Week 4 HW: Protein design part I
Part A. Conceptual Questions
- How many amino acid molecules are in 500 g of meat?
Ans: Average amino acid ≈ 100 Daltons (100 g/mol)
500 g ÷ 100 g/mol = 5 moles
1 mole = 6.022 × 10²³ molecules
So,
5 × 6.022 × 10²³ ≈ 3 × 10²⁴ amino acid molecules
- Why do humans eat beef but do not become a cow, eat fish but do not become fish?
Ans: When we eat beef or fish, our body breaks proteins into amino acids during digestion. Then we rebuild them into human proteins, not cow proteins.
- Why are there only 20 natural amino acids?
Ans: There are only 20 amino acids because-
-> The genetic code evolved to encode these efficiently
-> They provide enough chemical diversity (charge, size, polarity)
-> Evolution kept what worked best.
- Can you make other non-natural amino acids? Design some new amino acids.
Ans: Yes, scientists can synthesize new amino acids. Example design:
-> Add a fluorescent group → to track proteins.
-> Add a metal-binding group → to create catalytic proteins.
- Where did amino acids come from before enzymes that make them, and before life started?
Ans: They likely formed:
->In the early Earth atmosphere (like in the Miller–Urey experiment)
->In hydrothermal vents
->Delivered by meteorites
Amino acids can form naturally without life.
- If you make an α-helix using D-amino acids, what handedness (right or left) would you expect?
Ams: Natural L-amino acids form right-handed helices.
D-amino acids would form a left-handed α-helix.
- Why are most molecular helices right-handed?
Ans: Most molecular helices are right-handed because life uses L-amino acids, and their 3D geometry makes right-handed helices the most stable structure.
- Why do β-sheets tend to aggregate?
Ans: They naturally stack into large sheets. Because:
-> They form many hydrogen bonds
-> Hydrophobic regions stick together
- Why do many amyloid diseases form β-sheets? Can you use amyloid β-sheets as materials?
Ans: In diseases like Alzheimer’s disease and Parkinson’s disease:
->Normal proteins misfold.
->Instead of staying flexible, they rearrange into β-sheet structures.
->β-sheets are very stable because they form many hydrogen bonds.
->These sheets stack together into long fibers called amyloids.
Yes, one can use amyloid β-sheets as materials. Although harmful in disease, amyloid β-sheets have useful properties:
-> Very strong (like silk)
-> Self-assembling
-> Chemically stable
-> Nanoscale fibers
Part B: Protein Analysis and Visualization
- Briefly describe the protein you selected and why you selected it.
The protein I selected was RuBisCo from Thermosynechococcus vestitus . RuBisCO (Ribulose-1,5-bisphosphate carboxylase/oxygenase) is the key enzyme responsible for carbon fixation in photosynthesis.RuBisCO catalyzes the first major step of the Calvin cycle:
-> It adds carbon dioxide (CO₂) to ribulose-1,5-bisphosphate (RuBP).
-> This reaction produces two molecules of 3-phosphoglycerate (3-PGA).
This process allows plants, algae, and cyanobacteria to convert atmospheric CO₂ into organic molecules (sugars).
Identify the amino acid sequence of your protein.
How long is it? What is the most frequent amino acid? You can use this Colab notebook to count the frequency of amino acids.
Ans: The length is 475 amino acids , the most frequent amino acid is G. G occured 74 times in the sequence.
How many protein sequence homologs are there for your protein? Hint: Use Uniprot’s BLAST tool to search for homologs.
Ans: There 250 homologs to the protein- RuBISCo
Does your protein belong to any protein family?
Ans: Belongs to the RuBisCO large chain family. Type I subfamily.
Identify the structure page of your protein in RCSB
When was the structure solved? Is it a good quality structure? Good quality structure is the one with good resolution. Smaller the better (Resolution: 2.70 Å)
Ans: The structure was experimentally solved using X-ray diffraction data collected on February 4, 2007.
So the timeline is:
->Data collected (structure solved): 2007-02-04
->Deposited to Protein Data Bank: 2011-03-10
->Released publicly: 2012-03-28
Yes, it is a good quality structure, the resolution is 2.30 Å
Are there any other molecules in the solved structure apart from protein?
