Week 1 HW: Principles and Practices


Class Assignment
A Wearable or At-Home Androgen Biosensing Platform for PCOS Diagnosis and Management
I am interested in developing an easy-to-use biosensor/microfluidic kit that can detect and quantify androgen levels from blood or sweat to support the diagnosis and personalized management of Polycystic Ovary Syndrome (PCOS).
PCOS is a common hormonal disorder affecting 5-13% of reproductive-aged women and is characterized by androgen excess, irregular or absent ovulation, and polycystic ovaries. Symptoms can include severe acne, hirsutism, hair loss, and infertility, and the condition is associated with long-term risks such as diabetes, cardiovascular disease, and endometrial cancer.
Currently, one of the key diagnostic criteria for PCOS is elevated androgen levels, which are typically assessed through blood tests performed specifically on days 2-5 of the menstrual cycle. However, for many women with PCOS, menstrual cycles are irregular or absent, making it difficult to determine when testing should occur. As a result, diagnosis can be delayed or inconclusive.
A biosensing platform capable of continuously or repeatedly monitoring androgen levels throughout an entire cycle would provide a more accurate picture of hormonal dynamics. This could improve diagnostic reliability and enable more personalized treatment strategies by tracking hormonal responses to lifestyle changes or medications over time.
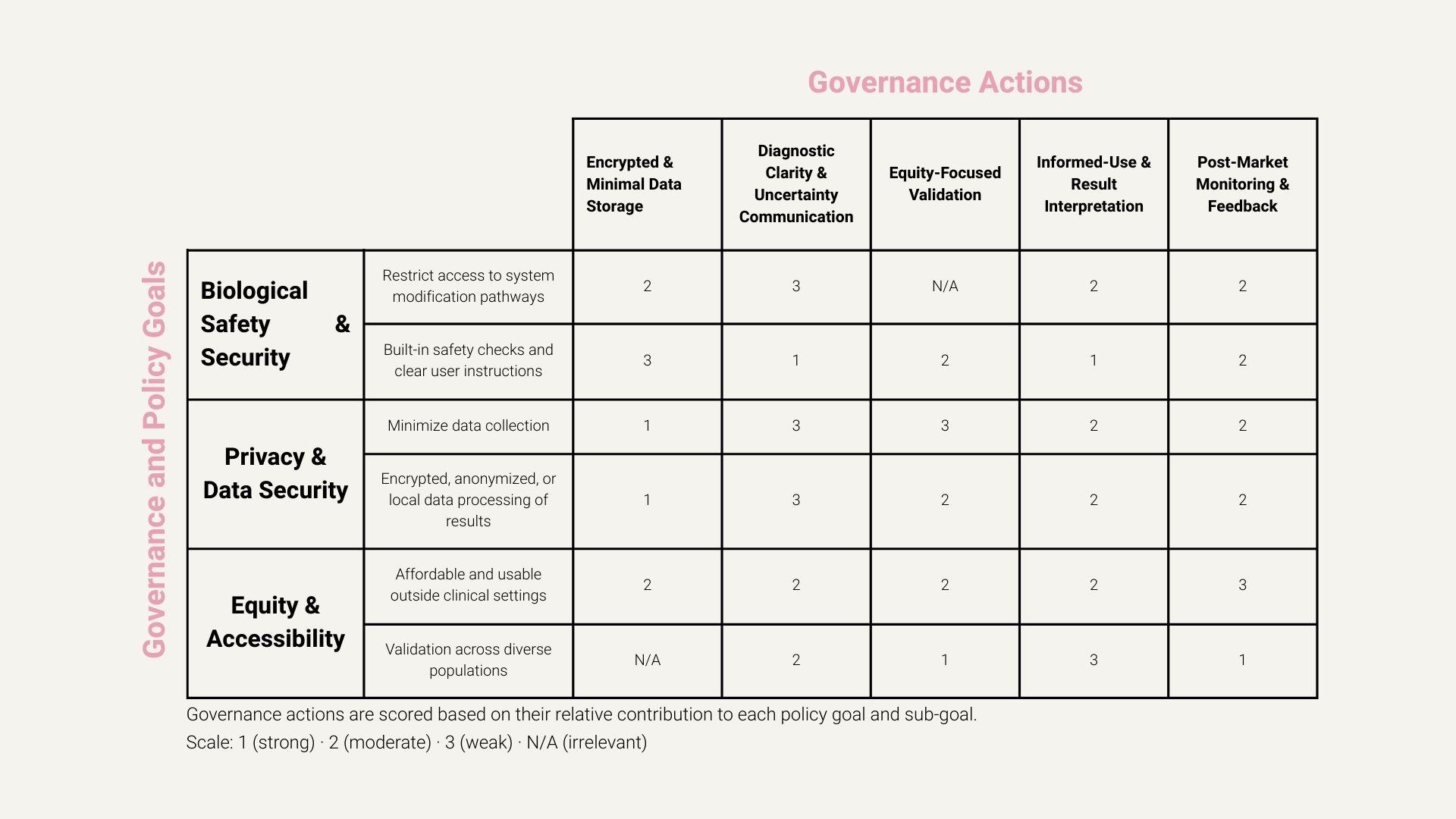
Governance and Policy Goals
1. Ensure Biological Safety and Security
Prevent misuse, unsafe operation, or unintended biological harm associated with the biosensing platform.
