<YOUR NAME HERE> — HTGAA Spring 2026
About me
Contact info
Homework
- Week 1 HW: Principles and Practices
- DNA Read, Write, and Synthesis
- Genetic Circuits - 1
- Protein design - 1
- Week-07 Genetic Circuits - 2
Week 1 HW: Principles and Practices
Biological Engineering Application or Tool The proposed application is an AI-guided protein therapeutic discovery and bioproduction platform. The system uses machine learning–based protein design models to generate novel therapeutic protein candidates, such as antimicrobial proteins, enzymes, or biologics optimized for stability and activity. These candidates are then evaluated for manufacturability and functional performance using controlled bioproduction workflows, including microbial expression or cell-free systems. This application reflects an emerging paradigm in biopharmaceutical development, where AI accelerates early-stage discovery while scalable bioproduction determines clinical and commercial feasibility. However, as AI enables rapid de novo protein design, many generated sequences may lack homology to known natural proteins, introducing novel biosecurity and safety risks if not properly governed.
DNA Read, Write, and Synthesis
This week, we were tasked to utilize different tools to be able to virtually read, write, and visualize using samples like lambda DNA from Escherichia coli and the Tumor suppressor gene from humans. Part 1 - Introduction and DNA digest. Gel Electrophoresis Gel - material Electro - Electric Phoresis - to transport It is a method used to transport charged materials using an electric field through a gel (a Semi-liquid substance). Digested fragments of Lambda DNA
Part - 1 What are some components in the Phusion High-Fidelity PCR Master Mix, and what is their purpose? Phusion High-Fidelity PCR Master Mix, commonly produced by Thermo Fisher Scientific, contains a high-fidelity DNA polymerase with proofreading ability, a reaction buffer that maintains optimal conditions, Mg²⁺ ions as a cofactor, dNTPs as building blocks, and stabilizing additives. Together, these components enable accurate and efficient DNA amplification with a low error rate. What are some factors that determine primer annealing temperature during PCR?
A. Conceptual Questions How many molecules of amino acids do you take with a piece of 500 grams of meat? (On average, an amino acid is ~100 Daltons) Answer 1 Dalton ≈ 1 g/mol Average amino acid ≈ 100 g/mol If you eat 500 g of (pure) amino acids: number of moles = Gm/ Tm = 500g/100g/mol
Assignment Part 1: Intracellular Artificial Neural Networks (IANNs) What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions? Intracellular artificial neural networks provide more flexible and nuanced behavior than traditional Boolean genetic circuits because they can process inputs in a graded, continuous manner rather than simple on or off states. This allows cells to integrate multiple signals and produce proportional responses, making them better suited for complex decision making and pattern recognition inside biological systems. Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
The proposed application is an AI-guided protein therapeutic discovery and bioproduction platform. The system uses machine learning–based protein design models to generate novel therapeutic protein candidates, such as antimicrobial proteins, enzymes, or biologics optimized for stability and activity. These candidates are then evaluated for manufacturability and functional performance using controlled bioproduction workflows, including microbial expression or cell-free systems.
This application reflects an emerging paradigm in biopharmaceutical development, where AI accelerates early-stage discovery while scalable bioproduction determines clinical and commercial feasibility. However, as AI enables rapid de novo protein design, many generated sequences may lack homology to known natural proteins, introducing novel biosecurity and safety risks if not properly governed.
The overarching governance goal is to ensure that AI-enabled protein drug discovery and bioproduction contribute to a safe, ethical, and socially beneficial future, while preventing misuse or unintended harm. This goal can be divided into the following sub-goals:
2.1. Non-malfeasance and biosecurity
Prevent the accidental or intentional creation of harmful, toxic, or dual-use proteins enabled by AI-assisted design.
2.2. Responsible scale-up and traceability Ensure that the transition from digital protein design to physical bioproduction is secure, auditable, and accountable.
2.3. Preservation of constructive innovation Maintain open scientific collaboration and efficient therapeutic development without imposing unnecessary regulatory burdens that would slow innovation.
These goals align with arguments advanced by Baker and Church, who emphasize that enhanced biosecurity should be embedded into protein design and DNA synthesis infrastructure without undermining transparency or information sharing.
3.1 Governance Action 1: Integrated Safety Screening and Secure Sequence Logging
Purpose
Currently, AI protein design pipelines primarily optimize for functional performance, and existing biosecurity measures rely heavily on sequence homology screening at the DNA synthesis stage. As Baker and Church note, this approach is increasingly insufficient for de novo designed proteins. This project proposes an integrated governance mechanism that embeds mandatory AI-based safety screening and secure sequence logging directly into the protein design and bioproduction pipeline.
Design
This governance approach would be implemented through collaboration among AI tool developers, biopharmaceutical companies, and DNA synthesis or bioproduction providers. All AI-generated protein sequences would undergo computational screening for toxicity, virulence, and dual-use potential before synthesis approval. Once synthesized, sequences would be logged in encrypted repositories tied to production systems, with access restricted to exceptional circumstances such as public health investigations. This design enables traceability and accountability while protecting intellectual property and minimizing interference with normal research workflows.
