Week 1 HW: Principles and Practices
Principles and Practices
Overview
Ethics, safety, and security are essential considerations throughout (and beyond!) this class. We have therefore designed the Class Assignment this week to give you a strong foundation, and then will ask you to reflect each week and in the design of your final project.
- First, describe a biological engineering application or tool you want to develop and why
Chagas disease is a chronic disease caused by the flagellate protozoan Trypanosoma cruzi. This disease represents a major public health problem in Latin America, due to it is endemic in 21 countries of the region. Recently, the active vectorial transmission has been also recognized in the United States (Beatty NL et al., 2025). Besides this, in the last few decades, the epidemiological pattern of the disease changed due to the population migrations, and it is estimated that more than 8 million people is infected around the world and more than 75 million people are at risk of infection, according to Pan American Health Organization (PAOH).
Considering that this disease particularly affects low and middle-income countries, especially the most isolated and lowest-income social sectors, both the World Health Organization (WHO) and PAHO consider it as part of the group of neglected diseases.
Currently, there are only tow drugs available for treating the disease: Benznidazole (Roche) and Nifurtimox (Bayer). However, the success of the treatment depends on the stage of infection, the age of the patient, and the region where the infection was contracted. In addition to theses limitations, trypanocide drugs have significant toxicity profiles associated with both drugs: adverse reactions occur in up to 40% of treated patients, leading to treatment discontinuation (Centers for Disease Control and Prevention [CDC]).
For this reason, it is essential to urgently develop new drugs that are effective and well tolerated by patients.
PROPOSE: PROTEIN-BASED DRUGS AGAINST CHAGAS DISEASE
The proposed idea consists of developing a new therapeutic alternative using the specialized nutrient uptake and endocytosis system of these parasites, through cell membrane receptor proteins, as anchors for mini-protein modules capable of triggering the internalization of protein drugs.
This is based on the fact that these protein drugs can be specifically designed to affect the parasite’s own processes, such as breaking protein-protein interactions involved in the formation of essential complexes, inhibiting their development without affecting the processes of human cells, since they do not release other cellular components and cannot be internalized by them.
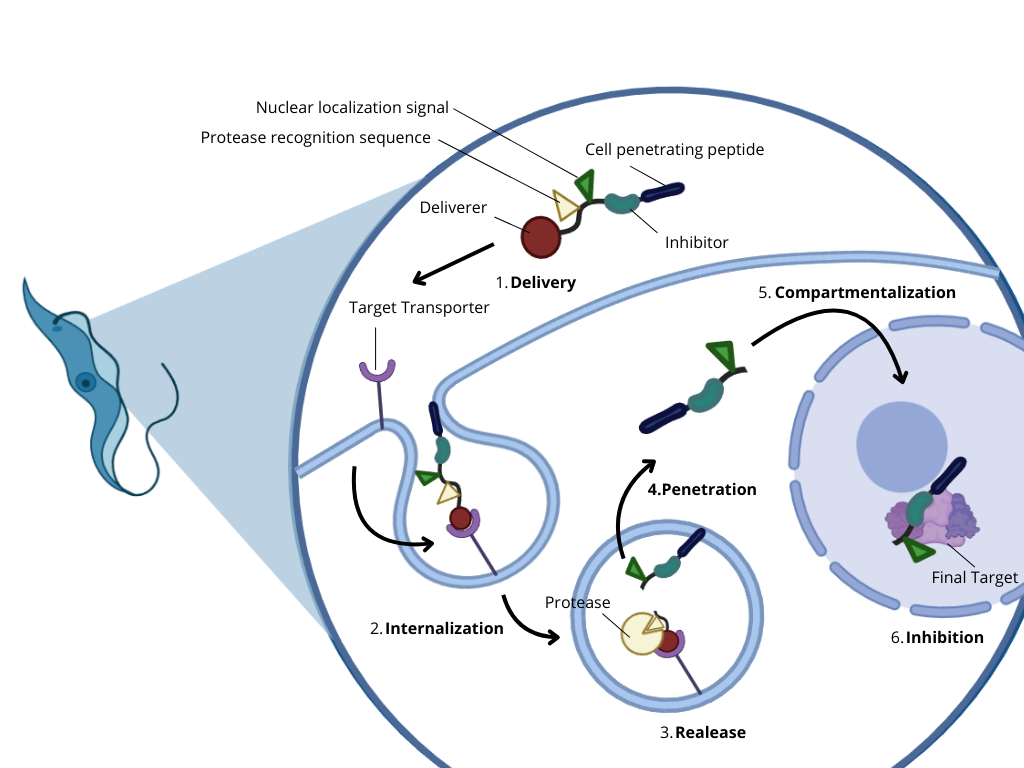

- Next, describe one or more governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future, like ensuring non-malfeasance (preventing harm). Break big goals down into two or more specific sub-goals
Is it ethical to develop a treatment that is then inaccessible to the affected populations? Is it valid that patents exist for this type of scientific advance, thereby blocking the possibility of generic drugs? What happens when these developments are carried out with public funds?