Sub-goals:
- Restrict access to raw sensor calibration, modification protocols, or firmware to reduce the risk of misuse.
- Incorporate built-in safety checks, clear user instructions, and warnings to minimize incorrect use or misinterpretation of results.
2. Protect User Privacy and Data Security
Protect sensitive hormonal and reproductive health data to prevent discrimination, stigma, or exploitation.
Sub-goals:
- Minimize data collection to only what is strictly necessary for hormonal monitoring and trend analysis.
- Ensure hormonal data is encrypted, anonymized, or processed locally when possible to reduce exposure.
3. Promote Equity and Accessibility in Women’s Health
Ensure the tool reduces existing barriers to diagnosis and care rather than reinforcing healthcare inequities.
Sub-goals:
- Design the biosensing kit to be affordable and usable outside specialized clinical settings.
- Validate the platform across diverse populations to reduce bias and improve diagnostic reliability for underrepresented groups.
Governance Actions
1. Encrypted and Minimal Data Storage System
Actor: Companies (product developers), with regulatory oversight
Ensure that hormonal data is encrypted and that only the minimum necessary data is collected or stored, preferably processed locally on-device.
2. Diagnostic Clarity and Uncertainty Communication
Actor: Companies + regulators
Ensure that diagnostic outputs clearly distinguish between high-confidence diagnoses and ambiguous or borderline results. The tool should explicitly communicate diagnostic confidence and uncertainty, and automatically prompt users to seek further clinical evaluation when results fall outside validated confidence thresholds.
3. Equity-Focused Validation Across Diverse Populations
Actor: Academic researchers, funders, public health agencies
Conduct and incentivize validation studies across diverse populations to reduce bias and improve diagnostic reliability for underrepresented groups.
4. Informed-Use and Result Interpretation Requirements
Actor: Companies + regulators
Require a short, standardized informed-use process before first use that explains the scope of the tool, the meaning of diagnostic confidence and uncertainty, and appropriate user actions in response to different types of results.
5. Post-Market Monitoring and Feedback Loop
Actor: Regulators + companies
Require ongoing, anonymized post-market monitoring of the tool after deployment, including reporting of false positives, false negatives, and aggregate user outcomes. This feedback should be used to identify unintended harms, performance gaps across populations, and opportunities for improvement over time.
Actions Ranking

Recommendation for Best Governance Action
Based on the scoring analysis, I would prioritize a combination of governance actions, with Encrypted and Minimal Data Storage as the primary action, alongside Informed-Use and Result Interpretation Requirements and Post-Market Monitoring and Feedback as complementary measures.
The highest priority should be the implementation of an encrypted and minimal data storage system. This action consistently scored strongest across privacy and data security sub-goals and provides a foundational safeguard against harm. Because hormonal and reproductive health data is highly sensitive, failures in data protection could lead to discrimination, stigma, or misuse at scale. Prioritizing data minimization and local or encrypted processing reduces these risks regardless of downstream user behavior or diagnostic accuracy. This action is also relatively feasible for companies to implement early and does not significantly postpone research or innovation.
As a second priority, I would emphasize informed-use and result interpretation requirements together with post-market monitoring. Informed-use mechanisms reduce misinterpretation and panic by helping users understand results and lower uncertainty, while post-market monitoring enables the detection of errors, biases, or unintended harms that emerge after deployment.
One key trade-off is the tension between data minimization and the need for a lot of data to support monitoring. There is also uncertainty around how effectively users will engage with and understand informed-use materials, which could limit their intended protective impact.
