Week 1 HW: Principles and Practices

1) Describe a biological engineering application or tool you want to develop and why.
Throughout my previous research as a new biodesigner, I have been particularly drawn to two themes: the intricate relationship between the gut microbiota and its effects on human physical and mental health, and the fascinating world of fungi and their broader implications for planetary health. I have deepened my research into psilocybin and related tryptamine alkaloids (substances that activate serotonin 5-HT receptors), which are already being investigated clinically for neuropsychiatric conditions (Xi et al., 2023), but what especially interests me is emerging evidence that some of their effects may be mediated by the gut microbiota rather than the brain alone (Caspani et al., 2024). There is still a lack of research on the precise impact that serotonergic psychedelics have on the structure and composition of the gut microbiota, even though animal studies suggest possible links (Császár-Nagy et al., 2022). A survey of wild‑type rats has shown changes in the abundance of spore‑forming gut bacteria following oral administration of serotonin, suggesting that serotonergic compounds such as psilocybin could directly or indirectly reshape microbial communities (Fung et al., 2019).
Beyond psilocybin, the traditional use of other psychoactive organisms also points to underexplored antimicrobial and immunomodulatory effects. For instance, the peyote cactus (Lophophora williamsii) has been reported to display activity against multiple penicillin‑resistant strains of Staphylococcus aureus (McCleary et al., 1960). Such examples raise broader questions about how these organisms interact with microbes and the host’s physiology. These kinds of plants, fungi, and compounds have long histories in traditional medicine yet remain only partially understood within Western biomedical frameworks (Doesburg-van Kleffens et al., 2023). I believe that the intersection of psychedelics, microbiota, and immune function is a promising area for synthetic biology yet to be further explored, with potential not only for already studied mental health conditions but also for inflammatory bowel diseases (IBD) (Qureshi et al., 2025). Rather than viewing these compounds solely through a brain‑centric lens, it would be interesting to investigate their therapeutic potential through the gut microbiota as a primary interface rather than a secondary effect.
Psychedelic‑assisted therapy is currently highly regulated, expensive, and accessible to very few people (Rea et Wallace, 2021). From a biodesign and synthetic biology perspective, biosynthesising psilocybin in a bacterial host such as Escherichia coli through recombinant DNA technology (Keller et al., 2025) could potentially provide a more controllable, scalable, and eventually more affordable route to producing psilocybin or related analogues.
In the context of the HTGAA course, I therefore propose to focus on a minimal and safe fragment of this broader concept, with the following objectives:
- Investigate how natural serotonergic psychedelics and related natural compounds might interact with the gut microbiota- immune axis, with a focus on IBD and gut inflammations.
- Design a conceptual framework for microbial psilocybin production in E. coli (Adams et al., 2019) as a platform for future microbiome‑targeted therapies.
- Prototype an E. coli experimental system expressing a heterologous lyase enzyme from a psilocybin-related tryptamine biosynthetic pathway (Abrahms et al., 2025), assembled via PCR-based cloning methods.
- Apply CRISPR‑based editing to modulate the expression of the heterologous enzyme and characterise the resulting change in a readout (through colour or fluorescence) to determine if the right sequence in E.Coli is being targeted.
- Develop a simple in silico pathway model to explore where metabolic bottlenecks might occur in a full psilocybin biosynthesis pathway in E. coli (Irvine et al., 2023).
- Reflect on ethical, regulatory, and accessibility questions around engineering psychoactive compounds and further document how these considerations shape the design of the project.
- Abrahms, Z.N., Sen, A.K. and Jones, J.A. (2025).Pathway engineering for the biosynthesis of psychedelics. Current Opinion in Biotechnology, 94, pp.103314–103314. doi:https://doi.org/10.1016/j.copbio.2025.103314.
- Adams, A.M., Kaplan, N.A., Wei, Z., Brinton, J.D., Monnier, C.S., Enacopol, A.L., Ramelot, T.A. and Jones, J.A. (2019). In vivo production of psilocybin in E. coli. Metabolic Engineering, [online] 56, pp.111–119. doi:https://doi.org/10.1016/j.ymben.2019.09.009.
