Week 11 HW: Bioproduction & Cloud Labs
Part A: The 1,536 Pixel Artwork Canvas | Collective Artwork
I contributed 7 pixels to the global artwork experiment, helping extend a horizontal yellow line in the top-left area (see screenshot below).
At first, I was cautious and tried to understand the ongoing ideas for each section and whether there was a unifying concept. I considered introducing something new, but ultimately decided to stick with what seemed to be the area’s goal (a horizontal yellow line). For next year, it might be fun to have an in-app chat within the same domain to coordinate contributions more easily and check the current vibes.

Part B: Cell-Free Protein Synthesis | Cell-Free Reagents
Question 1
| Component Category | Component | Corrected role in the cell-free reaction |
|---|---|---|
| Lysate | E. coli Lysate | Provides the endogenous transcription, translation, and metabolic machinery needed for in vitro gene expression. |
| BL21 (DE3) Star Lysate (includes T7 RNA Polymerase) | Provides the same core lysate machinery plus T7 RNA polymerase for strong transcription from T7 promoter templates. | |
| Salts / Buffer | Potassium Glutamate | Helps set intracellular-like ionic conditions that support enzyme activity, ribosome function, and overall reaction performance. |
| HEPES-KOH pH 7.5 | Maintains reaction pH in the range needed for stable transcription-translation activity. | |
| Magnesium Glutamate | Supplies Mg2+, an essential cofactor for ribosomes, polymerases, and many ATP-dependent enzymes. | |
| Potassium phosphate monobasic | Contributes phosphate and helps maintain buffer balance together with the dibasic form. | |
| Potassium phosphate dibasic | Works with the monobasic form to maintain phosphate buffering and reaction stability. | |
| Energy / Nucleotide System | Ribose | Supports nucleotide metabolism and regeneration pathways rather than serving as the main energy source. |
| Glucose | Serves as a metabolic energy substrate that helps regenerate ATP through endogenous lysate metabolism. | |
| AMP | Acts as a nucleotide monophosphate precursor that can be phosphorylated into higher-energy adenine nucleotides. | |
| CMP | Acts as a nucleotide precursor that can be converted into CTP for transcriptional needs. | |
| GMP | Acts as a nucleotide precursor that can be converted into GTP for transcription and translation-related processes. | |
| UMP | Acts as a nucleotide precursor that can be converted into UTP for transcriptional needs. | |
| Guanine | Serves as a salvage precursor for guanine nucleotide synthesis. | |
| Translation Mix (Amino Acids) | 17 Amino Acid Mix | Provides most of the amino acid building blocks required for protein synthesis. |
| Tyrosine | Provides a required amino acid for translation and may also be supplied separately because of formulation or pathway-specific needs. | |
| Cysteine | Provides a required amino acid for translation and is often added separately because of its chemical instability. | |
| Additives | Nicotinamide | Serves as a precursor for NAD-related cofactors that support extract redox metabolism. |
| Backfill | Nuclease Free Water | Brings the reaction to the target volume without introducing nucleases or contaminants. |
Question 2
The 1-hour PEP-NTP system supplies fully activated NTPs and high-energy phosphate (PEP) upfront, enabling fast, high-rate transcription and translation but with limited longevity due to rapid energy depletion.
In contrast, the 20-hour NMP-ribose-glucose system relies on metabolic regeneration, using NMPs and simple substrates (ribose, glucose) that are enzymatically converted into active nucleotides and ATP, trading peak speed for sustained, longer-duration protein production.
Part C: Planning the Global Experiment | Cell-Free Master Mix Design
Question 1
a. Superfolder Green Fluorescent Protein (sfGFP)
Description: a basic (constitutively fluorescent) green fluorescent protein published in 2005, derived from Aequorea victoria. It is reported to be a very rapidly-maturing weak dimer.
sfGFP has very efficient folding and fast maturation (~13 min), allowing it to produce fluorescence quickly and reliably even under suboptimal cell-free conditions. This makes it ideal for early and robust readout.
b. Monomeric Red Fluorescent Protein 1 (mRFP1)
mRFP1: Derived from DsRed, mRFP1 has slow maturation and lower photostability, which delays fluorescence signal and reduces effective brightness in short or energy-limited cell-free reactions.
c. mKusabira-Orange2 (mKO2)
mKO2 has moderate maturation speed but higher sensitivity to photobleaching and environmental conditions, which can reduce signal stability during long incubations or repeated excitation. This protein is relatively acid-sensitive (higher pKa), so its fluorescence can decrease if the cell-free reaction acidifies over time, affecting signal stability.
d. mTurquoise2
This protein has an exceptionally high quantum yield and photostability, making it one of the brightest CFP variants and ideal for strong signal readout even at low expression levels.
e. mScarlet_I
mScarlet-I is optimized for high brightness and improved maturation efficiency among red FPs, enabling stronger signal compared to earlier RFPs, though maturation still limits very early readouts compared to GFP variants.
f. Electra2
As a newer engineered FP (likely optimized variant), its performance is typically influenced by trade-offs between brightness, folding efficiency, and maturation kinetics, meaning signal output depends strongly on how well it folds and matures in the cell-free environment.