Assumptions
This approach assumes that predictive models for protein toxicity and risk are sufficiently accurate to identify high-risk candidates and that industry actors are willing to adopt shared security standards. It also assumes that secure logging can be implemented in a way that does not expose proprietary information or discourage legitimate research.
Risks of Failure and “Success”
Potential failure modes include false negatives that allow harmful proteins to proceed or false positives that block legitimate therapeutic candidates. Additionally, if logging systems are unevenly implemented, malicious actors may bypass regulated platforms. A potential risk of “success” is increased centralization of bioproduction infrastructure, which could disadvantage smaller labs or researchers in low-resource settings if access is not equitably managed.
3.2 Governance Action Option 2
Tiered Access and Credentialing for Advanced Protein Design Models
Purpose
Currently, many AI protein design tools are becoming increasingly accessible with minimal differentiation between low-risk exploratory use and high-risk de novo protein generation. This action proposes a tiered access system where more powerful generative protein design capabilities require additional credentials, training, or institutional affiliation.
Design
AI tool providers and research institutions would implement access tiers based on user role, training completion, and intended application. Basic design and analysis features would remain widely accessible, while advanced generative functions (e.g., unrestricted de novo protein design) would require completion of biosecurity and ethics training, institutional oversight, or project-level approval. This mirrors governance models used in high-performance computing, clinical data access, and human-subjects research.
Assumptions
This approach assumes that access restrictions can meaningfully reduce misuse without pushing users toward unregulated alternatives. It also assumes institutions are capable of fairly and consistently evaluating access requests.
Risks of Failure and “Success”
If too restrictive, tiered access could slow innovation or disadvantage independent researchers and low-resource institutions. If too permissive, it may fail to deter misuse. A risk of “success” is the normalization of credential-based gatekeeping that could reinforce existing inequities in global research participation.
3.3 Governance Action Option 3
Safety-by-Design Standards Linked to Incentives and Recognition
Purpose
While safety measures are often framed as compliance requirements, this action reframes governance as an incentive-based system that rewards early integration of biosecurity and safety considerations into AI-driven protein design and bioproduction.
Design
Funding agencies, journals, and investors would establish safety-by-design criteria as part of grant evaluation, publication standards, and due diligence. Projects that demonstrate integrated risk assessment, secure production workflows, and ethical reflection would receive preferential funding, expedited review, or public recognition. This approach aligns governance with existing academic and commercial reward structures rather than relying solely on enforcement.
Assumptions
This approach assumes that researchers and companies respond strongly to funding, publication, and reputational incentives. It also assumes evaluators have sufficient expertise to assess safety claims without turning the process into box-checking.
Risks of Failure and “Success”
If poorly designed, incentives may encourage superficial compliance rather than genuine risk mitigation. A risk of “success” is that safety standards become rigid or outdated, unintentionally discouraging novel approaches that do not fit existing evaluation frameworks.
| Does the option: | Option 1 | Option 2 | Option 3 |
|---|---|---|---|
| Enhance Biosecurity | |||
| • By preventing incidents | 1 | 2 | 2 |
| • By helping respond | 1 | 3 | 3 |
| Foster Lab Safety | |||
| • By preventing incident | 2 | 2 | 1 |
| • By helping respond | 1 | 3 | 2 |
| Protect the environment | |||
| • By preventing incidents | 2 | 3 | 2 |
| • By helping respond | 1 | 3 | 2 |
| Other considerations | |||
| • Minimizing costs and burdens to stakeholders | 2 | 1 | 1 |
| • Feasibility? | 1 | 2 | 2 |
| • Not impede research | 2 | 1 | 1 |
| • Promote constructive applications | 1 | 2 | 1 |
Evaluation and Prioritization of Governance Approach
Overall, this integrated governance approach performs well across the major policy goals of biosecurity, lab safety, and responsible innovation. By focusing on prevention at the design stage and accountability at the production stage, it strengthens biosecurity while remaining feasible and compatible with existing biopharmaceutical workflows. Although the approach introduces some additional cost and procedural overhead, it does not fundamentally impede research and instead helps reduce downstream failures and regulatory risk.
Based on this evaluation, the integrated safety screening and secure sequence logging approach should be prioritized as the primary governance mechanism for AI-enabled protein drug discovery and bioproduction. This strategy addresses the highest-risk stages—design and scale-up—while remaining technically feasible and aligned with existing biopharmaceutical practices. The key trade-off involves balancing innovation speed with safety and accountability. While additional screening and logging may introduce modest overhead, these costs are outweighed by reduced downstream failures, increased regulatory confidence, and improved public trust.
This recommendation is directed toward biopharmaceutical R&D leadership and regulatory agencies, where early alignment between AI-driven discovery and governance expectations can ensure that emerging therapeutic technologies are both innovative and trustworthy.
This week, we were tasked to utilize different tools to be able to virtually read, write, and visualize using samples like lambda DNA from Escherichia coli and the Tumor suppressor gene from humans.
For this assignment, I have chosen the Tumor Repressor protein 53 in humans. I chose this because I have previously made a comparative analysis with the Trp 53 protein from the mouse.
AAH03596.1 Tumor protein p53 [Homo sapiens] MEEPQSDPSVEPPLSQETFSDLWKLLPENNVLSPLPSQAMDDLMLSPDDIEQWFTEDPGPDEAPRMPEAA PRVAPAPAAPTPAAPAPAPSWPLSSSVPSQKTYQGSYGFRLGFLHSGTAKSVTCTYSPALNKMFCQLAKT CPVQLWVDSTPPPGTRVRAMAIYKQSQHMTEVVRRCPHHERCSDSDGLAPPQHLIRVEGNLRVEYLDDRN TFRHSVVVPYEPPEVGSDCTTIHYNYMCNSSCMGGMNRRPILTIITLEDSSGNLLGRNSFEVRVCACAGR DRRTEEENLRKKGEPHHELPPGSTKRALPNNTSSSPQPKKKPLDGEYFTLQIRGRERFEMFRELNEALEL KDAQAGKEPGGSRAHSSHLKSKKGQSTSRHKKLMFKTEGPDSD
atggaagaaccgcagagcgatccgagcgtggaaccgccgctgagccaggaaacctttagc gatctgtggaaactgctgccggaaaacaacgtgctgagcccgctgccgagccaggcgatg gatgatctgatgctgagcccggatgatattgaacagtggtttaccgaagatccgggcccg gatgaagcgccgcgcatgccggaagcggcgccgcgcgtggcgccggcgccggcggcgccg accccggcggcgccggcgccggcgccgagctggccgctgagcagcagcgtgccgagccag aaaacctatcagggcagctatggctttcgcctgggctttctgcatagcggcaccgcgaaa agcgtgacctgcacctatagcccggcgctgaacaaaatgttttgccagctggcgaaaacc tgcccggtgcagctgtgggtggatagcaccccgccgccgggcacccgcgtgcgcgcgatg gcgatttataaacagagccagcatatgaccgaagtggtgcgccgctgcccgcatcatgaa cgctgcagcgatagcgatggcctggcgccgccgcagcatctgattcgcgtggaaggcaac ctgcgcgtggaatatctggatgatcgcaacacctttcgccatagcgtggtggtgccgtat gaaccgccggaagtgggcagcgattgcaccaccattcattataactatatgtgcaacagc agctgcatgggcggcatgaaccgccgcccgattctgaccattattaccctggaagatagc agcggcaacctgctgggccgcaacagctttgaagtgcgcgtgtgcgcgtgcgcgggccgc gatcgccgcaccgaagaagaaaacctgcgcaaaaaaggcgaaccgcatcatgaactgccg ccgggcagcaccaaacgcgcgctgccgaacaacaccagcagcagcccgcagccgaaaaaa aaaccgctggatggcgaatattttaccctgcagattcgcggccgcgaacgctttgaaatg tttcgcgaactgaacgaagcgctggaactgaaagatgcgcaggcgggcaaagaaccgggc ggcagccgcgcgcatagcagccatctgaaaagcaaaaaaggccagagcaccagccgccat aaaaaactgatgtttaaaaccgaaggcccggatagcgat
ATGGAAGAACCACAAAGTGACCCCAGCGTTGAACCGCCGCTGAGCCAGGAAACCTTCAGTGATCTGTGGAAACTGCTGCCGGAAAACAACGTGCTGAGCCCGCTGCCGAGCCAGGCGATGGATGATCTGATGCTGTCTCCGGATGACATTGAGCAGTGGTTCACCGAAGACCCCGGACCGGATGAAGCGCCGCGTATGCCGGAAGCAGCACCGCGCGTAGCACCGGCACCGGCAGCACCGACCCCGGCTGCACCTGCACCGGCACCCTCATGGCCGCTCAGCAGCTCAGTGCCCAGCCAGAAAACCTATCAGGGCAGCTATGGCTTCCGCCTGGGCTTCCTGCACAGCGGCACGGCAAAATCGGTGACCTGCACCTACAGCCCTGCGCTGAACAAGATGTTCTGCCAGCTGGCGAAAACCTGCCCGGTGCAGCTGTGGGTTGACTCCACACCGCCGCCAGGCACCCGTGTGCGTGCGATGGCGATCTATAAACAGAGCCAGCACATGACCGAAGTGGTGCGTCGCTGCCCGCACCATGAGCGCTGCTCTGACAGCGACGGTCTGGCACCGCCGCAGCATCTGATCCGCGTTGAAGGTAACCTGCGTGTGGAGTATCTGGATGACCGCAACACCTTCCGCCACAGCGTGGTGGTGCCGTATGAACCGCCGGAAGTGGGCAGCGACTGCACCACCATCCACTACAACTACATGTGCAACTCCTCCTGCATGGGCGGTATGAACCGCCGTCCGATTCTGACCATTATCACCCTGGAAGACTCCAGCGGTAACCTGCTGGGCCGTAACAGCTTTGAAGTGCGTGTGTGTGCCTGTGCCGGCCGCGATCGCCGCACGGAAGAAGAAAACCTGCGCAAGAAAGGTGAACCGCACCACGAACTGCCGCCGGGCAGCACCAAGCGTGCGCTGCCGAACAACACCTCCTCCAGCCCGCAGCCGAAGAAGAAACCGCTGGATGGCGAGTACTTCACCCTGCAGATCCGTGGGCGTGAACGTTTTGAAATGTTCCGTGAGCTGAACGAAGCGCTGGAGCTGAAAGATGCGCAGGCGGGTAAAGAGCCGGGTGGCTCACGTGCGCACAGCAGCCACCTGAAATCCAAAAAAGGTCAGAGCACCAGCCGTCACAAAAAACTGATGTTTAAAACTGAAGGGCCGGACAGCGAT
The main reason that the same gene can produce different proteins at the transcriptional level is mainly because of :
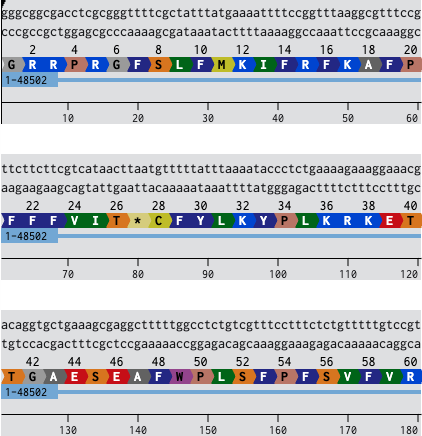

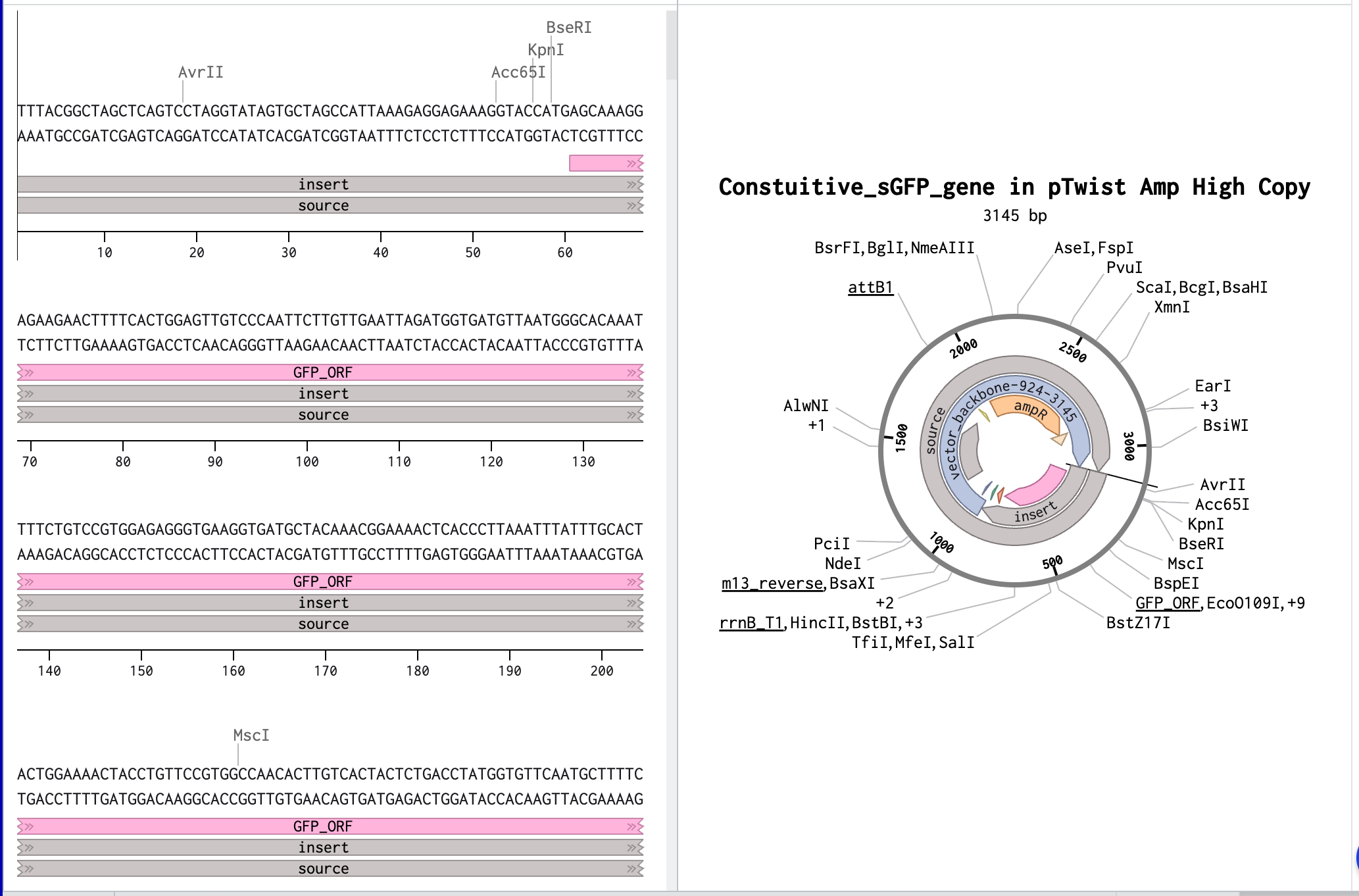
In this part, I was able to create an expression cassette that can be inserted into a vector plasmid and incorporated with a cell-free or a cell-dependent medium to express a desired protein. To exercise the entire procedure of making a construct and getting a customised plasmid vector benchling and Twist were used. I used the sGFP gene sequence from NCBI and annotated its promoter, ribosome-binding site, optimized codon region, and its terminator on benchling and later a pTwist Amp High Copy vector was used after downloading from Twist.

What are some components in the Phusion High-Fidelity PCR Master Mix, and what is their purpose?
What are some factors that determine primer annealing temperature during PCR?
There are two methods from this class that create linear fragments of DNA: PCR, and restriction enzyme digests. Compare and contrast these two methods, both in terms of protocol as well as when one may be preferable to use over the other.
How can you ensure that the DNA sequences that you have digested and PCR-ed will be appropriate for Gibson cloning?
How does the plasmid DNA enter the E. coli cells during transformation?
Describe another assembly method in detail (such as Golden Gate Assembly) Explain the other method in 5 - 7 sentences plus diagrams (either handmade or online).
(On average, an amino acid is ~100 Daltons)
1 Dalton ≈ 1 g/mol
Average amino acid ≈ 100 g/mol
If you eat 500 g of (pure) amino acids:
number of moles = Gm/ Tm = 500g/100g/mol
Using Avogadro’s number: 5×6.022×10^23 ≈ 3.0 × 10²⁴ molecules
So you consume roughly 3 septillion amino acid molecules.
Proteins are digested into individual amino acids in the stomach and small intestine.
Your body:
Because they have been created by an intelligent design in such a way.
Yes. Scientists create non-natural amino acids using synthetic biology.
Examples of designs:
• A fluorescent amino acid (attach a fluorophore to side chain) • A metal-binding amino acid (add a bipyridine group) • A photo-switchable amino acid (add an azobenzene group) • A redox-active amino acid
These can:
Natural proteins use L-amino acids and form right-handed α-helices.
If you use D-amino acids, you would expect a left-handed α-helix.
The handedness flips due to stereochemistry.
Yes.
Beyond the α-helix, proteins contain:
3₁₀ helix
π-helix
Collagen triple helix
Structural biology and protein design can reveal or engineer new helix types.


Because biological systems predominantly use L-amino acids.
Their stereochemistry naturally favors right-handed packing for minimal steric clash and optimal hydrogen bonding.
What is the driving force for β-sheet aggregation? Why do many amyloid diseases form β-sheets? Can you use amyloid β-sheets as materials? Design a β-sheet motif that forms a well-ordered structure.
Why β-sheets aggregate: β-strands expose backbone hydrogen bonding groups. They stack via intermolecular hydrogen bonds.
Driving force:
Hydrogen bonding
Hydrophobic interactions
π–π stacking (aromatic residues)
Amyloid diseases: Proteins misfold and form stable β-sheet fibrils.
Examples include:
Alzheimer’s disease
Parkinson’s disease
Amyloid β-peptides form cross-β sheet structures.
Materials applications: Yes — amyloid fibrils can be used as:
Nanowires
Hydrogels
Biocompatible scaffolds
Alternating hydrophobic residues promotes stacking and ordered β-sheet assembly.
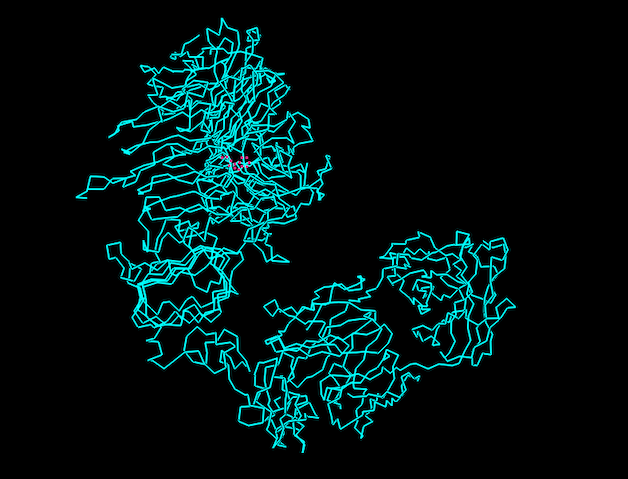
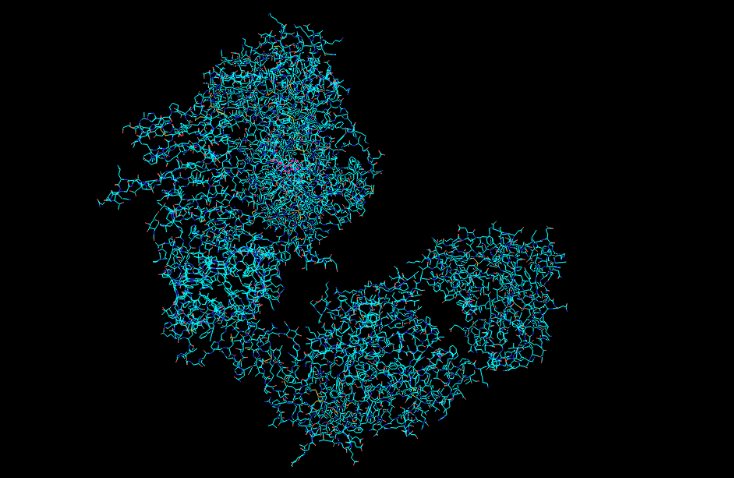
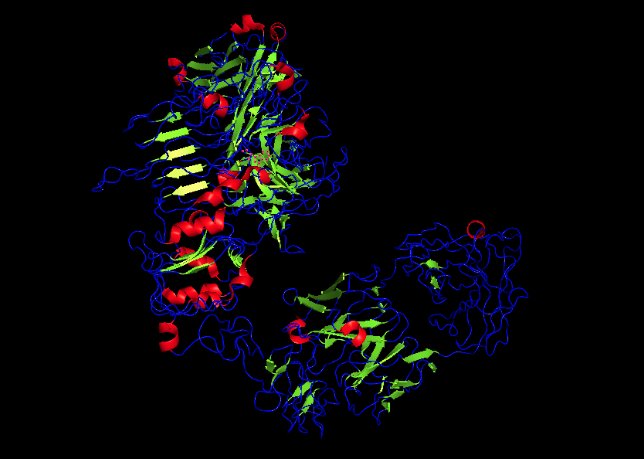
I have chosen Herceptin (trastuzumab) for this section. Herceptin is a monoclonal antibody mainly involved in recognising cancer cells. It binds specifically to the HER2 receptor on cancer cells and blocks signaling pathways that promote tumor growth. I selected this protein because it is an important example of a therapeutic antibody widely used in breast cancer treatment.
MELAALCRWGLLLALLPPGAASTQVCTGTDMKLRLPASPETHLDMLRHLYQGCQVVQGNLELTYLPTNASLSFLQDIQEVQGYVLIAHNQVRQVPLQRLRIVRGTQLFEDNYALAVLDNGDPLNNTTPVTGASPGGLRELQLRSLTEILKGGVLIQRNPQLCYQDTILWKDIFHKNNQLALTLIDTNRSRACHPCSPMCKGSRCWGESSEDCQSLTRTVCAGGCARCKGPLPTDCCHEQCAAGCTGPKHSDCLACLHFNHSGICELHCPALVTYNTDTFESMPNPEGRYTFGASCVTACPYNYLSTDVGSCTLVCPLHNQEVTAEDGTQRCEKCSKPCARVCYGLGMEHLREVRAVTSANIQEFAGCKKIFGSLAFLPESFDGDPASNTAPLQPEQLQVFETLEEITGYLYISAWPDSLPDLSVFQNLQVIRGRILHNGAYSLTLQGLGISWLGLRSLRELGSGLALIHHNTHLCFVHTVPWDQLFRNPHQALLHTANRPEDECVGEGLACHQLCARGHCWGPGPTQCVNCSQFLRGQECVEECRVLQGLPREYVNARHCLPCHPECQPQNGSVTCFGPEADQCVACAHYKDPPFCVARCPSGVKPDLSYMPIWKFPDEEGACQPCPINCTHSCVDLDDKGCPAEQRASPLTSIISAVVGILLVVVLGVVFGILIKRRQQKIRKYTMRRLLQETELVEPLTPSGAMPNQAQMRILKETELRKVKVLGSGAFGTVYKGIWIPDGENVKIPVAIKVLRENTSPKANKEILDEAYVMAGVGSPYVSRLLGICLTSTVQLVTQLMPYGCLLDHVRENRGRLGSQDLLNWCMQIAKGMSYLEDVRLVHRDLAARNVLVKSPNHVKITDFGLARLLDIDETEYHADGGKVPIKWMALESILRRRFTHQSDVWSYGVTVWELMTFGAKPYDGIPAREIPDLLEKGERLPQPPICTIDVYMIMVKCWMIDSECRPRFRELVSEFSRMARDPQRFVVIQNEDLGPASPLDSTFYRSLLEDDDMGDLVDAEEYLVPQQGFFCPDPAPGAGGMVHHRHRSSSTRSGGGDLTLGLEPSEEEAPRSPLAPSEGAGSDVFDGDLGMGAAKGLQSLPTHDPSPLQRYSEDPTVPLPSETDGYVAPLTCSPQPEYVNQPDVRPQPPSPREGPLPAARPAGATLERPKTLSPGKNGVVKDVFAFGGAVENPEYLTPQGGAAPQPHPPPAFSPAFDNLYYWDQDPPERGAPPSTFKGTPTAENPEYLGLDVPV
Total Length: 1255 Most Common Amino Acid: Leucine(L)
It belongs to the immunoglobulin G (IgG1) subclass within the immunoglobulin superfamily. And it is part of the L-domian family. (Immunoglobulin Light-chain domain.)
Resolution: 4.36 Å, which shows low resolution of the model.
The crystal structure of trastuzumab bound to HER2 was solved in 2004.






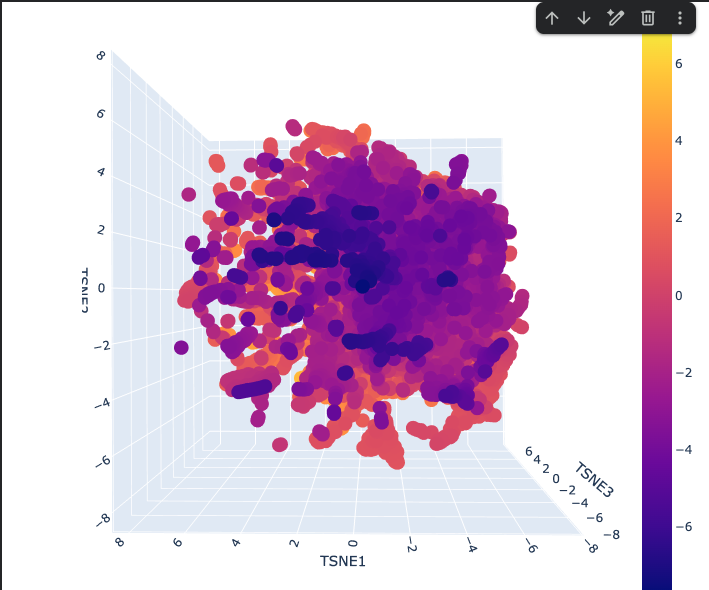

One large continuous cloud — no hard separate clusters Reflects that protein sequence space is smooth and gradual, not divided into distinct categories
Where most proteins sit These are common, well-represented protein families that ESM2 has seen many times
Outlier proteins that are unusual or specialized Score higher on whatever the colorbar is measuring (likely a biological property or cluster score ranging from -7 to +7)
Streaks radiating outward from the core Represent protein subfamilies that share a common origin but have diverged over evolution.
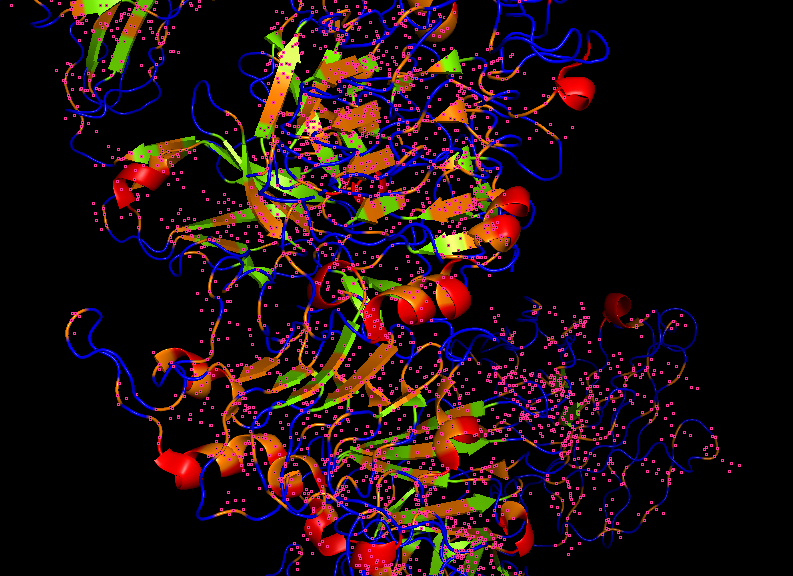
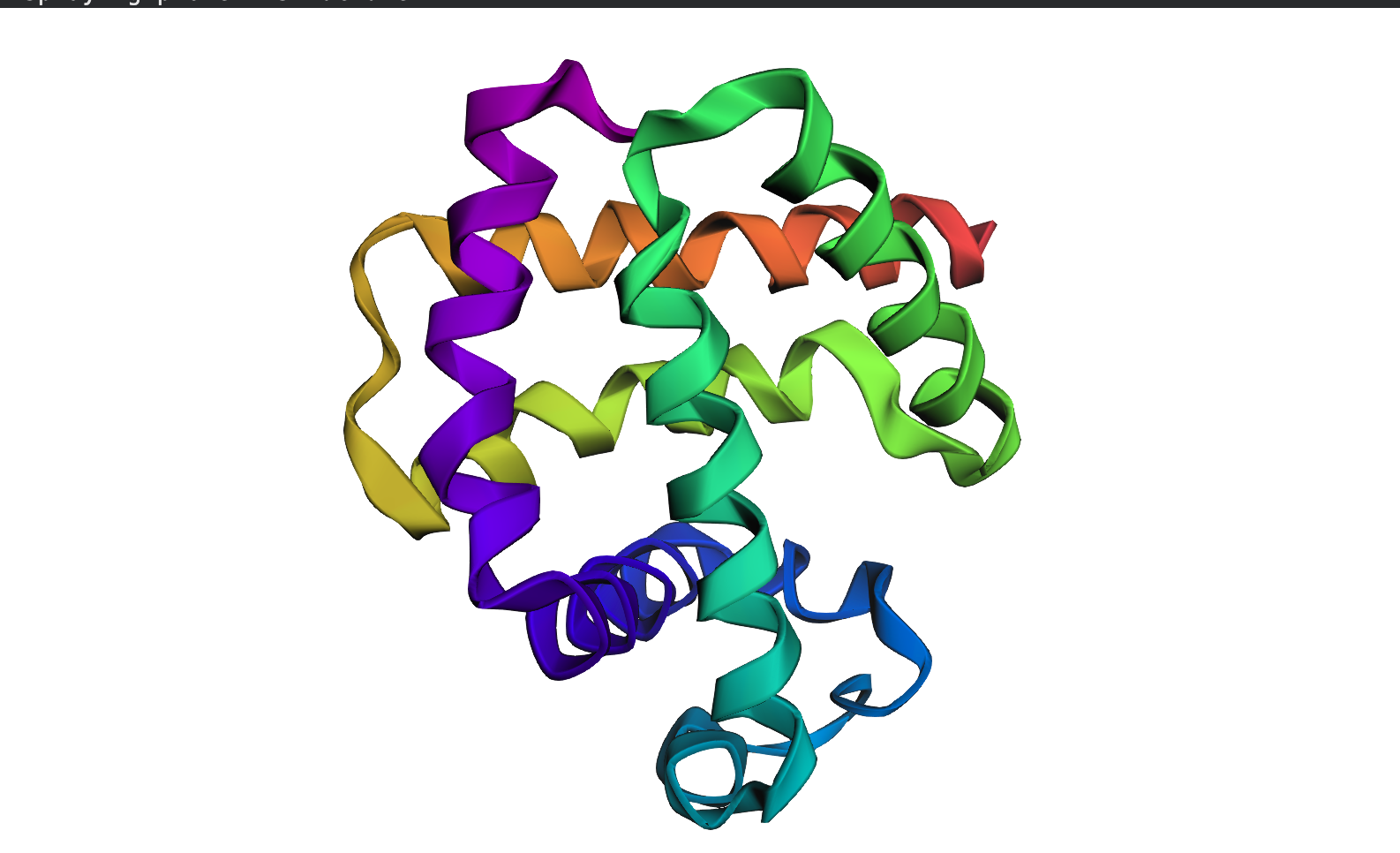
For this part, I used the PDB file of the HER2 protein. After uploading the pdb file, a reverse folding was run, and 20 possible candidates for the actual sequence of the protein was predicted. Among the results, the one with the lowest log score was identified through manual screeing and was folded using the ESMfold model. The predicted sequence and the folded protein are attached below.
ALTPEQAALLAAAWAPVFADREANARAFVLDLFRAYPSLADLFPEFKGKTLEQIAASPALGPYAGAFADRLAQFVASSDNAAKMATFWENYANEHIRRGITASHFEQVRAVFPGFVASVAEPPPGAAAAWDQFWGGIIDALKKAGG
T=0.5, sample=0, score=0.9440, seq_recovery=0.4932
T = 0.5 (Temperature)
Controls how creative/diverse the designed sequence is 0.5 is moderate — balanced between staying close to original and exploring new sequences Lower (0.1) = conservative, Higher (1.0) = very adventurous
sample = 0
This is the first designed sequence (counting starts from 0) If you generated 10 sequences, you’d see sample=0 through sample=9 Each sample is an independent design attempt for the same backbone
score = 0.9440
Negative log likelihood — measures model confidence Lower = better — model is very confident this sequence fits your backbone Your score of 0.9440 is excellent — it’s below 1.0 which is better than your insulin results (1.06 and 1.08)
seq_recovery = 0.4932
49.32% of positions match the original protein sequence exactly Roughly 1 in 2 residues is identical to the original This is your best recovery so far — slightly higher than insulin’s ~46%

What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions?
Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
Below is a diagram depicting an intracellular single-layer perceptron where the X1 input is DNA encoding for the Csy4 endoribonuclease and the X2 input is DNA encoding for a fluorescent protein output whose mRNA is regulated by Csy4. Tx: transcription; Tl: translation.
What are some examples of existing fungal materials and what are they used for? What are their advantages and disadvantages over traditional counterparts?
What might you want to genetically engineer fungi to do and why? What are the advantages of doing synthetic biology in fungi as opposed to bacteria?