This recommendation is directed toward the research and development team responsible for building the biosensing platform, including academic labs and early-stage startups developing the technology. Overall, the scoring highlights that no single action is sufficient on its own. Instead, a comprehensive approach that prioritizes privacy infrastructure while supporting safe use and continuous evaluation offers the most ethically robust path forward.
Reflection on Ethical Concerns
An ethical concern that became especially salient to me this week is the risk associated with centralized access to sensitive hormonal and reproductive health data. Considering what could happen if a single actor gained access to aggregated user data highlighted how data breaches or misuse could cause large-scale harm, including discrimination or loss of trust. This concern reinforced the importance of prioritizing encrypted and minimal data storage, local data processing when possible, and ongoing post-market monitoring, rather than treating privacy as a secondary or purely technical consideration.
Lab Preparation
🦠 Complete Lab Specific Training in Person DONE
🦠 Complete Safety Training in Atlas DONE
Week 2 Lecture Prep
Questions from Professor Jacobson:
- Nature’s machinery for copying DNA is called polymerase. What is the error rate of polymerase? How does this compare to the length of the human genome. How does biology deal with that discrepancy?
Answer: The error rate of polymerase is approximately 1 in 10⁶. The human genome is about 3 × 10⁹ bp, so that corresponds to roughly 3,000 potential errors per genome copy (and across many dividing cells, the total number of errors could be much higher). Biology deals with this discrepancy through multiple layers of error correction - using proofreading and repair mechanisms that reduce errors, such as the MutS repair system.
- How many different ways are there to code (DNA nucleotide code) for an average human protein? In practice what are some of the reasons that all of these different codes don’t work to code for the protein of interest?
Answer: Most amino acids can be encoded by multiple codons, so there are an astronomically large number of possible DNA sequences that could encode the same protein, with many choices per amino acid compounding across the full length of the gene.
In practice, not all of these sequences work because certain codons are translated more efficiently than others, and some sequences can create unfavorable secondary structures or other issues. As a result, only a small subset of possible DNA sequences is functional and practical for expressing the desired protein.
Questions from Dr. LeProust:
- What’s the most commonly used method for oligo synthesis currently?
Answer: The most commonly used method for oligo synthesis is chemical phosphoramidite DNA synthesis, which builds DNA sequences one nucleotide at a time through repeated cycles of coupling, protection, and deprotection on a solid support.
- Why is it difficult to make oligos longer than 200nt via direct synthesis?
Answer: It is difficult to synthesize oligos longer than 200nt via direct chemical synthesis because each nucleotide addition (chemical reaction) has an error rate, and these errors accumulate with every synthesis cycle, causing the correctness of the sequences to decrease as the length increases.
- Why can’t you make a 2000bp gene via direct oligo synthesis?
Answer: A 2000bp gene cannot be made via direct oligo synthesis because synthesis errors accumulate with each nucleotide addition. As sequence length increases, the probability of producing a correct full-length sequence drops dramatically. Since direct oligo synthesis is already unreliable beyond 200nt, synthesizing a 2000bp sequence in a single run is effectively impossible. In addition, oligo synthesis produces single-stranded DNA, and generating a correct double-stranded gene of this length would further compound errors, making direct synthesis impractical.
Question from George Church:
- Using Google & Prof. Church’s slide #4, what are the 10 essential amino acids in all animals, and how does this affect your view of the “Lysine Contingency”?
Answer: The 10 essential amino acids in animals are: Histidine, Isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Threonine, Tryptophan, Valine, and Arginine.
I did not watch Jurassic Park, but according to what I read online, the “Lysine Contingency” was a genetic alteration Henry Wu performed in the dinosaurs’ genome to knock out their ability to produce lysine, forcing them to rely on lysine supplements provided by park staff in an attempt to imprison them in the park and prevent them from destroying the global ecosystem.
After checking where lysine comes from (I asked Gemini: “How do animals produce lysine?”), I found that animals cannot produce lysine at all and must obtain it through nutrition. This means that humans, animals, and dinosaurs would all need dietary lysine regardless. Based on this, I conclude that Henry Wu’s lysine contingency was useless as a containment strategy, and it is unclear what genetic mechanism he could have targeted to achieve this goal, since there is no direct lysine biosynthesis pathway in animals to knock out.
Resource: Lysine contingency, Jurassic Park Wiki
My HTGAA Website
What I did to personalise the website