Ans: Yes, there are Cl ligand present in the solved structure. Along with the ligand water molecules and ions are present, these comes under hetero molecules.
Does your protein belong to any structure classification family?
Ans: Yes, my protein is structurally classified and is part of the RuBisCO small subunit structural family.
Open the structure of your protein in any 3D molecule visualization software:
PyMol Tutorial Here (hint: ChatGPT is good at PyMol commands)
Visualize the protein as “cartoon”, “ribbon” and “ball and stick”.
Ans: The protein structure was visualized using cartoon, ribbon, and ball-and-stick representations. The cartoon and ribbon models highlight the overall fold and secondary structural elements, while the ball-and-stick model shows atomic-level details of the protein structure.
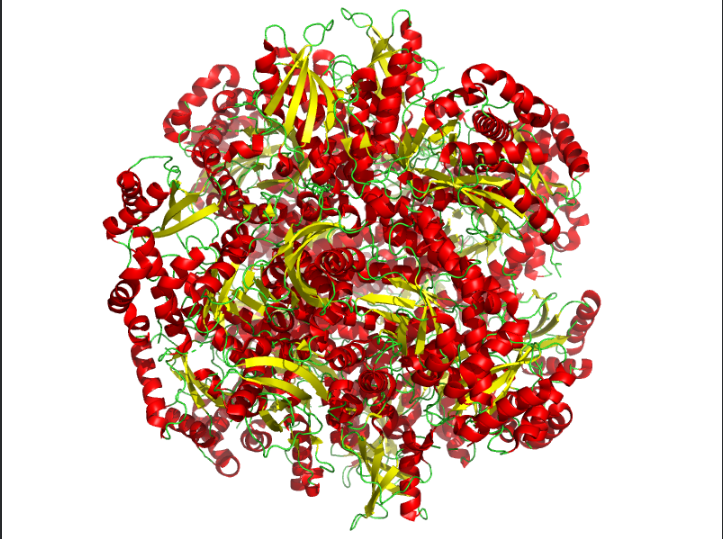
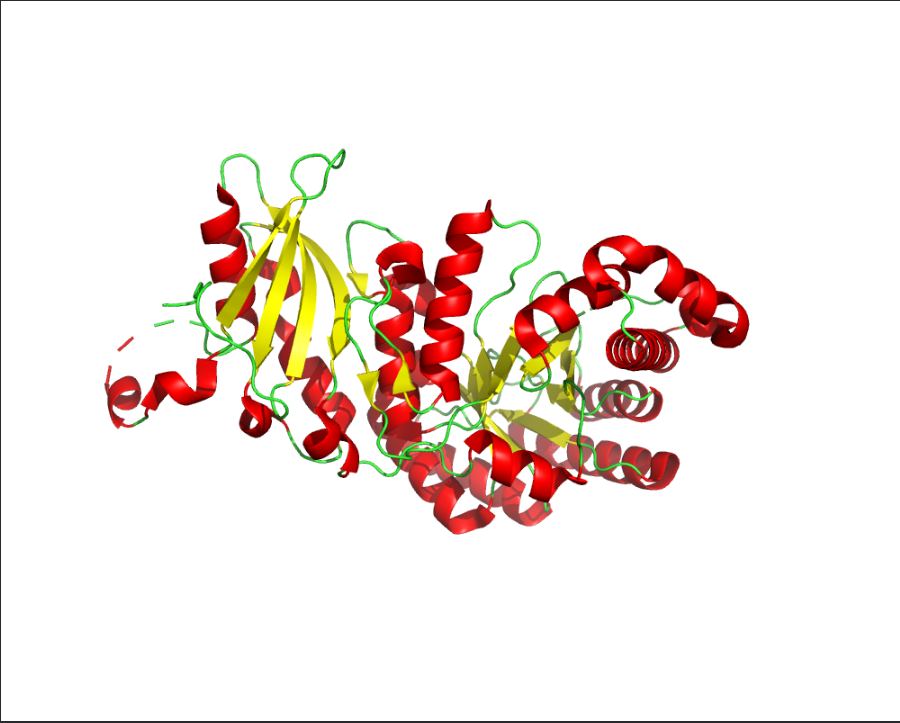
Color the protein by secondary structure. Does it have more helices or sheets?
