L} * 532 \frac{g}{mol} = 2,660 \frac{g}{L} $$ $$ 2,660 \frac{g}{L}* \frac{1L}{1,000 mL} = 2.66 \frac{g}{mL} $$
You will perform a serial dilution to get 100 uM of MS. Devise a plan to dilute a 5 M MS solution to 100 uM. How many dilution steps will we need? Which tubes should we use? Which pipettes?
We will need two empty microtubes. For the first step, we’ll use a P20 for the stock solution, and a P1000 for the water. For the second step, we’ll use a P20 for dilution 1, and a P200 for the water.
graph LR; A[stock solution 5M] -->|1µL stock into 499µL water| B[dilution 1: 10,000µM] B -->|1µL dilution1 into 99µL water| C[final dilution: 100 µM]
- Fill out the following chart to prepare a final reaction with 60 uL reaction volume. Why did we make 100 uM MS if we actually need 40 uM MS? Why not prepare 40 uM in serial dilutions?
| Reagent | Stock concentration | Desired concentration | Volume |
|---|---|---|---|
| Loading dye | 6X | 1X | 10 µL |
| MS | 100 uM | 40 uM | 24 µL |
| dH2O | n/a | n/a | 26 µL |
$$ C_{dye stock} V_{dye} = C_{dye final} V_{total} $$
$$ (6X) V_{dye} = (1X)* (60 µL) $$
$$ V_{dye} = \frac{60}{6} µL = 10 µL $$
$$ C_{MS stock} V_{MS} = C_{MS final} V_{total} $$
$$ (100 µM) V_{MS} = (40 µM)* (60 µL) $$
$$ V_{MS} = \frac{40*60 µM µL}{100 µM} = 24 µL $$
$$ V_{total} = V_{dye} + V_{MS} + V_{d H_2 O} $$
$$ 60 µL = 10 µL + 24 µL + V_{d H_2 O}$$
$$ V_{d H_2 O} = 60-10-24 µL = 26 µL $$
If we had 40 µM MS, then when we added the loading dye, it would be diluted below 40 µM. So we need to have a high enough concentration of MS, that we can add loading dye to 1X concentration and still reach a final MS concentration of 40 µM.
Lab
Part 1: Mixing Color
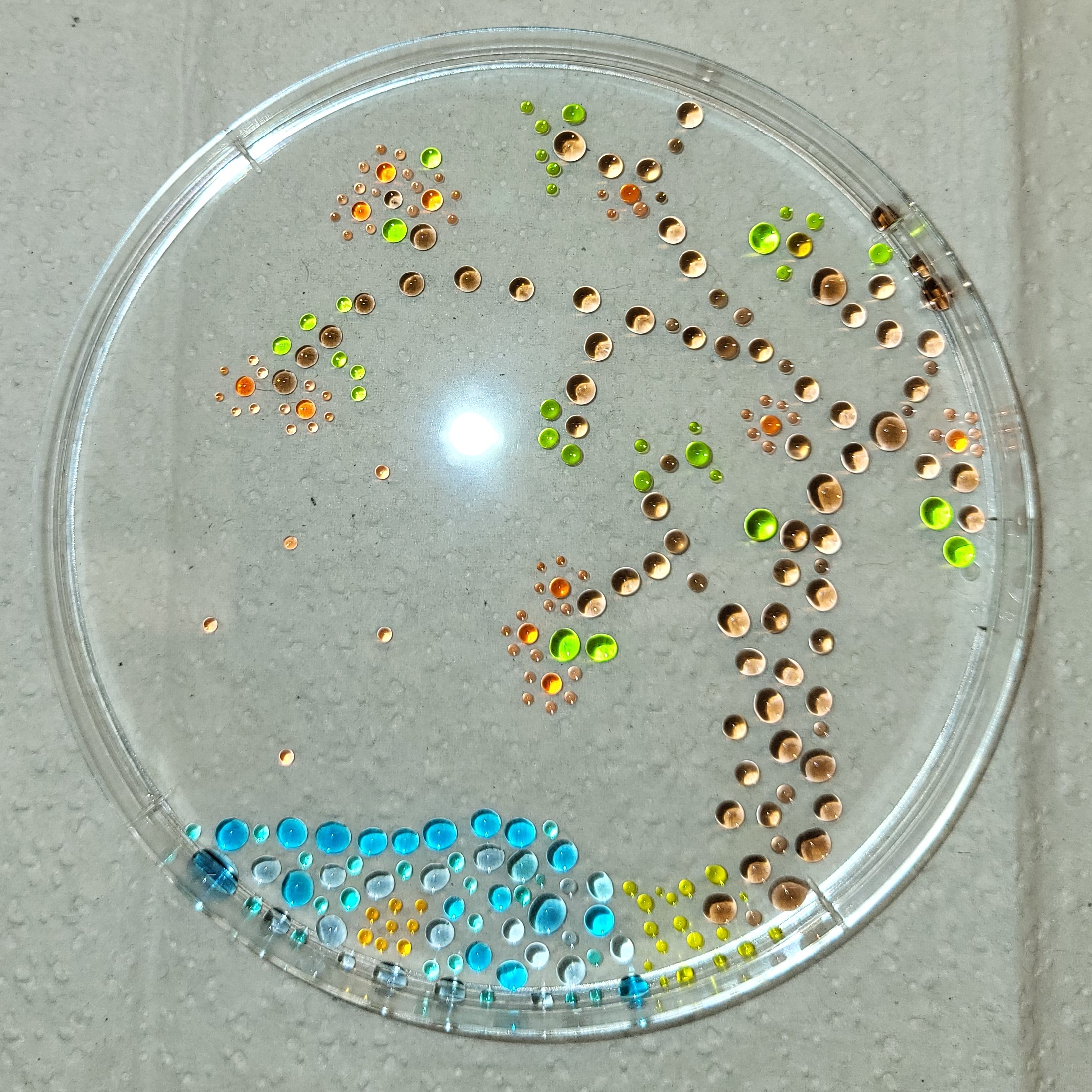
I made my stock color solutions by adding dye to approximately 5 ml water in three different 12 ml test tubes: 3 drops of yellow dye, 1 drop of blue dye, 2 drops of red dye, and then vortexing to mix.
Following the protocol, I obtained 6 colors. Step 4 was done with P20 and P200 in steps as described; steps 5 and 6 were done in single steps with the P1000 and P200 respectively.


