Week 2 Lab: Restriction Digests
Protocol
Part 0: Design

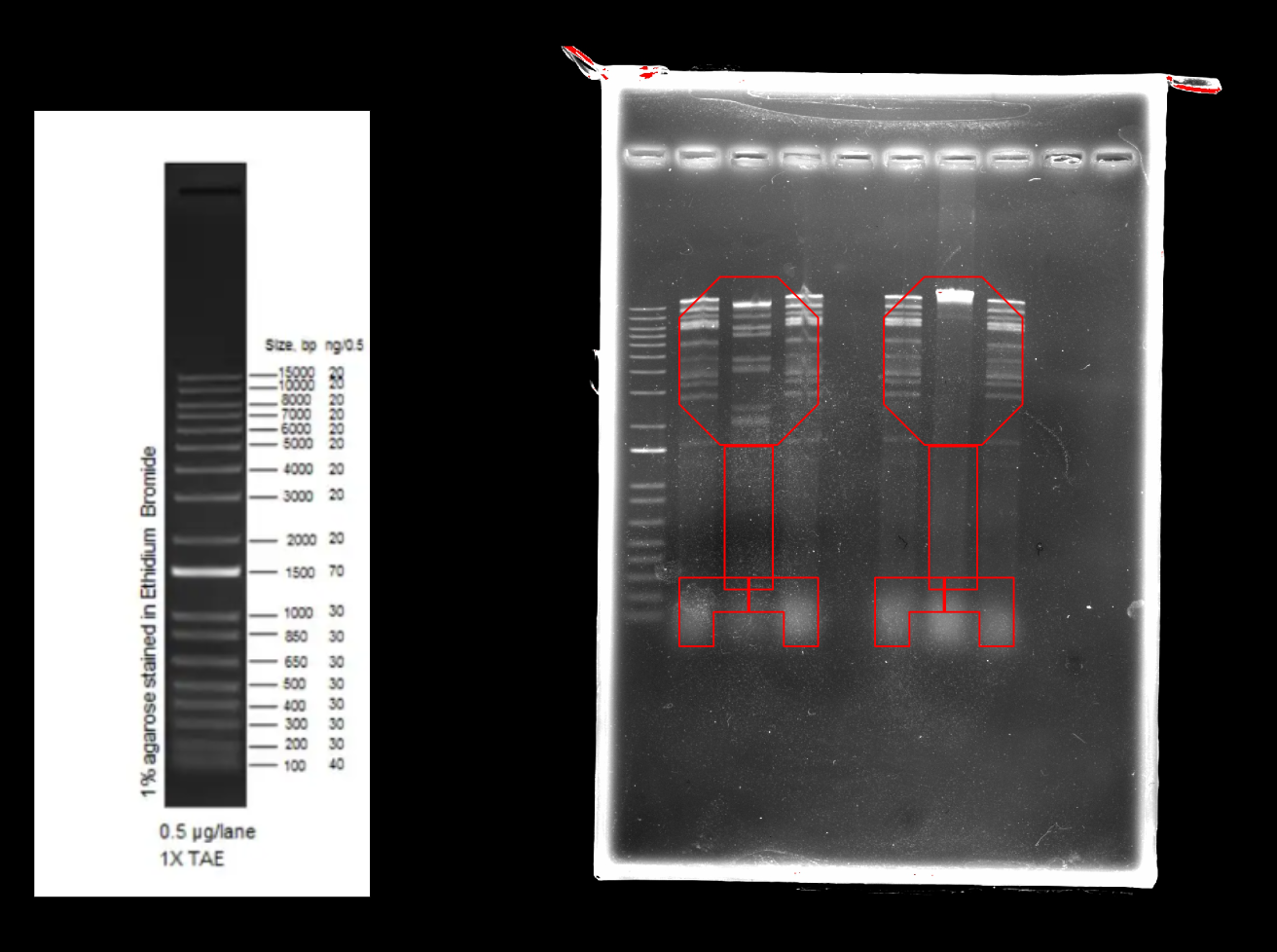
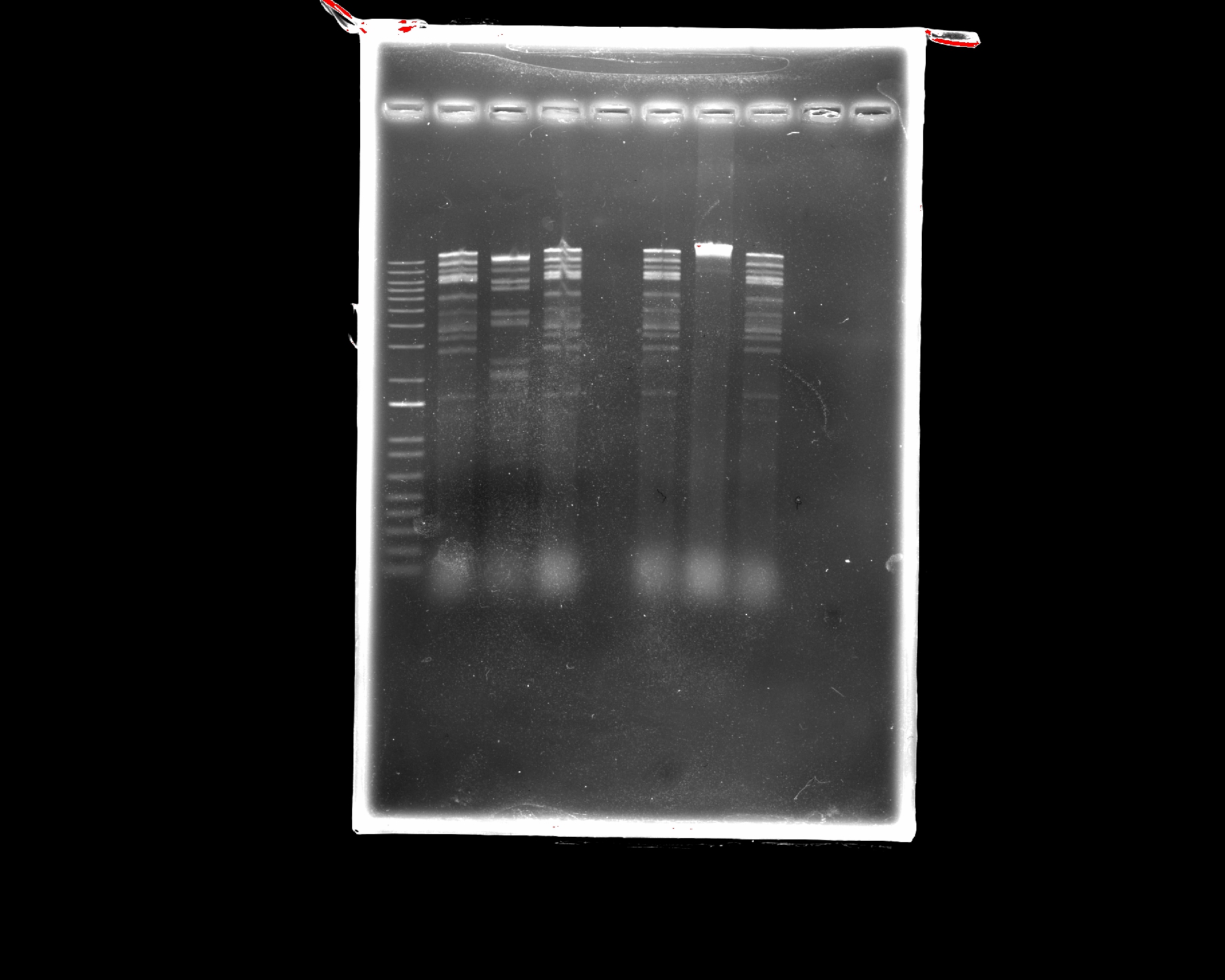
 Note: Ignore EcoRI lane: for visualization
Note: We are using DNA from mycobacteriophage Kampy
Note: Ignore EcoRI lane: for visualization
Note: We are using DNA from mycobacteriophage Kampy
Part 1a: Gel prep
Setting up a 1% agarose gel:
- 0.5 g agarose to plastic flask
- 50 ml 1x TAE to agarose in flask
- Microwave the mixture 1:30 / until agarose dissolves
- Sideways gel tray in box, pour hot agarose/TAE, add comb
- Let set 30 min
Part 1b: Restriction Digest
Chemicals
- 1X Lambda DNA
- 1 uL of each enzyme: EcoRI-HF, HindIII-HF, BamHI-HF, KpnI-HF, EcoRV-HF, SacI-HF, SalI-HF
- Nuclease-free water Equipment and Consumables
- -20ºC freezer
- Incubator (or use a thermocycler or heat block
- “PCR tube rack” (pipette tip holder)
Digest requirements
- 3 ul 0.5 ug/uL DNA – final conc 1.5 ug/20 ul total
- 2 uL 10x buffer
- 1 uL 20 units/uL enzyme – final conc 15 units
- NFW: enough to make 20 uL total mixture
