Week 1: Principles and Practices
Synthetic Bioogy in Regenerative Medicine: Drug Delivery
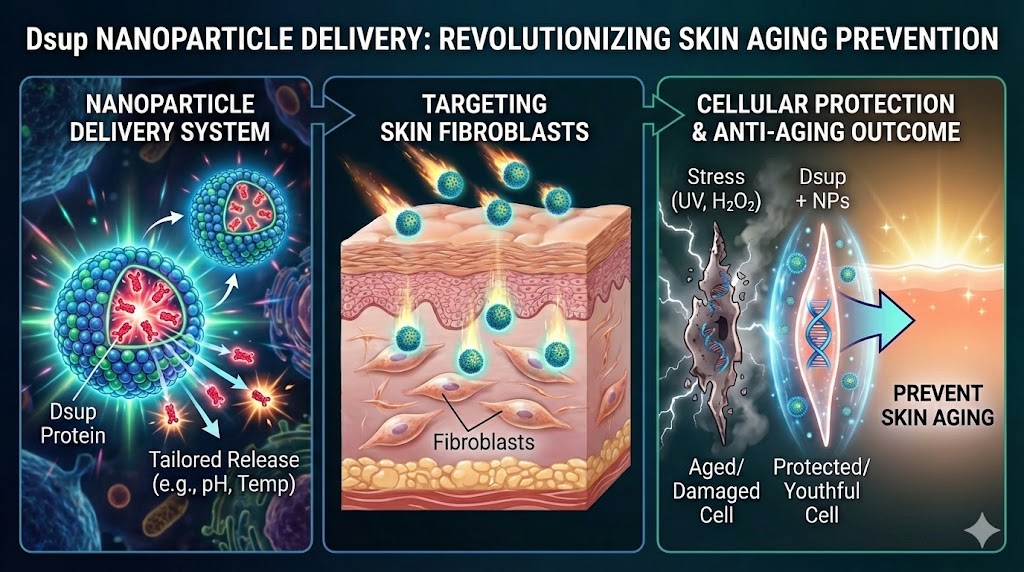

The convergence of biology and engineering offers innovative potential to address complex healthcare challenges. More specifically, regenerative medicine has advanced enormously through inspiration from nature. Nowadays, bioengineered materials can be adapted to mimic and integrate natural designs with intricate mechanisms found in living organisms, ecosystems, and evolutionary processes. The main goal is to develop new materials, devices, and systems that can restore and enhance tissue performance and function, leading to new therapeutic approaches. Several essential synthetic biology techniques are used toward this aim, such as genetic engineering, cellular reprogramming, cellular pathway engineering, CRISPR-Cas9, delivery systems, artificial cells and organs, stem cell engineering, biomechanics, and bioinformatics.
One particularly important tool is delivery systems. A common strategy to target specific locations is the use of nanoparticles (NPs). Due to their size and biocompatibility, NPs can breach biological barriers, penetrate deep tissues, and release therapeutic agents in a precisely controlled manner. Moreover, delivery system carriers can be tailored to respond to different conditions such as pH and temperature, enhancing treatment effectiveness while reducing unintended side effects.
Numerous studies have implemented this technique to deliver viral vectors for gene therapies in diseases such as hemophilia A and glioblastoma, as well as non-viral vectors, including proteins such as vascular endothelial growth factor. One particularly interesting protein that could be carried using NPs is the damage suppressor protein (Dsup), a nucleosome-binding protein found in the tardigrade Ramazzottius varieornatus (a resilient invertebrate commonly known as a water bear). This protein has been shown to significantly improve cell survival and growth by protecting against extreme stress conditions, specifically oxidative stress (hydrogen peroxide, H₂O₂) and UV-C irradiation in HEK293 human cells (human embryonic kidney cells).
Given the possibility of tailoring release conditions in nanoparticle-based delivery systems and the potential of Dsup to prevent DNA damage, I propose to study the delivery of Dsup using different types of nanoparticles into fibroblasts, the main cells found in skin. The skin is the largest organ of the human body and performs multiple functions, including homeostatic regulation; prevention of percutaneous loss of fluid, electrolytes, and proteins; temperature maintenance; sensory perception; and immune surveillance. This approach could help prevent cellular damage in degenerative phenomenon such as skin aging, which do affect every single person worldwide.
Governance and Policiy Goals for Ethical Future
Minimize Biological and Long-Term Risks
Ensure that nanoparticle carriers and delivered proteins do not induce genotoxicity, immune dysregulation, or unintended cellular adaptations.
Prevent long-term accumulation or persistence of nanoparticles in tissues.
Prevent Misuse or Dual-Use Risks
Avoid applications that could enable enhancement beyond therapeutic intent (e.g., extreme stress resistance for non-medical or military use).
Ensure that the technology is not repurposed for harmful or coercive applications.
Promote Responsible and Equitable Access
Ensure that benefits are not restricted to cosmetic or luxury applications while excluding broader public health needs.
Encourage transparency and public engagement regarding intended uses.