- Caspani, G., Ruffell, S.G.D., Tsang, W., Netzband, N., Rohani-Shukla, C., Swann, J.R. and Jefferies, W.A. (2024). Mind over matter: the microbial mindscapes of psychedelics and the gut-brain axis. Pharmacological Research, 207, p.107338. doi:https://doi.org/10.1016/j.phrs.2024.107338.
- Császár-Nagy, N., Bob, P. and Bókkon, I. (2022). A Multidisciplinary Hypothesis about Serotonergic Psychedelics. Is it Possible that a Portion of Brain Serotonin Comes From the Gut? Journal of Integrative Neuroscience, 21(5), p.148. doi:https://doi.org/10.31083/j.jin2105148.
- Doesburg-van Kleffens, M., Zimmermann-Klemd, A.M. and Gründemann, C. (2023). An Overview on the Hallucinogenic Peyote and Its Alkaloid Mescaline: The Importance of Context, Ceremony and Culture. Molecules, [online] 28(24), p.7942. doi:https://doi.org/10.3390/molecules28247942.
- Fung, T.C., Vuong, H.E., Luna, C.D.G., Pronovost, G.N., Aleksandrova, A.A., Riley, N.G., Vavilina, A., McGinn, J., Rendon, T., Forrest, L.R. and Hsiao, E.Y. (2019). Intestinal serotonin and fluoxetine exposure modulate bacterial colonization in the gut. Nature Microbiology. doi:https://doi.org/10.1038/s41564-019-0540-4.
- Keller, M.R., McKinney, M.G., Sen, A.K., Guagliardo, F.G., Hellwarth, E.B., Islam, K.N., Kaplan, N.A., Gibbons, W.J., Kemmerly, G.E., Meers, C., Wang, X. and Jones, J.A. (2025). Psilocybin biosynthesis enhancement through gene source optimization. Metabolic Engineering, [online] 91, pp.119–129. doi:https://doi.org/10.1016/j.ymben.2025.04.003.
- McCleary, J.A., Sypherd, P.S. and Walkington, D.L. (1960). Antibiotic activity of an extract of peyote (Lophophora Williamii (Lemaire) Coulter). Economic Botany, 14(3), pp.247–249. doi:https://doi.org/10.1007/bf02907956.
- Qureshi, O., Cowley, J., Pegg, A., Cooper, A.J., Gordon, J., Brady, C.A., Belli, A., Butterworth, S., Upthegrove, R., Andrews, N. and Barnes, N.M. (2025). Are we hallucinating or can psychedelic drugs modulate the immune system to control inflammation? British journal of pharmacology, [online] p.10.1111/bph.70138. doi:https://doi.org/10.1111/bph.70138.
- Xi, D., Berger, A., Shurtleff, D., Zia, F.Z. and Belouin, S. (2023). National Institutes of Health psilocybin research speaker series: State of the science, regulatory and policy landscape, research gaps, and opportunities. Neuropharmacology, [online] 230, p.109467. doi:https://doi.org/10.1016/j.neuropharm.2023.109467.
- Risk assessment for gut-specific harm: to safely develop this kind of medication or therapy, it will require a longitudinal patient gut-microbome monitoring prior to and post-dosing to refine safety protocols and the detection of any adverse shifts.
- Accessibility whilst respecting indviduality of the gut microbiome: creating an accessible medication targeting something so unique to the individual will be a challenge. There are now affordable baseline microbiome profiling kits (e.g. Feel Gut - Microbiome Test Kit - 250£) to screen high-risk patients pre-treatment, while developing 2–3 engineered E. coli strains producing dosage variants (low/medium/high) based on symptom severity or gut sensitivity.
- Value of traditional knowledge: crediting and collaborating with communities that have historically used psychedelic organisms for medical approaches through benefit-sharing agreements (BSAs).
- Transparency and accountability: Create traceable audit trails for strains and products to ensure responsibility if misuse occurs and build public trust through open data sharing.
- DiliTrust (2025). Understanding Audit Trails: Implementation, Types, and Best Practices. [online] Dilitrust. Available at: https://www.dilitrust.com/audit-trail/ [Accessed 7 Feb. 2026].
