Week 5 HW: Protein Design Part Two
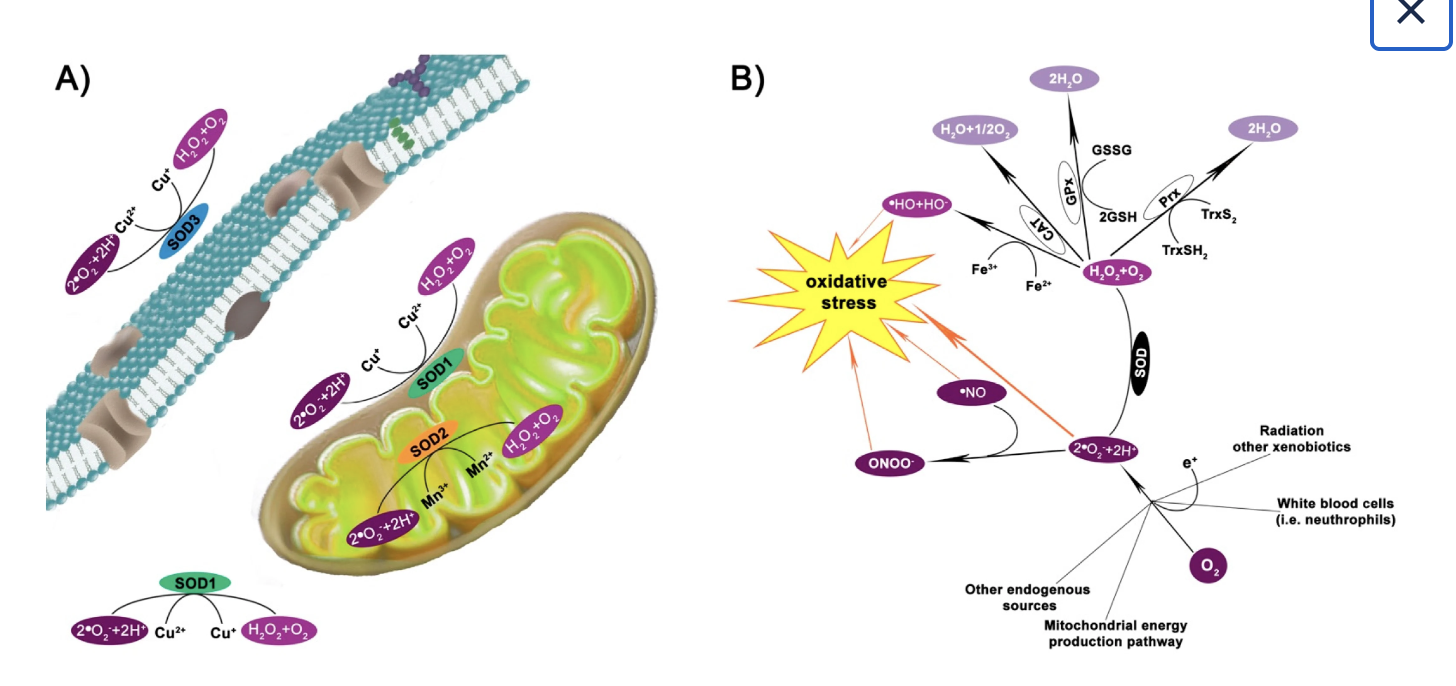
Superoxide dismutase 1 (SOD1) is a cytosolic antioxidant enzyme that converts superoxide radicals into hydrogen peroxide and oxygen. In its native state, it forms a stable homodimer and binds copper and zinc.
Mutations in SOD1 cause familial Amyotrophic Lateral Sclerosis (ALS). Among them, the A4V mutation (Alanine → Valine at residue 4) leads to one of the most aggressive forms of the disease. The mutation subtly destabilizes the N-terminus, perturbs folding energetics, and promotes toxic aggregation.
Your challenge:
Design short peptides that bind mutant SOD1. Then decide which ones are worth advancing toward therapy.
