Week 7 HW: Genetic Circuits Part 2: Neuromorphic Circuits
Assignment Part 1: Intracellular Artificial Neural Networks (IANNs)
Question 1
Traditional genetic circuits are usually implemented in Boolean logic (ON/OFF), hand-designed as fixed logic. so representing nuanced behaviors often requires many gates, sharp thresholds, and careful tuning, which can make designs bulky and brittle. As the number of inputs grows the circuit complexity can explode combinatorially, increasing burden by stacking multiple layers and adding intermediate nodes, which increases metabolic load, failure points, and sensitivity to part-to-part variability Also, adapting to new targets or shifting biological context often means redesigning the circuit architecture, not just re-tuning parameters.
Intracellular Artificial Neural Networks (IANNs) are parametric and trainable: you can adjust “weights” to fit a desired behavior from data (calibration/learning), then iterate as conditions change. This is more condizent with the noisy and complex nature of biological signals. IANNs are parametric and trainable, designed to operate on analog inputs, tolerate noise through distributed computation, and approximate complex decision boundaries without enumerating every logic case. Using IANN you can adjust “weights” to fit a desired behavior from data (calibration/learning), then iterate as conditions change, which is in general a very wanted feature for biological modelling.
Question 2
A useful application for an IANN could be a multi-signal “smart probiotic” controller that decides when to express a therapeutic payload in the gut based on a noisy inflammation signature. This could be a proposed pipeline:
Sensors detect several analog inputs. These can be related to a mesurable intracellular signal (i.e. information on promoters/sensors response to nitrate/NO, tetrathionate, ROS, and low pH <-> measurable intracellular signal like transcription rate or a regulator concentration)
The IANN integrates these signals as weighted contributions and computes a graded output: a continuously tunable expression level of a payload gene (e.g., an anti-inflammatory cytokine mimic, a barrier-protective peptide, or a locally acting enzyme), plus an optional reporter for monitoring.
Instead of requiring all conditions to be “true” or “false,” like Boolerian models the IANN can implement a “risk score” that turns on strongly only when the combined pattern matches inflammation, while remaining low for benign fluctuations. In practice, you would calibrate the weights using training data from known conditions (healthy vs inflamed models) so the output tracks the probability or intensity of the target state.
Limitations / failure modes: IANNs still face real biological constraints such as sensor cross-talk and context effects. These can shift input distributions. Also, weights can drift as cells evolve, and metabolic burden can reduce growth or change the very physiology being measured. The dynamic range and noise of biological parts can compress signals, making it hard to separate “moderate” from “high” states without careful normalization and controls. Time dynamics also matter: inputs arrive on different timescales (transcription vs metabolites), so the network may need memory/filters to avoid reacting to transient spikes, which can substantially increase the complexibility of the network. Finally, safety and containment become part of the spec, thus important to define acceptance balance between error type 1 and 2 defining if you’d likely need a kill switch and strict limits on maximum output to avoid unintended activation in off-target contexts.
Question 3
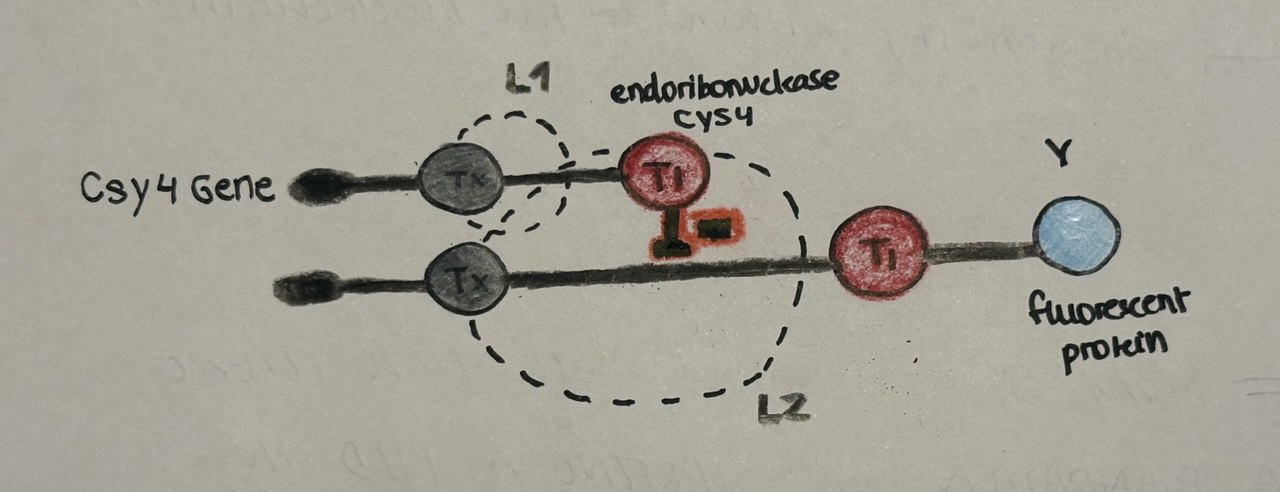
Assigment Part 2: Fungal Materials
Question 1
Example 1: Mycelium composite foams (grown on agricultural waste)
Used for protective packaging, insulation panels, acoustic damping, and lightweight cores.