Question 2
Hypothesis: For mKO2, increasing the HEPES-KOH buffer concentration and maintaining sufficient glucose in the cell-free mastermix will improve fluorescence over a 36-hour incubation by reducing pH drift and sustaining ATP regeneration.
Rationale: Because mKO2 is relatively acid-sensitive, stronger pH buffering should help preserve fluorescence, while sustained glucose-dependent energy regeneration should support continued protein expression and chromophore maturation, resulting in a higher final fluorescence signal.
Small caveat: glucose can also contribute to acidification depending on the metabolism of the lysate, so the strongest version is really HEPES-KOH + controlled glucose, not just “more glucose.”
Question 3
sfGFP → system calibration (TX-TL health) Melanin has a broad absorbance spectrum, but it absorbs much more strongly at shorter wavelengths (blue/green) than at longer wavelengths (red). Melanin interferes with optical readout since we will be trying to measure fluorescence in a reaction that is simultaneously getting darker, which creates optical interference broadening the wavelengh espectrum of signal.
mScarlet-I → expression readout for melA tyrosinase especifically fluorescence is less sensitive to melanin, so it better tracks expression alone (sfGFP → Ex ~488 nm / Em ~510 nm → high overlap with melanin absorbance; mTurquoise2 → even worse (blue region); mScarlet-I → Ex ~569 nm / Em ~594 nm → less overlap).
Question 4 For optimizing the Master Mix design for mScarlet-I in my melA tyrosinase cell-free system, I’d supplement CuSO4 since my analyte is a cooper dependent enzyme, HEPES-KOH pH 7.5 to have an additional buffer against acidification and magnesium glutamate to improve translation capacity.
At first I thought about adding glucose since it could extend energy regeneration, but then I wondered that it may also increase acidification. Since you’re worried about fluorescence readout in a pigment-producing system, I’d prioritize pH stability over extra glucose.
I’d actually supplement L-tyrosine that serve as a functional validation that my protein of interest MelA tyrosinase is being expressed and active.
Master Mix designs to be tested using mScarlet-I and sfGFP:
REACTION 1
My preparation before have received email (to your email address as registered here on the Forum) providing your personal link to participate in the Cell-Free Master Mix Cloud Lab Global Experiment:
my melA-tyrosine cell-free system
mScarlet-I
| Supplement | Volume | Purpose |
|---|---|---|
| HEPES-KOH pH 7.5 | 1.0 µL | Buffer against pH drift over 36h, helping preserve mScarlet-I fluorescence and MelA activity. |
| L-tyrosine | 0.75 µL | Provides additional substrate for MelA-driven melanin-like pigment production. |
| CuSO4, very low concentration | 0.25 µL | Supports MelA tyrosinase activity as a copper-dependent enzyme while minimizing toxicity/inhibition. |
MelA-specific bottlenecks: tyrosine substrate, copper cofactor
Increasing buffering capacity with HEPES-KOH seems also a good idea because prolonged cell-free reactions coupled with melanin production lead to progressive acidification, which can reduce fluorescent protein signal, impair MelA activity, and shorten the productive lifetime of the TX-TL system.
REACTION 2
my melA-tyrosine cell-free system
sfGFP
| Supplement | Volume | Purpose |
|---|---|---|
| HEPES-KOH pH 7.5 | 1.0 µL | Buffer against pH drift over 36h, helping preserve mScarlet-I fluorescence and MelA activity. |
| L-tyrosine** | 0.75 µL | Provides additional substrate for MelA-driven melanin-like pigment production. |
| CuSO4, very low concentration | 0.25 µL | Supports MelA tyrosinase activity as a copper-dependent enzyme while minimizing toxicity/inhibition. |
MelA-specific bottlenecks: tyrosine substrate, copper cofactor
Increasing buffering capacity with HEPES-KOH seems also a good idea because prolonged cell-free reactions coupled with melanin production lead to progressive acidification, which can reduce fluorescent protein signal, impair MelA activity, and shorten the productive lifetime of the TX-TL system.