GENERAL OBJECTIVE I: Ensure equitable access to treatment distribution
SPECIFIC OBJECTIVES:
- Create joint working groups between the public and private sectors to prevent a monopoly in both the development and distribution of treatment.
- Create differential regulations for neglected diseases so that access to treatment is commensurate with the affected populations.
Which populations are considered for participation in clinical trials? Is this population truly informed about the risks, obligations, and responsibilities of each actor involved in the trials? Is it ethical to withhold treatment or use placebos in affected individuals when a drug is available? What happens when research is conducted in countries where this disease is not a health problem?
GENERAL OBJECTIVE II: Ensure biosafety in the development and use of the new therapeutic alternative
SPECIFIC OBJECTIVES:
- Avoid scientific extractivism by promoting international cooperation and conducting multicenter clinical trials.
- Promote clinical evaluation by ensuring equitable population representation, i.e., including different sectors such as patients infected through different routes of transmission, both sexes, and different ages.
How can we expect progress if knowledge remains confined to healthcare stakeholders and scientists? Why do we, as a scientific community, believe that only we can bring about positive change? Are we caring for the community in this way?
GENERAL OBJECTIVE III: Protect and involve the community
SPECIFIC OBJECTIVES:
- Avoid power asymmetry by ensuring training for both the healthcare professionals involved and the general population, guaranteeing clear information not only about treatment but also about prevention and diagnosis, thereby promoting social participation as active generators of change.
- Promote transparency in the process of communicating the advantages/benefits and possible risks related to treatment options, with the aim of leading to an informed decision.
- Next, describe at least three different potential governance “actions” by considering the four aspects below (Purpose, Design, Assumptions, Risks of Failure & “Success”)
To ensure that these objectives are met, various strategies can be proposed, including the following actions:
I. Develop a specific regulatory and normative framework for neglected diseases
The objective of creating this regulatory framework is to ensure equitable access, regardless of purchasing power and the geographical area in which the affected people are located, a key aspect when dealing with a disease that mainly affects vulnerable populations.
To this end, a multidisciplinary commission should be formed to develop regulations that bring together both public sector actors involved in developing this therapy and private sector actors who will be responsible for conducting clinical trials and scaling up treatment, as well as doctors and healthcare personnel who will act as the link between them and patients, in addition to representatives of the national bodies responsible for regulating, supervising, and authorizing all medicines. Of course, this commission must include members of the state/government who promote the existence of this commission and support the necessary measures both legislatively and financially.
It should be noted that this regulatory framework will not only focus on regulating accessibility from an economic perspective, but will also consider other aspects such as access to nearby public health facilities and basic hygiene conditions, among many other key aspects for controlling the disease.
II. Conduct awareness campaigns and create a monitoring system to ensure biosafety
Although there are currently epidemiological and awareness campaigns targeting the general population in Argentina, misinformation is a significant barrier, and since this is a new therapy, it is essential to reinforce certain aspects to promote its acceptance and correct use.
In this regard, part of the objective of this action is to develop training programs for health professionals on the mechanism of this new therapy and the key aspects to consider for its use, as well as training stages for the general population that contribute to ensuring its correct use, reinforcing the concept that regardless of existing therapies, it is always essential to reduce the risk of new transmissions. To implement this, specific workshops will be held for professionals, workshops that connect them with the affected populations, and information campaigns in the media to involve the general population.
On the other hand, in conjunction with these measures, it is also important to have a monitoring system in place to evaluate the long-term impact of the therapy, which allows for the detection of adverse effects and unforeseen events of any kind, improves its implementation, and gauges public opinion.
III. Establish a specific regional coordination system for neglected diseases
The objective of this action is to promote joint international government actions, at least in Latin America, for the evaluation, use, and financing of therapies such as the one proposed in this case. Furthermore, considering that the tool can be edited specifically against the parasite, it can be adapted to different strains of the parasite and its use can be extended to all strains present in the Americas.
In turn, the existence of this regional coordination promoted by high-impact international organizations such as PAHO or WHO means that this tool is also known to the entire community and can be modified and applied to treat other trypanosomatids, promoting technology transfer. Ultimately, it can also be evaluated for the treatment of other diseases or scientific applications.