Advantages: renewable feedstocks, low-temperature manufacturing, biodegradable or compostable end-of-life, and tunable density via growth conditions.
Disadvantages: mechanical properties can vary batch-to-batch, moisture sensitivity unless coated, and long-term durability and standards testing can be harder than for petrofoams.
Example 2: Mycelium “leather” (mycelium-based sheets)
Used for footwear, bags, apparel, and upholstery as a leather alternative.
Advantages: avoids the animal leather supply chain, potentially lower land and chemical burden, and tunable texture and thickness.
Disadvantages: still often needs finishing steps for durability and water resistance, performance can lag high-grade leather, and cost and scale are still improving.
Example 3: Fungal biocement or mycelium-bound “bio-bricks”
Used for low-load building blocks, interior architectural elements, and decorative panels.
Advantages: low-energy fabrication, can use local waste substrates, lightweight, and potentially lower embodied carbon than fired bricks or some concretes.
Disadvantages: typically not comparable to concrete for structural strength, humidity and fire performance require careful engineering, and regulatory acceptance is slower.
Example 4: Fungal pigments and dyes (fermentation-derived)
Used for textiles, inks, coatings, and cosmetics.
Advantages: renewable production, avoids some petroleum-derived dye routes, and potentially lower toxic byproducts depending on the process.
Disadvantages: stability and colorfastness can be challenging, purification costs can be nontrivial, and some pigment pathways have safety constraints depending on the organism and compound.
Question 2
One may want to tune mycelium architecture (hyphal branching, wall composition, and crosslinking) to achieve specific strength, flexibility, porosity, and water resistance for composite materials. Another application is producing programmable functional materials by engineering fungi to secrete adhesives, hydrophobins, melanin-like coatings, or crosslinking enzymes so the final material is tougher or more water-stable without heavy post-processing.
Beyond material applications, genetically engineered fungi can be used for biosensing if we add genetic circuits that turn on a visible reporter in response to VOCs, toxins, inflammation markers, or pollutants, enabling living “sensor materials.” They can also be used for biomanufacturing high-value enzymes, small molecules, and therapeutics that benefit from eukaryotic processing or secretion, and for bioremediation by enhancing the breakdown of lignin, plastic additives, dyes, PFAS-like contaminants (where feasible), or heavy-metal binding, depending on pathway and safety constraints.
Fungi can be advantageous over bacteria because filamentous growth lets them act as a self-assembling scaffold, so the organism is both the “factory” and the “fabrication method.” They also offer eukaryotic protein processing because fungi handle disulfide bonds, folding, secretion, and many post-translational modifications better than most bacteria, which matters for secreted enzymes and complex proteins. In addition, fungi naturally secrete many enzymes, which is ideal for biomass conversion and environmental breakdown workflows. Another advantage relative to bacteria is metabolic breadth since fungi often tolerate more extreme acidic conditions and diverse feedstocks, and many are strong at producing secondary metabolites.
However, bioprocesses with engineered fungi may have practical limitations compared with bacteria, such as slower growth and iteration, more complex regulation and morphology (heterogeneity in filamentous cultures can make outputs less uniform), and genetic tools that can be trickier because strain engineering and predictable expression are often less plug-and-play than in E. coli.