REACTION 3
my melA-tyrosine cell-free system
mScarlet-I
| Reagent | Volume | Purpose |
|---|---|---|
| L-tyrosine | 0.8 µL | Direct substrate for MelA pigment production |
| HEPES-KOH pH 7.5 | 0.6 µL | Reduces pH drift over 36h |
| Magnesium glutamate | 0.4 µL | Supports sustained transcription-translation |
| Low CuSO4 | 0.2 µL | Supports tyrosinase catalytic activity |
As copper is required as a cofactor for MelA tyrosinase activity, but must be carefully controlled because excess Cu²⁺ can inhibit cell-free expression and promote nonspecific oxidative reactions I decided to test reducing it and supplement magnesium glutamate since it improves TX-TL capacity by supporting ribosomes, RNA polymerase, and Mg-ATP/GTP chemistry.
REACTION 4
my melA-tyrosine cell-free system
sfGFP
| Reagent | Volume | Purpose |
|---|---|---|
| L-tyrosine | 0.8 µL | Direct substrate for MelA pigment production |
| HEPES-KOH pH 7.5 | 0.6 µL | Reduces pH drift over 36h |
| Magnesium glutamate | 0.4 µL | Supports sustained transcription-translation |
| Low CuSO4 | 0.2 µL | Supports tyrosinase catalytic activity |
REACTION 5
my melA-tyrosine cell-free system
mScarlet-I
| Reagent | Volume | Purpose |
|---|---|---|
| HEPES-KOH pH 7.5 | 1.25 µL | Stronger buffering against pH drift over 36h. |
| Low CuSO4 | 0.25 µL | Enables MelA tyrosinase activity as a copper-dependent enzyme. |
| Nuclease-free water | 0.50 µL | Keeps total supplement volume at 2 µL without adding more substrate. |
This reaction tests whether the main limitation is pH stability + copper availability, rather than additional tyrosine. It is useful because the base mastermix already contains tyrosine, so this condition asks whether MelA can produce pigment when copper is supplied and pH is stabilized without further increasing substrate concentration.
REACTION 6
my melA-tyrosine cell-free system
sfGFP
| Reagent | Volume | Purpose |
|---|---|---|
| HEPES-KOH pH 7.5 | 1.25 µL | Stronger buffering against pH drift over 36h. |
| Low CuSO4 | 0.25 µL | Enables MelA tyrosinase activity as a copper-dependent enzyme. |
| Nuclease-free water | 0.50 µL | Keeps total supplement volume at 2 µL without adding more substrate. |
This reaction tests whether the main limitation is pH stability + copper availability, rather than additional tyrosine. It is useful because the base mastermix already contains tyrosine, so this condition asks whether MelA can produce pigment when copper is supplied and pH is stabilized without further increasing substrate concentration.
REACTION 7
my MelA-tyrosine cell-free system
sfGFP
| Reagent | Volume | Purpose |
|---|---|---|
| L-tyrosine | 1.50 µL | Pushes substrate availability to test whether pigment formation is substrate-limited. |
| CuSO4, very low concentration | 0.25 µL | Enables MelA catalytic activity. |
| HEPES-KOH pH 7.5 | 0.25 µL | Minimal pH support. |
This is the pigment-stress condition: it intentionally pushes melanin production to test whether sfGFP fluorescence collapses when the reaction darkens. If sfGFP drops while pigment rises, that supports using mScarlet-I as the better reporter.
REACTION 8
my MelA-tyrosine cell-free system
sfGFP or mScarlet-I
| Reagent | Volume | Purpose |
|---|---|---|
| HEPES-KOH pH 7.5 | 1.50 µL | Strongly buffers against acidification over 36h. |
| CuSO4, very low concentration | 0.25 µL | Enables MelA activity. |
| L-tyrosine | 0.25 µL | Keeps substrate present but avoids overloading the system. |
This is the long-incubation preservation condition: it tests whether the best 36h outcome comes not from maximizing substrate, but from preventing reaction decay. If fluorescence and pigment both remain stronger at 36h, pH stability is the key design variable.
My actual experiments submitted
Now that I’ve seen the inferface better, I got that the goal here is to focus on DNA construct performance, so I’ll treat this as an expression/readout experiment rather than enzyme validation.
Went too far into broader bioprocess hypotheses 😅 in my brainstorm composition hypothesis above.