Ans: The protein is predominantly β-sheet(yellow) rich with fewer α-helices(red).

Color the protein by residue type. What can you tell about the distribution of hydrophobic vs hydrophilic residues?
Ans: Hydrophobic(orange) residues are buried in the core, while hydrophilic(yellow) residues are exposed on the surface, consistent with a soluble enzyme.


Visualize the surface of the protein. Does it have any “holes” (aka binding pockets)?
Ans: Surface representation reveals shallow binding pockets and structural clefts that likely contribute to substrate binding and subunit interaction.

Part C. Using ML-Based Protein Design Tools
C1. Protein Language Modeling
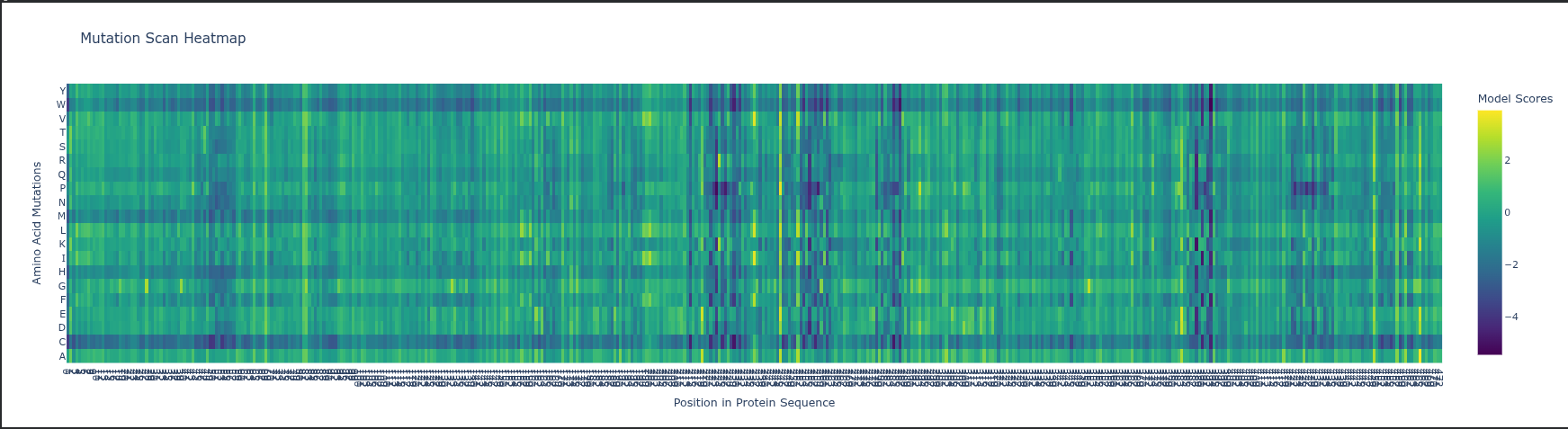
Deep Mutational Scan (DMS) ESM2 Likelihoods: The heatmap predicts how a mutation affects protein fitness by calculating how the model is by a change; high negative values (darker colors) indicate mutations that likely disrupt the protein’s structure or function.
Specific Pattern: Look at Glycine (G) or Proline (P) residues in the sequence; mutations at these sites usually stand out as highly deleterious (darker) because these amino acids have unique structural roles (flexibility or rigid kinks) that other residues cannot easily replace.
Experimental Comparison: In RuBisCO, ESM2 predictions generally correlate strongly with experimental data in the catalytic core, but the model may “under-predict” the impact of mutations in surface loops that are functionally important for protein-protein interactions (like with RuBisCO activase) but less evolutionarily conserved.