- FeelGut (2026). Gut Microbiome Health Test. [online] Feel Gut. Available at: https://feelgut.co.uk/products/gut-microbiome-health-test?gad_source=1&gad_campaignid=21790867659&gbraid=0AAAAAqF2ctBqNuD8W2LgDdGB8xmh-dJkI&gclid=Cj0KCQiAhaHMBhD2ARIsAPAU_D4EnlJcLWOuT0Ol-Gz0qDHbGOnE0a_08EaWu5OmccHvfelHGAJLgeUaAmI0EALw_wcB [Accessed 7 Feb. 2026].
- Ogilvy.com.au (2024). Shared Success: What is benefit sharing and why does it matter? - Insight - MinterEllison. [online] Minterellison.com. Available at: https://www.minterellison.com/articles/what-is-benefit-sharing-and-why-does-it-matter [Accessed 7 Feb. 2026].
- Secretariat of the Convention on Biological Diversity (2011). Convention on Biological Diversity: ABS Theme Access and benefit-sharing. [online] Available at: https://www.cbd.int/abs/infokit/revised/web/factsheet-abs-en.pdf [Accessed 7 Feb. 2026].
- COLLECTIVE DATABASE:
- PATIENT GUT-MICROBIOME SCREENING: Call for the pre-screening of patients’ gut microbiome to ensure appropriate candidates. This also calls for collaboration with a pharmaceutical partner to deliver these kits directly to potential patients or to hospitals and institutions where the samples will be analysed to determine whether they are suitable for treatment of the patient's gut microbiome. This will ensure safety and a reduction of side effects, and better results by than accordingly selecting which strain would benefit the patient the most. There are still some risks considering ‘false negative’ testing, which could therefore make it difficult to fully confirm the appropriate treatment, as well as the potential increase in price of the treatment if the gut-microbiome data were to become a Pharma IP monopoly, limiting access due to often increasing prices (Dosi et al., 2023).
- CROSS COLLABORATION Call for the implementation of royalty funds for indigenous cultures that have contributed to the development of this project with their knowledge on the medical application of psychedelics to ensure an ethical and transparent development of the project. Furthermore, developing a wider web of collaboration with governments, scientists, hospitals, psychologists, and doctors to meet and discuss/review strains, trials and dosages. This complex cross-collaboration would only be possible with an FDA approval of the usage of such psychedelics to begin with, which would require a long time, many trials and solid evidence of success at a high success rate level (Lamkin, 2022).
- Dosi, G., Marengo, L., Staccioli, J. and Virgillito, M.E. (2023). Big Pharma and Monopoly capitalism: a long-term View.Structural Change and Economic Dynamics, [online] 65, pp.15–35. doi:https://doi.org/10.1016/j.strueco.2023.01.004.
- Lamkin, M. (2021). Prescription Psychedelics: The Road from FDA Approval to Clinical Practice. The American Journal of Medicine, 135(1). doi:https://doi.org/10.1016/j.amjmed.2021.07.033.
- Grogan, S. and Preuss, C. (2023). Pharmacokinetics. [online] PubMed. Available at: https://www.ncbi.nlm.nih.gov/books/NBK557744/ [Accessed 9 Feb. 2026].
- Aylin Elçi (2025). Switzerland is home to Europe’s only psychedelics treatment. [online] SWI swissinfo.ch. Available at: https://www.swissinfo.ch/eng/multinational-companies/switzerland-is-home-to-europes-only-psychedelics-treatment/89195943 [Accessed 7 Feb. 2026].
- Bates, S. (2019). Base Pair. [online] Genome.gov. Available at: https://www.genome.gov/genetics-glossary/Base-Pair [Accessed 8 Feb. 2026].
- Clent Life Science (2024). High-fidelity DNA Polymerases & When to use them. [online] Clent Life Science. Available at: https://clentlifescience.co.uk/high-fidelity-dna-polymerases-and-when-to-use-them/ [Accessed 8 Feb. 2026].
- Liu, Y., Yang, Q. and Zhao, F. (2021). Synonymous but Not Silent: The Codon Usage Code for Gene Expression and Protein Folding. Annual Review of Biochemistry, 90(1), pp.375–401. doi:https://doi.org/10.1146/annurev-biochem-071320-112701.