Given the broader objective of optimizing the cell-free master mix for maximal fluorescence across six proteins, I will test the 2 reporters:
- mScarlet-I = better reporter under melanin/dark pigment interference
- sfGFP = system health / pigment-interference control
This 1st round I will test these 8 reactions - Table Followed by textual arguments
| Reaction | Reporter | Testing | HEPES-KOH | Tyrosine | Magnesium glutamate | Water/backfill | Main purpose |
|---|---|---|---|---|---|---|---|
| 1 | mScarlet-I | Low buffer / low substrate | 0.25 µL | 0.25 µL | 0 µL | 1.50 µL | Baseline condition for mScarlet-I. |
| 2 | sfGFP | Low buffer / low substrate | 0.25 µL | 0.25 µL | 0 µL | 1.50 µL | Baseline condition for sfGFP. |
| 3 | mScarlet-I | pH drift | 1.00 µL | 0.25 µL | 0 µL | 0.75 µL | Tests whether stronger buffering improves mScarlet-I signal. |
| 4 | sfGFP | pH drift | 1.00 µL | 0.25 µL | 0 µL | 0.75 µL | Tests whether stronger buffering preserves sfGFP signal. |
| 5 | mScarlet-I | substrate limitation | 0.25 µL | 1.00 µL | 0 µL | 0.75 µL | Tests whether extra tyrosine increases pigment formation with mScarlet-I. |
| 6 | sfGFP | substrate limitation / pigment interference | 0.25 µL | 1.00 µL | 0 µL | 0.75 µL | Tests whether extra tyrosine-driven pigment formation interferes with sfGFP. |
| 7 | mScarlet-I | pH drift, substrate limitation, TX-TL capacity | 1.00 µL | 0.75 µL | 0.25 µL | 0 µL | Tests combined support for fluorescence and pigment production. |
| 8 | sfGFP | pH drift, substrate limitation, TX-TL capacity + reporter comparison | 1.00 µL | 0.75 µL | 0.25 µL | 0 µL | Same as Reaction 7, but tests sfGFP under melanin-producing conditions. |
| Reaction | Hypothesis |
|---|---|
| 1 | Low HEPES and low tyrosine will provide a baseline fluorescence condition for comparison across proteins. |
| 2 | The same low HEPES / low tyrosine condition will reveal whether sfGFP is more sensitive to pigment-related interference than mScarlet-I. |
| 3 | Increasing HEPES will improve fluorescence over 36h by reducing pH drift. |
| 4 | Increasing HEPES will help determine whether pH stabilization benefits sfGFP fluorescence under the same conditions. |
| 5 | Increasing tyrosine will test whether extra substrate/pigment formation reduces fluorescence through optical interference. |
| 6 | High tyrosine with sfGFP will test whether green fluorescence is especially affected by pigment accumulation. |
| 7 | Combining HEPES, tyrosine, and magnesium glutamate will improve fluorescence by supporting pH stability, substrate context, and TX-TL capacity. |
| 8 | The same combined condition with sfGFP will test whether translation support and buffering can preserve fluorescence despite stronger pigment-forming conditions. |
REACTION 1
Testing: Baseline condition (low buffer, low substrate)
Hypothesis: Under minimal buffering and substrate availability, both melanin production and mScarlet-I fluorescence will be limited, providing a baseline to compare improvements from other conditions.
System: melA system mScarlet-I
Supplements: HEPES-KOH → 0.25 µL Tyrosine → 0.25 µL
REACTION 2
Testing: Baseline condition + reporter comparison
Hypothesis: This condition mirrors Reaction 1 but uses sfGFP to evaluate baseline fluorescence without strong pigment production, serving as a reference for how each reporter behaves under minimal conditions.
System: melA system sfGFP
Supplements: HEPES-KOH → 0.25 µL Tyrosine → 0.25 µL
REACTION 3
Testing: pH drift
Hypothesis: Increasing buffering capacity with HEPES-KOH will improve mScarlet-I fluorescence over 36 hours by reducing pH drift, even without increasing substrate availability.
System: melA system mScarlet-I
Supplements: HEPES-KOH → 1.0 µL Tyrosine → 0.25 µL
REACTION 4
Testing: pH drift + reporter comparison
Hypothesis: This condition mirrors Reaction 3 but uses sfGFP to test whether stronger buffering preserves green fluorescence, or if signal is still affected by pigment formation and optical interference.
System: melA system sfGFP
Supplements: HEPES-KOH → 1.0 µL Tyrosine → 0.25 µL
REACTION 5
Testing: Substrate limitation
Hypothesis: Increasing tyrosine concentration will enhance melanin-like pigment production, indicating that MelA activity may be limited by substrate availability under baseline conditions.
System: melA system mScarlet-I
Supplements: HEPES-KOH → 0.25 µL Tyrosine → 1.0 µL
REACTION 6
Testing: Substrate limitation + pigment interference
Hypothesis: This condition mirrors Reaction 5 but uses sfGFP to evaluate whether increased pigment formation interferes with green fluorescence, compared to the red-shifted mScarlet-I signal.