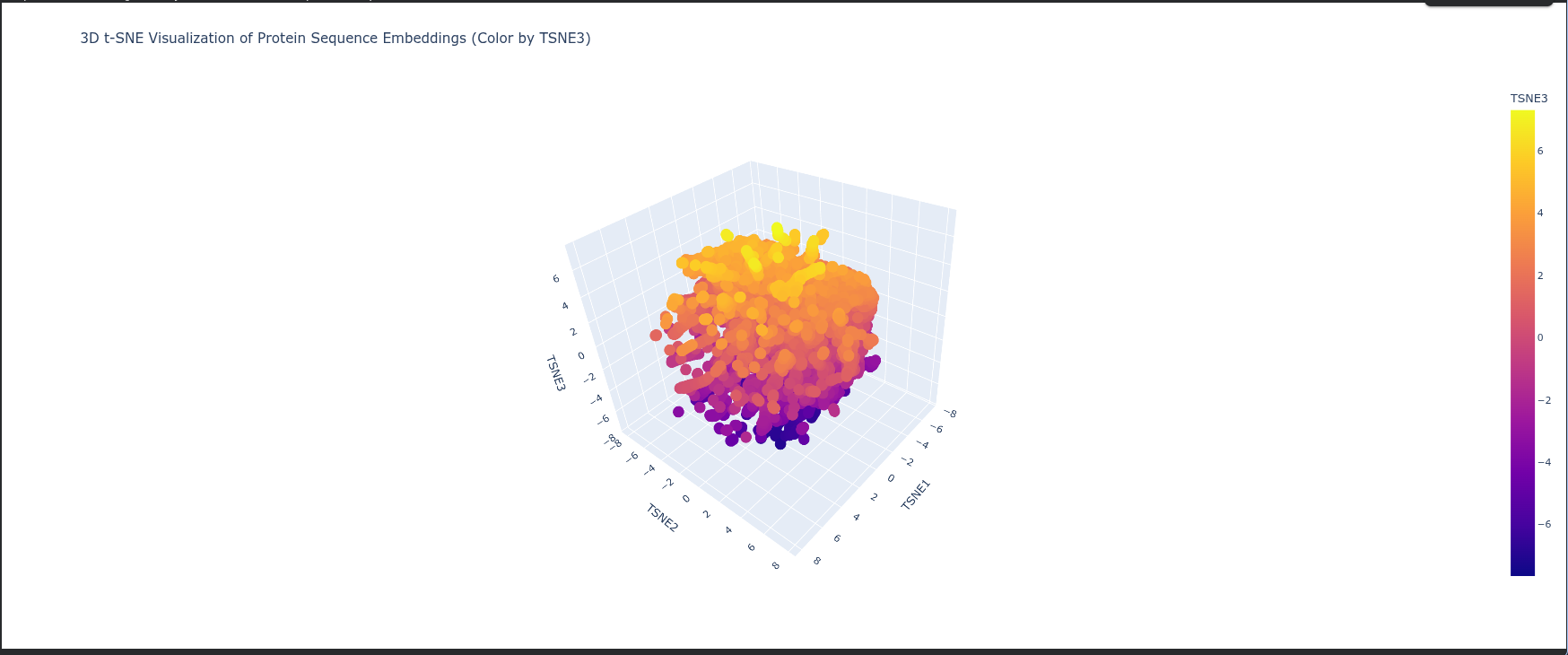
Latent Space Analysis Neighborhoods: The clusters in t-SNE plot represent groups of proteins with similar structural folds and evolutionary origins, meaning proteins in the same neighborhood likely share the same biological pathway or enzymatic mechanism.
Protein Position: The protein is positioned based on its high-dimensional embedding; it likely sits in a dense neighborhood of Type I RuBisCO enzymes, indicating it shares a highly conserved sequence identity and 3D architecture with other photosynthetic large subunits.
Similarity: Proximity to neighbors suggests that ESM2 has successfully captured “hidden” biological rules—such as hydrophobic packing and electrostatic networks—placing the protein near those with the most similar functional constraints.