- Rajbanshi, B. and Guruacharya, A. (2025). codonGPT: reinforcement learning on a generative language model enables scalable mRNA design. Nucleic Acids Research, [online] 53(22). doi:https://doi.org/10.1093/nar/gkaf1345.
- ATDBio Ltd (2005). ATDBio - Solid-phase oligonucleotide synthesis. [online] atdbio.com. Available at: https://atdbio.com/nucleic-acids-book/Solid-phase-oligonucleotide-synthesis [Accessed 8 Feb. 2026].
- Glen Research (2026). Glen Report 21.211 - TECHNICAL BRIEF – Synthesis of Long Oligonucleotides. [online] Glenresearch.com. Available at: https://www.glenresearch.com/reports/gr21-211 [Accessed 8 Feb. 2026].
- McLaughlin, L. (2025). What Is Oligonucleotide Synthesis? Phosphoramidite oligonucleotide synthesis. [online] Biotechnologyreviews.com. Available at: https://www.biotechnologyreviews.com/p/what-is-oligonucleotide-synthesis [Accessed 8 Feb. 2026].
- Michael Mühlegger (2025). Oligonucleotide manufacturing – challenges & solutions. [online] Single Use Support: Pionneering Biopharma. Available at: https://www.susupport.com/blogs/biopharmaceutical-products/oligonucleotide-manufacturing-challenges-solutions [Accessed 8 Feb. 2026].
- Lopez, M.J. and Mohiuddin, S.S. (2024). Biochemistry, Essential Amino Acids. [online] PubMed. Available at: https://www.ncbi.nlm.nih.gov/books/NBK557845/ [Accessed 8 Feb. 2026].
- Jurassic-Pedia (2024). Lysine Contingency (S/F) / (S/F-T/G) – Jurassic-Pedia. [online] Jurassic-Pedia. Available at: https://www.jurassic-pedia.com/lysine-contingency-sf/ [Accessed 9 Feb. 2026].
Reference List:
2) Next, describe one or more governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future, like ensuring non-malfeasance (preventing harm). Break big goals down into two or more specific sub-goals.
Policies Goal: To ensure microbial psilocybin production for microbiome-targeted IBD therapy/medication prioritises safety, equitable access despite microbiome variability, respect for traditional knowledge and transparency to build public trust and prevent harm.
Sources:
3) Next, describe at least three different potential governance “actions” by considering the four aspects below (Purpose, Design, Assumptions, Risks of Failure & “Success”).