System: melA system sfGFP
Supplements: HEPES-KOH → 0.25 µL Tyrosine → 1.0 µL
REACTION 7
Testing: pH drift, substrate limitation, and TX-TL capacity
Hypothesis: Combining buffering (HEPES-KOH), substrate availability (tyrosine), and translation support (magnesium glutamate) will help sustain melanin production and mScarlet-I fluorescence over 36 hours by addressing the main system bottlenecks.
System: melA system mScarlet-I
Supplements: HEPES-KOH → 1.0 µL Tyrosine → 0.75 µL Magnesium glutamate → 0.25 µL
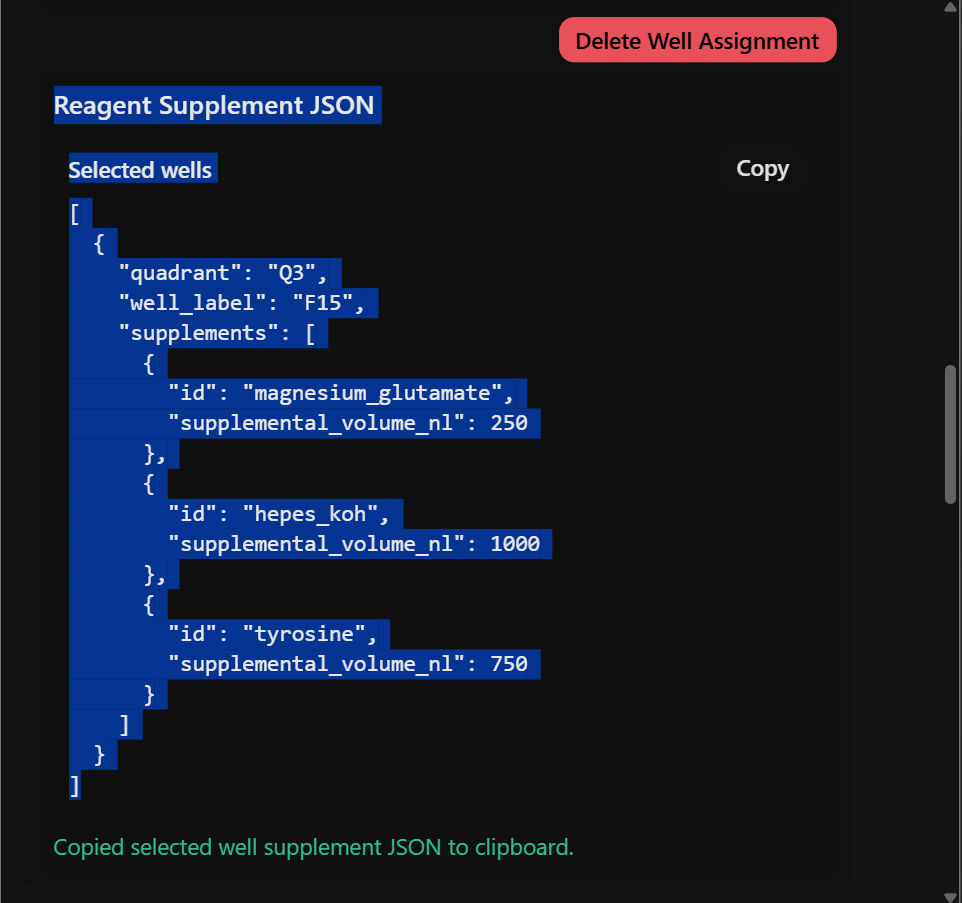
REACTION 8
Testing: pH drift, substrate limitation, TX-TL capacity + reporter comparison
Hypothesis: This condition mirrors Reaction 7 but uses sfGFP to evaluate how green fluorescence behaves under melanin-producing conditions, serving as a control to assess pigment interference relative to mScarlet-I.
System: melA system sfGFP
Supplements: HEPES-KOH → 1.0 µL Tyrosine → 0.75 µL Magnesium glutamate → 0.25 µL
Reactions submitter on 5/1/2026.
Unfortunately not possible to add copper, which is MelA tyrosinase cofactor in the form of CuSO4 now.
Keeping the designs aligned with my Part B logic I wish I could test this 5 hypothesis:
- I’m testing mScarlet-I for melanin-compatible readout
- sfGFP as the expression/pigment-interference control
- HEPES for 36h pH stability,
- Tyrosine for MelA substrate availability,
- Magnesium glutamate for TX-TL capacity.