Governence Actions
Mandatory Preclinical Risk Assessment Framework
Actors: Academic researchers, funding agencies, Institutional Review Boards (IRBs)
Purpose: Current research on nanoparticle-based protein delivery systems often prioritizes short-term efficacy and cytotoxicity. This action proposes expanding existing requirements to include standardized assessments of long-term genomic stability, epigenetic alterations, immune responses, and nanoparticle persistence prior to clinical translation.
Design:
Funding agencies require comprehensive long-term safety evaluations as a condition for grant approval.
Scientific journals mandate extended safety datasets for publication.
IRBs implement nanoparticle-specific risk assessment protocols during project approval.
Assumptions:
Long-term biological effects can be reasonably predicted using advanced in vitro and animal models.
Research institutions possess or can access the infrastructure needed for extended safety testing.
Risks of Failure & “Success”:
Failure: Increased regulatory requirements may slow innovation or disproportionately impact under-resourced laboratories.
Success Risk: Excessive standardization could discourage exploratory research or unconventional delivery strategies.
Use-Based Regulatory Classification of Nanoparticle Applications
Actors: Federal regulators, public health authorities, regulatory agencies
Purpose: Instead of regulating nanoparticle delivery systems solely based on their material composition, this action proposes classifying them according to intended use (therapeutic, preventive, cosmetic, or enhancement-related), allowing for proportional oversight.
Design:
Regulatory agencies establish distinct approval pathways based on application category.
Therapeutic and disease-prevention uses receive prioritized evaluation.
Enhancement-oriented or cosmetic applications are subject to stricter scrutiny or limitations.
Assumptions:
Clear and enforceable distinctions between therapeutic and enhancement uses can be maintained.
Developers will accurately disclose the intended use of their products.
Risks of Failure & “Success”:
Failure: Ambiguous classifications could create regulatory loopholes.
Success Risk: Over-restriction may incentivize unregulated or informal markets for enhancement applications.
Technical Safeguards Embedded in Delivery System Design
Actors: Bioengineers, biotechnology companies, translational researchers
Purpose: This action promotes the integration of safety and misuse-prevention mechanisms directly into nanoparticle delivery systems to reduce the risk of unintended or unethical applications.
Design:
Nanoparticles engineered to degrade rapidly outside specific tissue microenvironments.
Activation of therapeutic cargo dependent on cell-type-specific enzymes or physiological conditions.
Regulatory agencies offer expedited review pathways for designs incorporating built-in safeguards.
Assumptions:
High levels of biological specificity can be reliably engineered into delivery systems.
Added design complexity does not compromise therapeutic performance.
Risks of Failure & “Success”:
Failure: Biological variability may limit the effectiveness of technical safeguards.
Success Risk: Increased development costs could reduce accessibility, particularly in low-resource settings.
| Does the option: | Option 1 | Option 2 | Option 3 |
|---|---|---|---|
| Enhance Biosecurity | |||
| • By preventing incidents | 1 | 2 | 1 |
| • By helping respond | 2 | 1 | 3 |
| Foster Lab Safety | |||
| • By preventing incidents | 1 | 2 | 2 |
| • By helping respond | 1 | 2 | 3 |
| Protect the environment | |||
| • By preventing incidents | 2 | 2 | 1 |
| • By helping respond | 2 | 3 | 2 |
| Other considerations | |||
| • Minimizing costs and burdens to stakeholders | 3 | 1 | 2 |
| • Feasibility | 2 | 1 | 2 |
| • Not impede research | 3 | 1 | 2 |
| • Promote constructive applications | 1 | 1 | 1 |
Governance Prioritization and Recommendation
Based on the scoring, I recommend prioritizing Option 1 (Mandatory Preclinical Risk Assessment) and Option 2 (Use-Based Regulatory Classification), with selective use of Option 3 (Embedded Technical Safeguards) in later-stage applications.
Option 1 provides the strongest protection against biological and laboratory risks, which is essential given uncertainties around the long-term effects of nanoparticle-mediated protein delivery. Although it increases research burden, this trade-off is justified at early stages where prevention is most effective.
Option 2 adds proportional, use-based oversight that is highly feasible and minimizes unnecessary constraints on innovation, particularly during translation and commercialization.
Option 3 should be incentivized rather than required, as embedded safeguards reduce misuse risks but may increase complexity and costs. Together, this layered approach balances safety, feasibility, and responsible innovation.
References
Springer Reference. (2016). Nanoparticles in drug delivery. In Encyclopedia of Nanotechnology. Springer.
https://doi.org/10.1007/978-3-662-47398-6_4Springer. (2024). Advances in regenerative medicine and tissue engineering. In Handbook of Regenerative Medicine (pp. 521–525). Springer.
https://doi.org/10.1007/978-3-031-87744-5Madkour, L. H., et al. (2021). Nanoparticles and their biomedical applications. Biology, 10(10), 970.
https://doi.org/10.3390/biology10100970World Health Organization. (2022). Global guidance framework for the responsible use of the life sciences: Mitigating biorisks and governing dual-use research. WHO.
https://iris.who.int/handle/10665/362313Church, G. M., & Baker, D. (2024). Protein design meets biosecurity. Science, 383(6679), eado1671.
https://doi.org/10.1126/science.ado1671