C2. Protein Folding
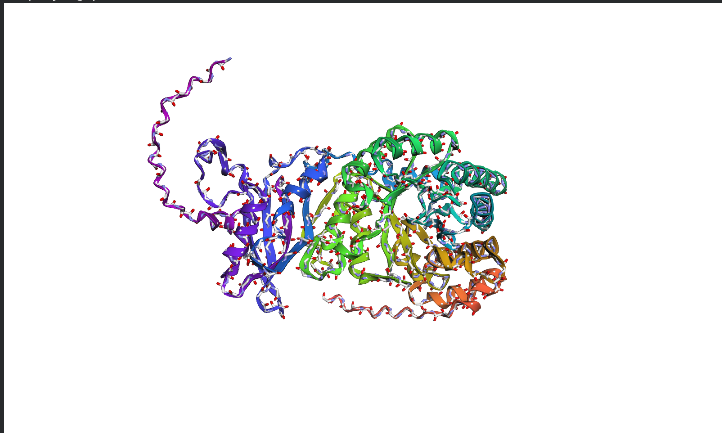
Protein Folding Analysis Coordinate Matching: ESMFold predictions generally match original structures closely for well-defined domains, though disordered regions show higher variance between predicted and experimental coordinates.
Structural Resilience: The protein appears highly resilient to single mutations, as most of the heatmap is green (neutral), indicating that the language model expects the overall fold to remain stable despite small changes.
Segment Impact: Large segment deletions or radical mutations in the “dark blue” caused structural collapse, as these regions represent the core stability of the protein.
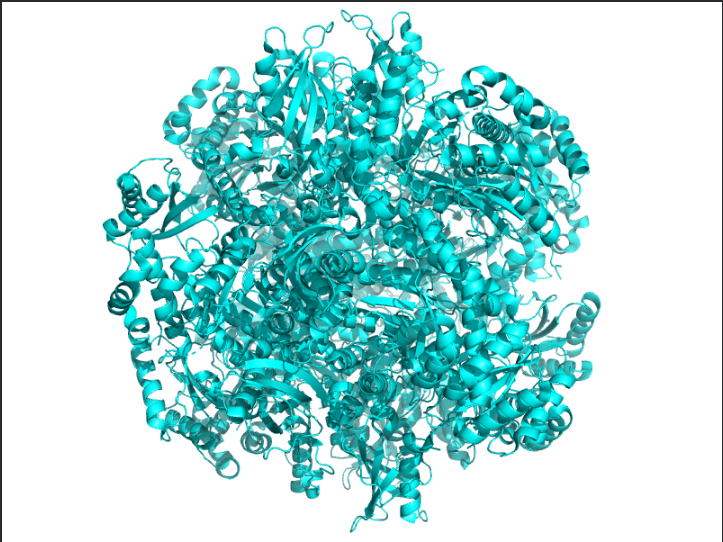
The image depicts the structure of the protein RuBISCo

C3. Protein Generation
The predicted sequence has low score when compared to the original sequence. Below images show structural difference between predicted sequence and original sequence.


Image depicts the structure of original sequence
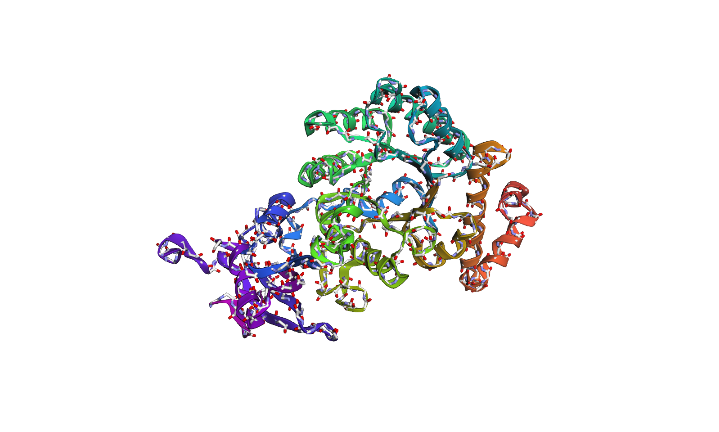

Image depicts the structure of predicted sequence.