our DNA = 324 ng/uL = 0.324 ug/uL– to get 1.5 ug, we need 4.62 uL of DNA
Plan for Mixtures
4.62 uL DNA
2 ul 10x buffer
BstXI: to get final amt 15 ug – 37 – H buffer
- 1.5 ul of 10 u/ul
- 11.88 uL NFW
KpnI: to get final amt 15 ug – 37 – MC buffer
- use 1.25 ul of 12 u/ul
- 12.13 uL
SfiI: to get final amt 15 ug – 50 – B buffer
- use 1.5 ul of 10 u/ul
- 11.88 uL NFW
Incubate at temp – 1 hr
Notes/Scratch work
Done: NFW, DNA, Buffer, Enzyme
Running
BstXI – + assoc buffer – 37oC
- stock buffer: promega 10x
- enzyme: 10 u / ul
BstXI – + assoc buffer – 37oC
KpnI – + assoc buffer – 37oC
- stock buffer: promega 10x buffer
- enzyme: 12 u / ul
SfiI – + assoc buffer – 50oC
- stock buffer: promega 10x
- enzyme: 10 u / ul
See chart on site for reagent concs and quantities
Part 2: Gel Run
- pour TAE over fill line
- load 12 ul ladder
- add 4 ul 6x dye to ea. 20 ul digest
- load 10 ul of the above on the gel
- set up electrophoresis and run ~30 min
- let stain in ethidium bromide 20 min
- image with gel imager
Results