To meet these policy goals, I propose the following three potential governance actions:
References:
4) Next, score (from 1-3 with, 1 as the best, or n/a) each of your governance actions against your rubric of policy goals. The following is one framework but feel free to make your own:
Scale: 1-3 (1: Most effective, 2: Moderately effective, 3: Least effective, or N/A)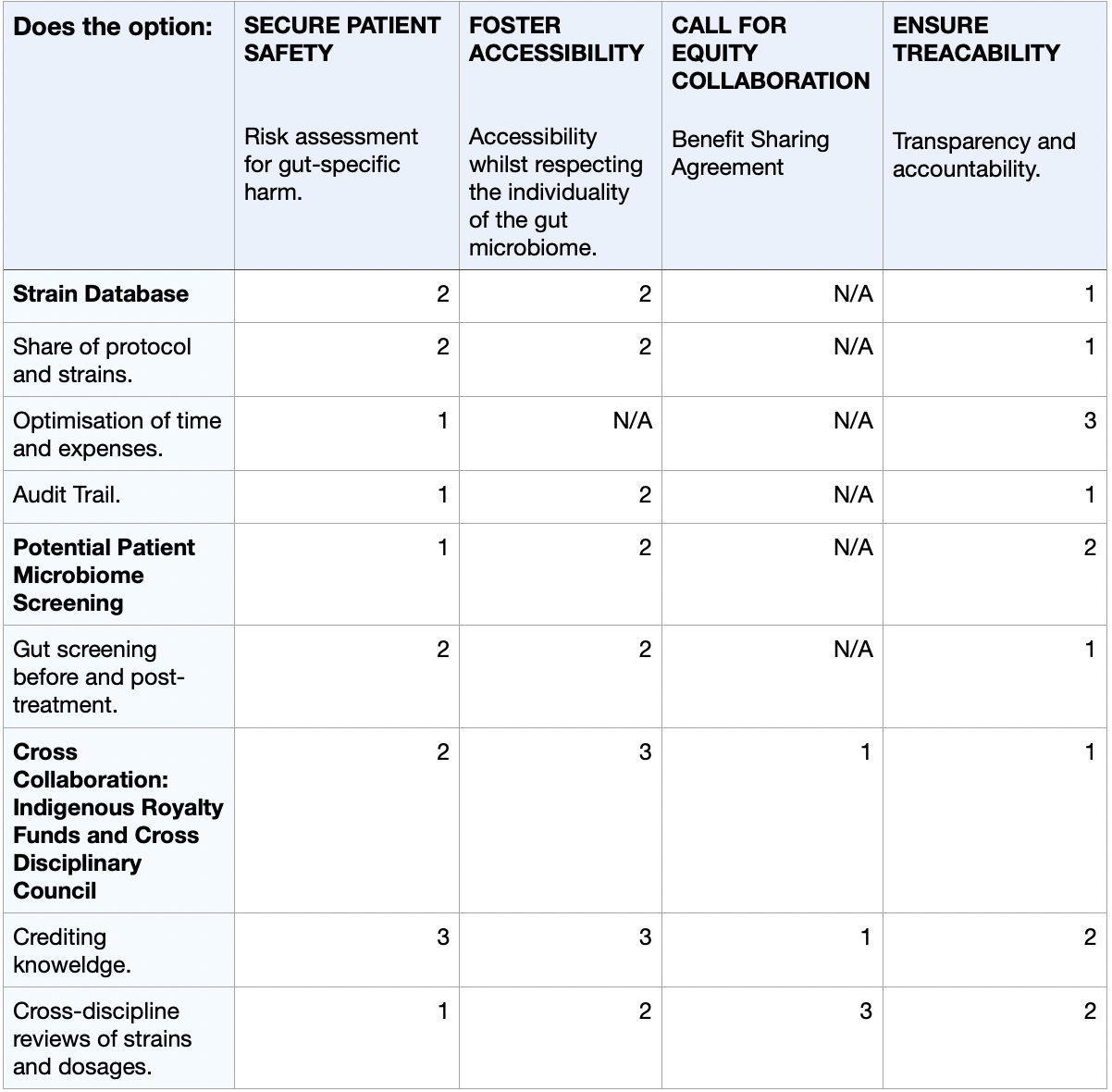

5) Last, drawing upon this scoring, describe which governance option, or combination of options, you would prioritize, and why. Outline any trade-offs you considered as well as assumptions and uncertainties.
I propose the prioritisation of the strain database, as well as the microbiome pre-screening, as the foundational governance actions for the development of microbial psilocybin. Given the strict regulations surrounding the use of psychedelic drugs in the medical world, transparency, feasability and safety are essential for governmental approval. Strategically, developing this project in countries like Switzerland or the Netherlands would offer optimal conditions. In Zurich and Geneva (Switzerland), research on the medical use of psychedelic compounds, such as psilocybin or LSD, in clinical psychiatric trials is continuously growing (Elçi, 2025). Additionally, their biotechnological infrastructure would allow for an ideal environment to conduct the development of such a project. On the other hand, the legal use of psilocybin truffles in the Netherlands should also be considered as a place of interest. Overlooking this as phases, the initial phase should consist of the launch of the strain database for safety and optimisation, followed by mandating affordable gut-microbiome screening. Lastly, validating via in-silico computational models the prediction of human gut-microbiome-psilocybin interactions through pharmacokinetic data (Grogan and Preuss, 2023) to build towards the potential commercialisation of such a product.
List of References:
Assignment (Week 2 Lecture Prep)
Homework Questions from Professor Jacobson:
1)Nature’s machinery for copying DNA is called polymerase. What is the error rate of polymerase? How does this compare to the length of the human genome. How does biology deal with that discrepancy?