PART 1
1/ The mutant SOD1 sequence MATKVLCVLKGDGPVQGIINFEQKESNGPVKVWGSIKGLTEGLHGFHVHEFGDNTAGCTSAGPHFNPLSRKHGGPKDEERHVGDLGNVTADKDGVADVSIEDSVISLSGDHCIIGRTLVVHEKADDLGKGGNEESTKTGNAGSRLACGVIGIAQ
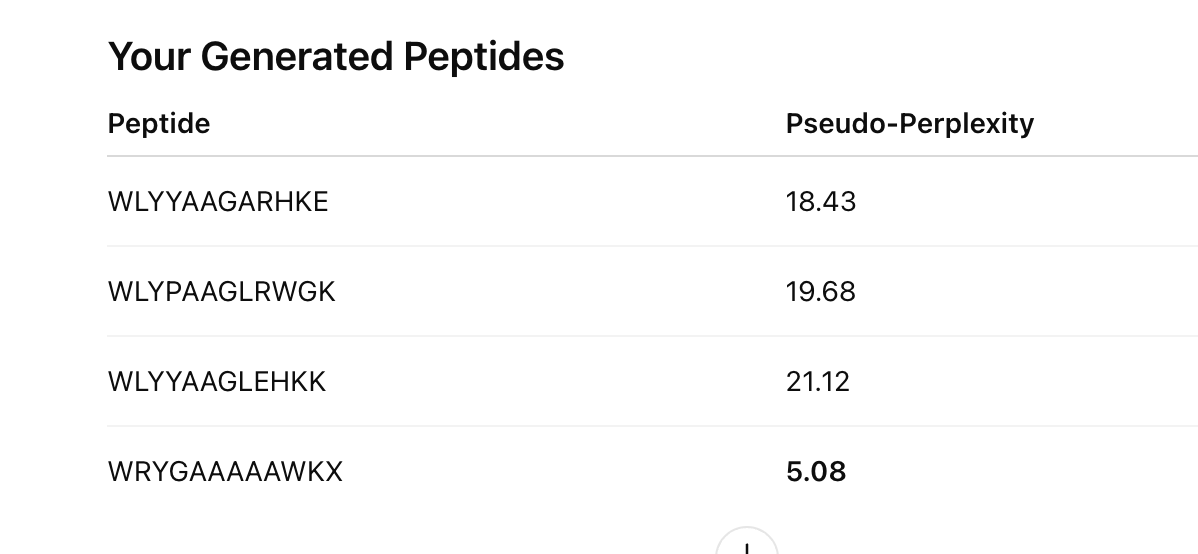
4 generated 12-aa peptides from PepMLM
the comparison peptide: FLYRWLPSRRGG
the perplexity score for each generated peptide, since the prompt says to record PepMLM confidence values
probably a small table you can later paste into your homework document

Part 2: Evaluate Binders with AlphaFold3
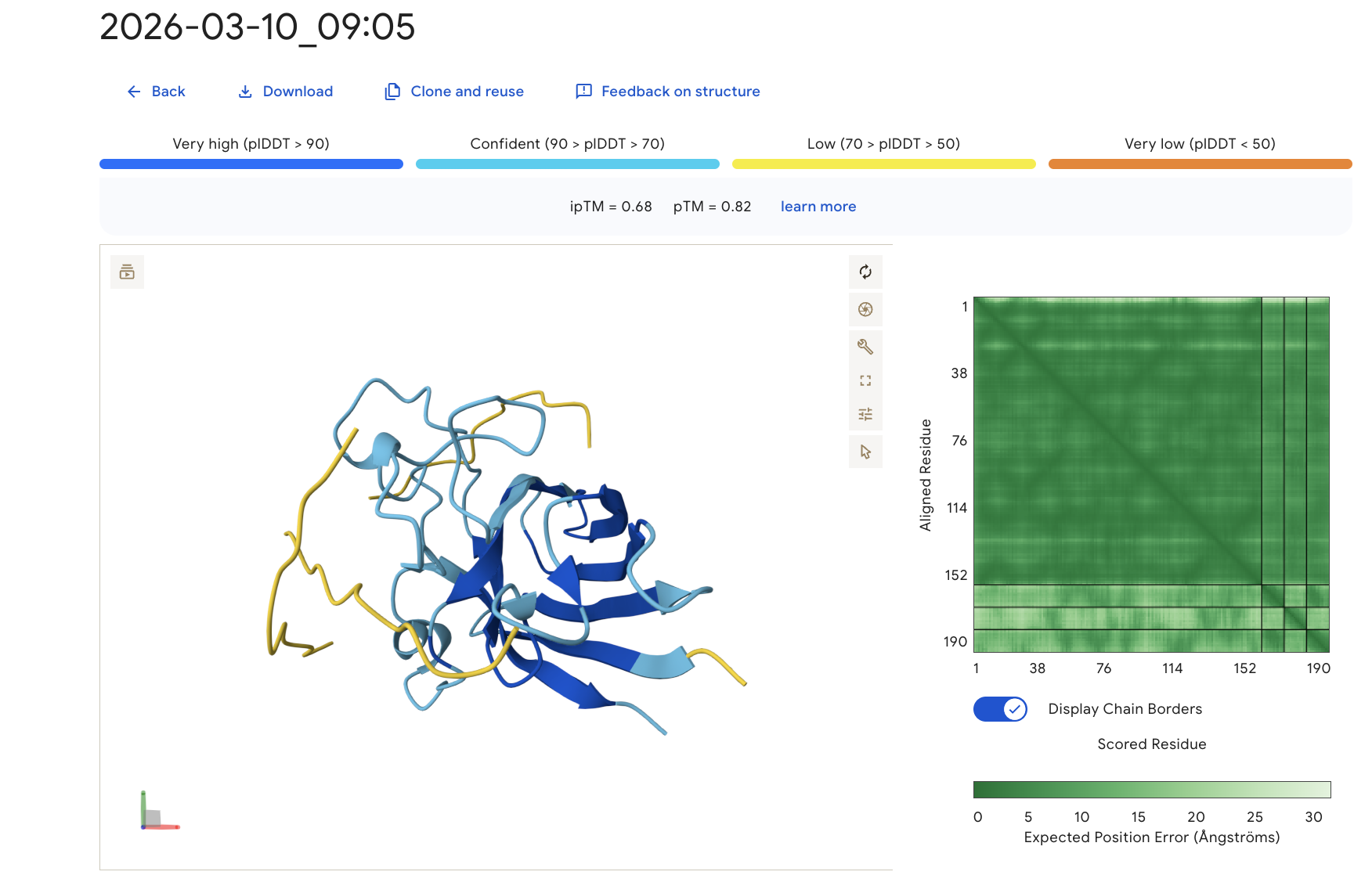
 The predicted complex produced an ipTM score of 0.68, suggesting a moderately confident interaction between the peptide and mutant SOD1. The SOD1 β-barrel core is predicted with very high confidence (dark blue), while the peptide shows lower confidence values, which is typical for short flexible peptides. The peptide appears primarily surface-associated rather than deeply buried within the protein structure.
The predicted complex produced an ipTM score of 0.68, suggesting a moderately confident interaction between the peptide and mutant SOD1. The SOD1 β-barrel core is predicted with very high confidence (dark blue), while the peptide shows lower confidence values, which is typical for short flexible peptides. The peptide appears primarily surface-associated rather than deeply buried within the protein structure.
Part 3: Evaluate Properties of Generated Peptides in the PeptiVerse (in progress) Structural confidence alone is insufficient for therapeutic development. Using PeptiVerse, let’s evaluate the therapeutic properties of your peptide! For each PepMLM-generated peptide:
Paste the peptide sequence. Paste the A4V mutant SOD1 sequence in the target field. Check the boxes Predicted binding affinity Solubility Hemolysis probability Net charge (pH 7) Molecular weight Compare these predictions to what you observed structurally with AlphaFold3. In a short paragraph, describe what you see. Do peptides with higher ipTM also show stronger predicted affinity? Are any strong binders predicted to be hemolytic or poorly soluble? Which peptide best balances predicted binding and therapeutic properties?
Choose one peptide you would advance and justify your decision briefly.
Part 4: Generate Optimized Peptides with moPPIt Now, move from sampling to controlled design. moPPIt uses Multi-Objective Guided Discrete Flow Matching (MOG-DFM) to steer peptide generation toward specific residues and optimize binding and therapeutic properties simultaneously. Unlike PepMLM, which samples plausible binders conditioned on just the target sequence, moPPIt lets you choose where you want to bind and optimize multiple objectives at once.
Open the moPPit Colab linked from the HuggingFace moPPIt model card Make a copy and switch to a GPU runtime. In the notebook: Paste your A4V mutant SOD1 sequence. Choose specific residue indices on SOD1 that you want your peptide to bind (for example, residues near position 4, the dimer interface, or another surface patch). Set peptide length to 12 amino acids. Enable motif and affinity guidance (and solubility/hemolysis guidance if available). Generate peptides. After generation, briefly describe how these moPPit peptides differ from your PepMLM peptides. How would you evaluate these peptides before advancing them to clinical studies?