I made an additional 4 colors as follows:
- Lime: 300 ul yellow, 50 ul blue
- Teal: 25 ul yellow, 600 ul blue
- Coral: 300 ul red, 50 ul yellow, 25 ul blue, 300 ul water
- Slate: 100 ul red, 300 ul blue, 300 ul water


My step 7 artwork is below and also the above cover image.


Part 2: Performing Serial Dilution
I don’t know what the Mystery Substance (MS) is supposed to be. I used some purified pUC19 plasmid, at a concentration of 197 ng/ul because that’s something I had available. It’s a double-stranded DNA, so the molecular weight would be around 660 g/mol per base pair, or a total of $660 \frac{g/mol}{bp} * 2.7 kb = 1,800 kg/mol$ approximately. Therefore, my stock concentration is $ 0.197 \frac{g}{L} * \frac{mol}{1,800,000 g} = 1.094E-7 mol/L = 0.11 uM = 110 nM$.
To get an arbitrarily chosen 1 nM stock, I did the following serial dilution:
$$ C_1 V_1 = C_2 V_2 $$ $$ (110 nM) V_1 = (10 nM)(50 ul) $$ $$ (110 nM) V_1 = 500 nM*ul $$ $$ V_1 = 4.5 ul $$
graph LR; A[0.11 uM stock solution ] -->|4.5 uL stock into 45.5 uL water| B[dilution: 0.01 uM]
Then I made the final solution according to the table. Again, the MS desired concentration was chosen arbitrarily.
| Reagent | Stock concentration | Desired concentration | Volume |
|---|---|---|---|
| Loading dye | 6X | 1X | 10 µL |
| MS | 10 nM | 1 nM | 6 µL |
| dH2O | n/a | n/a | 44 µL |
I added 20 ul of the final solution to an agarose gel (1% w/v). I made the agarose gel by measuring out 0.5 g of agarose, and adding it to 50 ml of 1x TAE buffer, then microwaving until melted. I poured it into a gel mold with a well comb and let set fully before putting into the electrophoresis set-up to practice loading into a well.