The error rate ratio of polymerase is 1:10^6 base pairs, equivalent to one mistake every million bases. In comparison, the human genome consists of approximately 3 billion base pairs (3 x 10^9) (National Human Genome Research Institute, 2026), meaning each replication introduces around 3000 errors (3x10^9 x 10^6), leading to genetic mutations. In synthetic biology, high-fidelity DNA polymerases have proven to have much lower error rates for PCR cloning of long biosynthetic pathways. These polymerases possess 3’ to 5’ exonuclease proofreading domains that detect mismatches through structural perturbations and immediately remove them before continuing synthesis. (Clent Life Science, 2024).
2)How many different ways are there to code (DNA nucleotide code) for an average human protein? In practice, what are some of the reasons that all of these different codes don’t work to code for the protein of interest?
There are astronomically many possible ways to code for an average human protein. For example, a 300-amino-acid protein may have 10^100 different synonymous codon combinations (Rajbanshi and Guruacharya, 2025). However, most fail in practice because cells prefer specific codons (codon usage bias), which causes rare codons to slow down the tRNA process. Consequently, this creates nonuniform ribosome decoding rates on mRNAs and in turn disrupts the contranslational protein folding process, which is essential for proper protein function (Liu et al., 2021).
References:
Homework Questions from Dr. LeProust:
1)What’s the most commonly used method for oligo synthesis currently?
Solid-phase phosphoramidite is currently the most commonly used method for oligo synthesis. It is a cyclical four-step phosphoramidite synthesis method (McLaughlin, 2025), which was developed in 1981 by Marvin Caruthers.
2) Why is it difficult to make oligos longer than 200nt via direct synthesis?
Making oligos longer than 200 nucleotides via direct synthesis is difficult due to cumulative yield losses. During this synthesising method, one nucleotide is added at a time. Although there is a high success rate (99%), each added nucleotide will contribute to yield loss (ATDBio Ltd, 2005). At the 200th nucleotide added (0.99^200), the overall yield production will have a new success rate of 0.135%, resulting in extremely low yield and consequently resulting in a waste of expensive reagents and purification time (Mühlegger,2025).
3) Why can’t you make a 2000bp gene via direct oligo synthesis?
As mentioned in the last question, chemical DNA synthesis adds only one nucleotide at a time, at a 99% success rate. Taking into consideration that we only use one strand of DNA for the synthesis, 0.99^2000 would reduce the success rate by 0.0002% yield for a 2000bp gene. Considering this extremely low amount, it would make it practically impossible for the yield to be detected for the purification process.
References:
Homework Question from George Church:
1) What are the 10 essential amino acids in all animals and how does this affect your view of the “Lysine Contingency”?
The essential amino acids are: Histidine (His), Isoleucine (Ile), Leucine (Leu), Lysine (Lys), Methionine (Met), Phenylalanine (Phe), Threonine (Thr), Tryptophan (Trp), Valine (Val), and Arginine (ARG). (Lopez and Mohiuddin, 2024).
The Lysine Contingency was a genetically engineered fail‑safe created by Dr Henry Wu in the late 1980s to prevent Jurassic Park’s dinosaurs from synthesising lysine. The idea was that park staff would have to supplement the animals with lysine to keep them alive, and if any dinosaur escaped Isla Nublar, it would die without this dietary supply. This was intended as a strategy to protect outside ecosystems. However, this design was fundamentally flawed. In 1997, Dr Sarah Harding showed that the dinosaurs could obtain enough lysine from their environment through their normal diet, making the contingency ineffective in practice. Since all animals already depend on dietary lysine in the first place, the Contingency never provided the precise control the park claimed (Jurassic-Pedia, 2024).
I agree with Dr Sorkin’s view that, even though these dinosaurs were cloned, they still deserved rights as living animals. Humans chose to recreate this extinct species, so it seems ethically wrong that we should also reserve the right to let them die for our convenience or as a simplistic safety measure. This raises a wider question about how much power humans should have over organisms they create or modify. In my view, these ethical discussions should happen before such cloning work begins, not after problems appear. Overall, the Lysine Contingency feels poorly thought through and aimed at solving a problem that should never have been framed that way in the first place.
References: