Week 1: Principles and Practices
1. Describe a biological engineering application or tool you want to develop and why.
I was reading this review paper, “Engineering bacterial warriors: harnessing microbes to modulate animal physiology”[1]. There’s a section that talks about engineering bacteria that can help corals survive heat stress, like making bacteria that clean up harmful reactive oxygen species (ROS) when water gets too warm [Figure 1]. The paper also discusses transplanting entire communities of beneficial microbes from resilient corals to vulnerable ones (Coral Microbiome Transfer, or CMT) [1,2]. The whole concept of using Beneficial Microorganisms for Corals (BMC) is really promising [3].
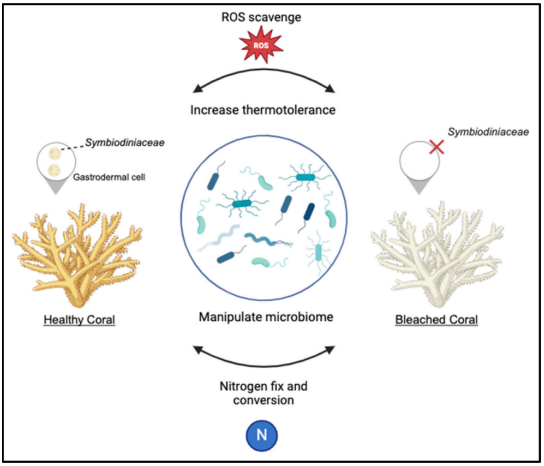
 Figure 1. Engineered microbes have the capacity to assist corals in alleviating environmental stresses [1].
Figure 1. Engineered microbes have the capacity to assist corals in alleviating environmental stresses [1].
However, the authors point out major deployment challenges [1, 4]. The effects of introducing new bacteria are unpredictable, and they could outcompete or disrupt the coral’s natural microbiome. Currents could also scatter free bacteria before they even reach the target corals. At the moment, there’s no way to control the dosage or how long the coral is exposed, making the treatment inefficient and potentially wasteful. In essence, it’s difficult to introduce these engineered microbes effectively in the ocean.
The next section of the paper was about human health, discussing how engineered bacteria are being developed for targeted disease therapy [1]. This got me thinking about medical delivery systems, things like timed-release pills, biodegradable implants, or hydrogels that release drugs right where they’re needed in the body. The principle for all these applications is the same, to protect the therapeutic agent and control its release at the target site. So I thought something similar could be done for coral reefs.
I looked into existing coral probiotic delivery systems and came across a smart underwater microbial delivery system for coral reef habitat recovery developed by researchers at KAUST [5]. This system uses a buoy, an FPGA computer, cameras, and AI to monitor coral color and automatically pump probiotics into the water [Figure 2]. However, its complexity, cost, and reliance on pumping microbes into the water column still face some of the core challenges mentioned in the review paper, like dosage control and localization.

 Figure 2. Smart underwater microbial delivery system for coral reef habitat (KAUST) [5].
Figure 2. Smart underwater microbial delivery system for coral reef habitat (KAUST) [5].
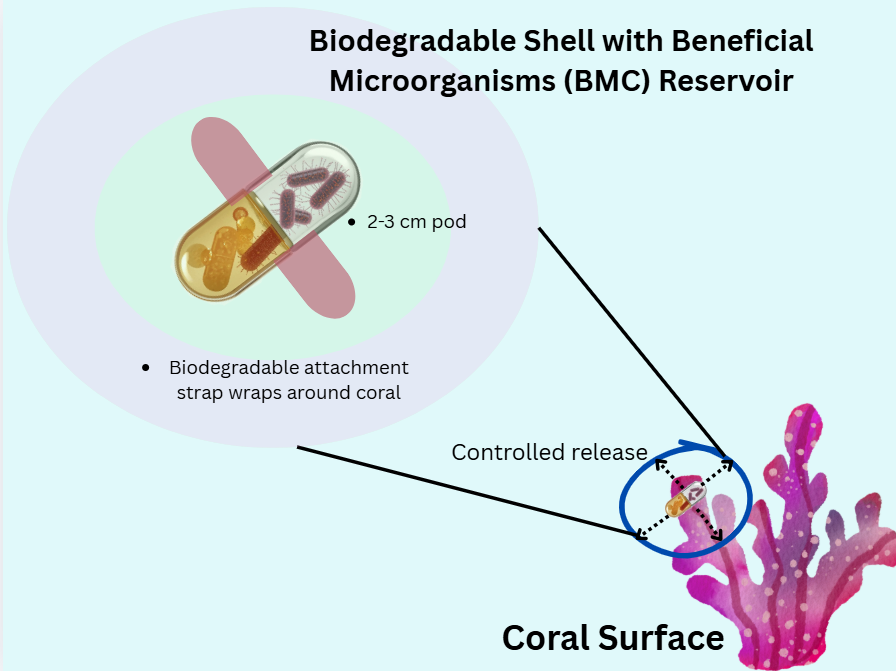
To address the limitations of bulk water delivery, I propose a more targeted approach: a small, biodegradable pod, like a micro-infusion pump specifically for a coral. The pod would be able to attach directly to the coral’s surface to create a localized environment where the release of beneficial bacteria can be controlled. This would allow for a slow and sustained colonization of the coral’s microbiome, making the delivery more efficient and less wasteful than other methods. Diver or robotic systems would be able to precisely deploy the pods which allows for targeted intervention on corals identified as the most at-risk or ecologically valuable for reef recovery.
This idea of targeted, contained delivery isn’t completely new. In agriculture, technologies like coated seeds or biodegradable granules are standard for protecting and delivering beneficial microbes to plant roots [6]. For corals, scientists are already exploring direct methods like “Bacterioplankton” or gels [4]. A small, attachable pod builds on these principles and turns a massive ecological problem into a tangible engineering challenge to deliver a single, effective device.
Sketch of the idea:

References
Gao B, Ruiz D, Case H, Jinkerson RE, Sun Q. Engineering bacterial warriors: harnessing microbes to modulate animal physiology. Curr Opin Biotechnol. 2024;87:103113. doi:10.1016/j.copbio.2024.103113.
Doering T, Wall M, Putchim L, Rattanawongwan T, Schroeder R, Hentschel U, et al. Towards enhancing coral heat tolerance: a “microbiome transplantation” treatment using inoculations of homogenized coral tissues. Microbiome. 2021;9(1):102. doi:10.1186/s40168-021-01053-6.
Peixoto RS, Rosado PM, Leite DCA, Rosado AS, Bourne DG. Beneficial microorganisms for corals (BMC): proposed mechanisms for coral health and resilience. Front Microbiol. 2017;8:341. doi:10.3389/fmicb.2017.00341.
Damjanovic K, van Oppen MJH, Menéndez P, Blackall LL. The contribution of microbial biotechnology to mitigating coral reef degradation. Microb Biotechnol. 2017 Jul 11;10(5):1236–1243. doi: 10.1111/1751-7915.12769.
Filho J. A smart underwater microbial delivery system for coral reef habitat recovery. InnovateFPGA. 2022 (cited 2026 February 8). Available from: https://www.innovatefpga.com/cgi-bin/innovate/teams.pl?Id=EM043.
Bashan Y, de-Bashan LE, Prabhu SR, Hernandez JP. Advances in plant growth-promoting bacterial inoculant technology: formulations and practical perspectives (1998-2013). Plant Soil. 2014;378(1):1-33. doi:10.1007/s11104-013-1956-x.
2. Describe one or more governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future, like ensuring non-malfeasance (preventing harm). Break big goals down into two or more specific sub-goals.
To ensure the ethical development and deployment of the coral probiotic pod, I propose adapting the governance framework outlined in the Synthetic Genomics: Options for Governance report [7]. The framework focuses on Safety, Security, Responsibility, and Oversight which together provide a clear structure to address the environmental and social challenges of engineering interventions in sensitive ocean ecosystems.
Goal 1: Safety – Establishing a Risk Assessment Protocol
Preventing unintended harm to reef ecosystems requires a phased approach that mirrors established biocontainment principles. This begins with pre-deployment screening in which any engineered microbial strain undergoes standardized laboratory assessment to confirm:
- Non-pathogenic to a panel of key reef organisms (corals, fish, crustaceans)
- Non-toxic in terms of metabolites produced
- Assessed for its competitive impact on the native coral microbiome
Following lab validation, a mandatory phased testing pathway should be implemented. Initial trials would occur in a controlled aquarium system simulating reef conditions. Successful results could then progress to caged in-situ trials, where pods are deployed within permeable enclosures on actual reefs to monitor ecological interactions under natural but contained conditions.
Goal 2: Security – Implementing a Function-Limited Use Framework
To address dual-use concerns inherent to programmable delivery systems, governance should focus on restricting applications to predefined beneficial functions. One approach involves establishing a “Positive List” of approved microbial functions, such as ROS-scavenging or nutrient provision, for coral restoration. An organization like the European Marine Research Network (EuroMarine) could maintain this public, evidence-based registry. Delivery of any agent performing functions not on this list would be prohibited. Complementing this, procedural security measures would include certification training for users in secure handling and deployment protocols, and restricting access to advanced engineered strains to institutions operating under established biosafety and biosecurity frameworks.
Goal 3: Responsibility – Ensuring Equitable Access and Procedural Justice
To align development with principles of environmental justice and accessibility, the pod’s mechanical design should follow an open-source model. 3D printable files and assembly guides should be published under a non-commercial conservation license to democratize access and enable local adaptation. Additionally, formal stakeholder consultation processes should be integrated into deployment planning. Engaging coastal communities, fishery cooperatives, and tourism boards from the start of the project would ensure local ecological knowledge informs implementation and aligns with community-defined priorities.
Goal 4: Oversight – Creating an Adaptive Governance Structure
Effective implementation requires mechanisms for ongoing evaluation and adaptation. A multidisciplinary advisory panel should comprise marine ecologists, synthetic biologists, social scientists, ethicists, and community representatives. They would provide oversight by reviewing field trial proposals and periodically updating the “Positive List” and testing protocols based on emerging research. This could be supplemented by a centralized deployment registry documenting key metadata (location, probiotic function, scale, responsible entity) to enable accountability and long-term impact monitoring.
References
- Garfinkel MS, Endy D, Epstein GL, Friedman RM. Synthetic Genomics: Options for Governance. J. Craig Venter Institute; 2007. Available from: https://www.jcvi.org/research/synthetic-genomics-options-governance.
3. Describe at least three different potential governance “actions” by considering the four aspects below (Purpose, Design, Assumptions, Risks of Failure & “Success”)
Action 1: Establishing a Mandatory ‘Reef-Safe’ Biopolymer Certification
- Purpose: Current material standards, such as the EU’s compostability criteria or general marine biodegradation tests, are not designed for devices in direct, sustained contact with sensitive coral reef ecosystems [8]. A new certification is needed to prevent pollution or toxic leaching from pod materials.
- Design: The European Committee for Standardization (CEN) could develop this certification in consultation with groups like the International Coral Reef Initiative (ICRI). It would build on existing marine biodegradation tests (like ISO 18830) but add specific ecotoxicity assays [9,10]. In the UK, Defra could require this certification for Marine Management Organisation (MMO) deployment permits [11].
- Assumptions: This assumes lab tests accurately predict real-world impacts and that certification won’t be too burdensome for conservation projects.
- Risks of Failure & “Success”: The action could fail if perceived as bureaucratic overreach, leading to low adoption or non-compliance. A significant risk of success is ‘greenwashing,’ where achieving material certification creates a false sense of security and diverts attention from necessary ecological monitoring of the probiotic function itself.
Action 2: Creating an Open-Source Hardware Repository Hosted by a Research Consortium
- Purpose: Open-source approaches have worked well in conservation technology. Applying this model to the pod system would prevent proprietary lock-in and encourage adaptation, aligning with funder open-access policies [12,13].
- Design: A consortium like EuroMarine could host the repository. Designs would use licenses like CERN Open Hardware, with governance similar to successful citizen science platforms, allowing vetted contributions while maintaining quality control.
- Assumptions: Without an active community, the repository could stagnate. Success might paradoxically lead to commercial co-option, where companies create proprietary versions that limit equitable access.
- Risks of Failure & “Success”: Failure could stem from repository stagnation without dedicated curation. Additionally, success could lead a for-profit entity to create a proprietary, locked version of the core open design, potentially undermining the goal of equitable access.
Action 3: Implementing Mandatory Pre-Deployment Registration in Existing Public Registers
- Purpose: While transparency is required by EU and UK marine laws, no registry exists for coral interventions. This action would fill that gap by extending existing systems.
- Design: In the UK, the MMO could add a coral intervention module to its Marine Case Management System [11]. For international waters, the UN’s Biosafety Clearing-House (BCH) already tracks living modified organisms and could serve as a model [14]. Funders could require registration as a grant condition.
- Assumptions: This assumes existing systems can be adapted affordably and that transparency itself encourages responsible behavior.
- Risks of Failure & “Success”: Cumbersome processes might reduce compliance. If successful, registry clutter could obscure important projects, and public listings might attract opposition to legitimate research.
References
- Wei L, McDonald AG, Stark NM. Biodegradable polymers in the marine environment: current status and future perspectives. Environ Sci Technol. 2021;55(9):4203-17.
- International Organization for Standardization. Plastics — determination of aerobic biodegradation of non-floating plastic materials in a seawater/sandy sediment interface — method by measuring the oxygen demand in closed respirometer. ISO 18830:2018.
- European Committee for Standardization. Who we are (Internet). Brussels: CEN; 2023 (cited 2026 Feb 8). Available from: https://www.cen.eu.
- Department for Environment, Food & Rural Affairs. Marine and Coastal Access Act 2009: guidance (Internet). (cited 2026 Feb 8). Available from: https://www.legislation.gov.uk/ukpga/2009/23/contents.
- European Commission. Horizon Europe programme guide (Internet). Brussels: European Commission; 2021 (cited 2026 Feb 8). Available from: https://ec.europa.eu/info/funding-tenders/opportunities/docs/2021-2027/horizon/guidance/programme-guide_horizon_en.pdf.
- UK Research and Innovation. UKRI open access policy (Internet). Swindon: UKRI; 2022 (cited 2026 Feb 8). Available from: https://www.ukri.org/publications/ukri-open-access-policy/.
- Secretariat of the Convention on Biological Diversity. The Biosafety Clearing-House (BCH) (Internet). Montreal: SCBD; 2023 (cited 2026 Feb 8). Available from: https://bch.cbd.int/.
4. Score (from 1-3 with, 1 as the best, or n/a) each of your governance actions against your rubric of policy goals. The following is one framework but feel free to make your own:
| Does the option: | Option 1 | Option 2 | Option 3 |
|---|---|---|---|
| Enhance Biosecurity | |||
| • By preventing incidents | 1 | 3 | 2 |
| • By helping respond | 2 | 1 | 1 |
| Foster Lab Safety | |||
| • By preventing incident | 2 | n/a | n/a |
| • By helping respond | 2 | n/a | n/a |
| Protect the environment | |||
| • By preventing incidents | 1 | 2 | 2 |
| • By helping respond | 3 | 3 | 1 |
| Other considerations | |||
| • Minimizing costs and burdens to stakeholders | 3 | 1 | 1 |
| • Feasibility? | 2 | 1 | 1 |
| • Not impede research | 2 | 1 | 1 |
| • Promote constructive applications | 1 | 1 | 1 |
5. Drawing upon this scoring, describe which governance option, or combination of options, you would prioritize, and why. Outline any trade-offs you considered as well as assumptions and uncertainties.
Based on the scoring, I would prioritize implementing Option 2 and Option 3 combined. Looking at the table, Option 2 and Option 3 both score a “1” on all of the “Other considerations” section. That means that they minimize burden, are highly feasible, don’t impede research, and promote good applications. This is important for a new tool meant to address an urgent problem like coral bleaching. If governance is too heavy-handed from the start, it could hinder the innovation and collaboration that’s needed. The trade-off is that Option 2, by itself, scores a “3” on preventing biosecurity incidents. Making designs freely available could, in theory, make misuse easier. This is where Option 3 creates counterbalance because it scores a “1” on helping respond to incidents across biosecurity and environmental protection. By pairing them, it would create a system with the tools to build the coral probiotic pod and help reefs, and it would create transparency and accountability by asking users to outline where, when, and what they’re doing with the tools.
Although Option 1 is undeniably important, it scores low on burden, feasibility and potential to impede research. Strict certification could potentially stall projects, which is why it’s important to begin by requiring the use of characterized and safe materials documented in the open-source repository (Option 2) and logged in the registry (Option 3). As the field matures and the most effective materials become clear, a formal certification like Option 1 can be developed based on real-world data, making it more practical and accepted.
This decision comes with the assumption that the primary users, such as researchers, and conservation NGOs, would be acting in good faith. The governance is primarily designed to support and guide a responsible community, not solely to thwart a malicious one. There’s also uncertainty regarding compliance with the registration system of Option 3. The system would only work if people actually use it, which depends heavily on enforcement tactics like making it a requirement for grant funding from UKRI or Horizon Europe, for example [12,13]. The main trade-off is accepting a potentially higher risk of a small-scale indicent by lowering barriers to entry with Option 2 in exchange for greater capacity for widespread, adaptive learning and rapid scaling. For a crisis like reef degradation, where rapid experimentation is needed, this seems like the necessary and ethical choice.
References
- European Commission. Horizon Europe programme guide (Internet). Brussels: European Commission; 2021 (cited 2026 Feb 8). Available from: https://ec.europa.eu/info/funding-tenders/opportunities/docs/2021-2027/horizon/guidance/programme-guide_horizon_en.pdf.
- UK Research and Innovation. UKRI open access policy (Internet). Swindon: UKRI; 2022 (cited 2026 Feb 8). Available from: https://www.ukri.org/publications/ukri-open-access-policy/.
Homework Questions from Professor Jacobson
- Nature’s machinery for copying DNA is called polymerase. What is the error rate of polymerase? How does this compare to the length of the human genome. How does biology deal with that discrepancy?
The error rate of DNA polymerase with its intrinsic proofreading function is approximately 1 error per 10⁶ bases incorporated (1:10⁶ or 10⁻⁶). the human genome has approximately 3.2 billion base pairs (3.2 x 10⁹ bp), that means there are about 3,200 errors per genome duplication. To deal with the discrepancy, there’s a third layer of correction called DNA Mismatch Repair (MMR). This is a post-replication system that acts after the polymerase has moved on. Specialized proteins scan newly synthesized DNA, identify mismatches that escaped proofreading, excise the erroneous segment, and resynthesize it correctly. In this way, MMR improves fidelity by an additional 100 to 1000-fold, so that the new error rate is about 1 error per 10⁹ to 10¹⁰ bases. Compared to the human genome, that’s about 0.3 errors per genome duplication.
- How many different ways are there to code (DNA nucleotide code) for an average human protein? In practice what are some of the reasons that all of these different codes don’t work to code for the protein of interest?
An average human protein has around 400 amino acids, and each of the 20 amino acids is encoded by 1-6 codons. Using average codon degeneracy (3 codons/amino acid), the possible sequences is around 3400, or roughly 10190 different DNA sequences. In practice all of these codes don’t work to code for the protein for several reasons. First, cells prefer certain codons for efficient translation because rare codons slow down protein production. Also, some sequences form stable structures that hinder ribosome binding or translation initiation, and these sequences could also create unintended splice sites that disrupt mRNA processing. Additionally, sequences like repeats or high GC content can cause recombination or mRNA degradation.
Homework Questions from Dr. LeProust
- What’s the most commonly used method for oligo synthesis currently?
The most commonly used method for oligo synthesis currently is the phophoramidite method developed by Caruthers in 1981.
- Why is it difficult to make oligos longer than 200nt via direct synthesis?
It’s difficult to make oligos longer than 200nt via direct synthesis because of the cumulative stepwise yield. Since the synthesis occurs in a cycle, so it compounds with each added based becoming inefficient and costly. After 200 cycles, even at 99.5% efficiency, only about 37% of the growing chains are the correct full-length product. The other shorter chains would be failed sequences that have to be removed.
- Why can’t you make a 2000bp gene via direct oligo synthesis?
The yield for a 2000nt strand would be very low which is impractical. Due to the high intrinsic error rate of chemial synthesis, in a 2000bp molecule there would be around 20 random errors which would make the gene non-functional.
Homework Question from George Church
[Using Google & Prof. Church’s slide #4] What are the 10 essential amino acids in all animals and how does this affect your view of the “Lysine Contingency”?
The 10 essentiall amino acids in all animals are: Cysteine (Cys), Histidine (His), Isoleucine (Ile), Leucine (Leu), Lysine (Lys), Methionine (Met), Phenylalanine (Phe), Threonine (Thr), Tryptophan (Trp), and Valine (Val) [1].
The “Lysine Contingency” is a fictional genetic modification from Jurassic Park that was designed to make engineered dinosaurs dependent on lysine supplements, causing them to die without them [2]. However, knowing the full list of essential amino acids significantly weakens the possibility of the contingency since lysine is not uniquely essential. It’s one of ten amino acids that animals cannot synthesize de novo. A predator deficient in any of these would most like have severe growth impairment, immune dysfunction, and eventually death. Additionally, animals get these essential amino acis from their diet. That means the contingency relies on the assumption that lysine is not available in the natural environment. However, lysine, like all the other essential amino acis, is abundant in protein-rich foods which a carnivorous dinosaur would depend on. Even herbivorous dinosaurs would acquire these amino acis through a varied plant diet. Therefore, the dinosaurs would probably meet their lysine requirement through their normal feeding behavior, which would make the contingency ineffective.
References
- Hou Y, Wu G. Nutritionally Essential Amino Acids. Advances in Nutrition (Internet). 2018 Sep 15;9(6):849–51. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6247364/.
- Lysine contingency (Internet). Jurassic Park Wiki. Available from: https://jurassicpark.fandom.com/wiki/Lysine_contingency.