- Next, score (from 1-3 with, 1 as the best, or n/a) each of your governance actions against your rubric of policy goals. The following is one framework but feel free to make your own
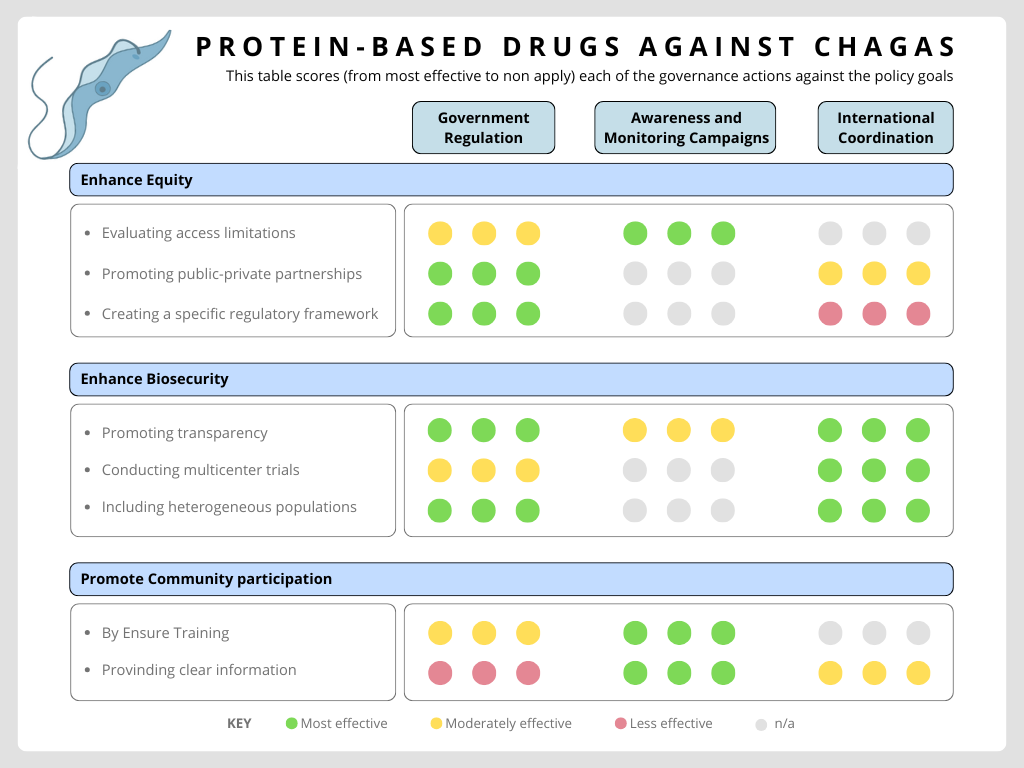

- Last, drawing upon this scoring, describe which governance option, or combination of options, you would prioritize, and why. Outline any trade-offs you considered as well as assumptions and uncertainties
According to the scores, it can be seen that not all measures met all the proposed objectives. Personally, I believe that the priority measure would be government regulation. In Argentina, there is a law, Law 26,281 on Chagas disease (passed in 2007, regulated in 2022), which declares the prevention, diagnosis, and free treatment of Chagas disease to be of national interest, with the aim of eradicating it. I believe it is essential that, when proposing a new treatment, the limitations on access for the current population be addressed and agreements between the public and private sectors be promoted so that the law is actually enforced.
Although the proposal assumes greater technical effectiveness and lower costs as a result, given that it not only benefits the infected person but also reduces costs for the public health system by reducing complications and side effects, as well as the assumption of collective work with the private sectors, it should be noted that compliance with laws and funds allocated to public health policies in countries such as Argentina and all of Latin America is subject to the government in power. In addition, it is assumed that there will be consensus between public and private actors, but it is known that this depends on various interests that do not always prioritize the affected population.
On the other hand, another priority measure is to promote international cooperation. For some years now, non-endemic countries have shown interest in studying the problem, and international collaboration has been encouraged in the field of scientific research. However, I believe that this aspect still needs to be further strengthened and supported by a regulatory framework. This is important not only to promote the biosafety of the new treatment but also to contribute to equity in different countries and to encourage advances in the search for treatments for other trypanosomiasis diseases. The latter is based on the assumption that the proposed technology will work similarly in real conditions to those in the laboratory and with the ability to adapt to different types of trypanosomiasis.
References:
- Pérez-Molina JA, Molina I (2018) Chagas disease. The Lancet 391:82–94. https://doi.org/10.1016/S0140-6736(17)31612-4.
- (2026) World Health Organization (WHO) web site. https://www.who.int/. Accessed 1 Feb 2026.
- Pérez-Molina JA, Crespillo-Andújar C, Bosch-Nicolau P, Molina I (2021) Trypanocidal treatment of Chagas disease. Infectious Diseases and Clinical Microbiology (English ed) 39:458–470. https://doi.org/10.1016/j.eimce.2020.04.012
- Muratspahić E, Feldman D, Kim DE, et al (2025) De novo design of miniprotein agonists and antagonists targeting G protein-coupled receptors. bioRxiv 1–34. https://doi.org/10.1101/2025.03.23.644666.
- Lee J, Case JB, Park Y, et al (2024) A pan-variant miniprotein inhibitor protects against SARS-CoV-2 variants.
- Chazin-Gray AM, Thompson TR, Lopatto EDB, et al (2025) De Novo Design of Miniprotein Inhibitors of Bacterial Adhesins. BioRxiv 135390:1–56.
- Ciesiołkiewicz A, Lizandra Perez J, Skalniak L, et al (2024) Miniprotein engineering for inhibition of PD-1/PD-L1 interaction. Protein Science 33:1–14. https://doi.org/10.1002/pro.5106.
- Luciano G. Levin, Pablo R. Kreimer & Pablo Jensen (2021): Chagas Disease across Contexts: Scientific Knowledge in a Globalized World, Medical Anthropology, DOI: 10.1080/01459740.2021.1946805