Week 1 HW: Principles and Practices


First, describe a biological engineering application or tool you want to develop and why. This could be inspired by an idea for your HTGAA class project and/or something for which you are already doing in your research, or something you are just curious about.
Thyroid implant for canines
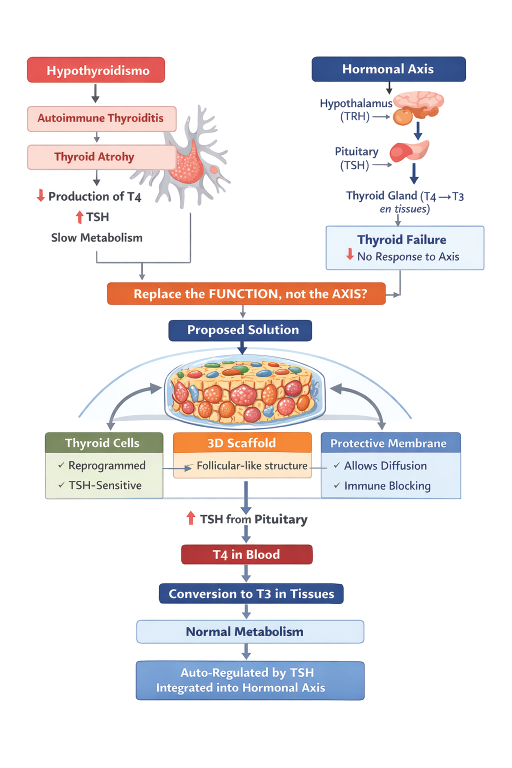
In the canine world, there is a very specific problem that owners face: hypothyroidism. It is a common condition that dogs develop around the ages of 2 or 3. Still, most of the time it is mistaken for other health conditions, such as intestinal problems, allergies, dermatological conditions, and so on. As this health condition has many impacts on the body of dogs, sometimes veterinarians can lead to a false positive.
There are three types of hypothyroidism:
- Primary
- Secondary
- Tertiary
- Congenituous
We are going to center our attention on primary hypothyroidism, caused by an immunological condition or an idiopathic problem.
The origin of Primary Hypothyroidism lies in the intercommunication that exists between the thyroid gland and the brain´s instructions. There is a malfunction that doesn´t allow the gland to produce more T4. Although the communication between the brain, the hypothalamus, and the hipophysis are fine and working at its normal pace, the gland receives the message, the gland cannot keep the production of T4 because it has been attacked by the immunological cells or it has started to transform into fat with no returning point.
The interesting thing about this system is that it is only partially broken. The T4-T3 hormones do not depend on the gland to be absorbed by the body; they depend on the genetic switches that are inside the organs they enter: liver, kidneys, brain, and muscles. Another interesting fact is that, actually, T4 hormones are passive cells and can travel around the body through the circulatory system.
In our present times, we have a pharmaceutical solution called: levotiroxin. This pill is actually a concentration ot T4 that enters the body through the stomach, and as it is absorbed, it can be delivered to the different organs to be transformed to T3. Although it is a simple solution, it is not exactly accurate all of the time, and it has to be monitored every 6 months to be adjusted according to the requirements of multiple blood tests until the dose is accurate.
Now, what if we could implant a thyroid substitute that could read the TSH directly through the blood and produce T4 as needed in the body, with a more precise response? To achieve this objective, we must face the subsequent situations:
The body needs to accept the cells as its own. The most accurate way to do this is by extracting a piece of endodermic or glandular cells from the body of the pet and reprogram it to be thyroid cells. This should be done in a lab, and the cells must reach a stable state so that they can operate accurately and not stop working when they are inside the body. The importance of having cells from the same body is that they won´t be read as a giant thread, as other materials could.
Since the body has a very acute immune system, it is necessary to put the reprogrammed cells into a container or membrane that filters immune cells and directs infiltrations. This membrane should be soft and with the right amount of pores that allow the entrance of oxygen, TSH, and nutrients, but also allow the filtration of T4 into the blood system. This membrane should be made out of biomimetic biogels, which are very hydrated and can be read as neutral elements by the immune cells.
To grow an implant, it is important to guide the cells so that they can reproduce the architecture of the thyroid gland. This could be guided by a biopolymer that suggests how cells should grow together.
Finally, this implant should be placed in the subcutaneous region so that it is surrounded by blood; therefore, TSH can reach the implant and start the reactions of production of T4.
The implant must be tested and programmed to react over a period of weeks, because the levels of T4 are regulated not in the exact moment that the TSH goes up or down, but rather when the TSH stays in this levels for the period of weeks. These slow reactions are similar to how the original gland reacts, and also, are not a red flag to immune cells. The implant would be reacting accurately and on its natural pace without the induction of not accurate dosis of T4.
Another important addition, and not a very stable one for the body, would be to integrate a nanochip that could scan and give feedback on the situation inside the body, but, for now, it is not a stable solution because the immune system would read it as a major danger that must be eliminated.
Next, describe one or more governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future, like ensuring non-malfeasance (preventing harm). Break big goals down into two or more specific sub-goals. Below is one example framework (developed in the context of synthetic genomics) you can choose to use or adapt, or you can develop your own. The example was developed to consider policy goals of ensuring safety and security, alongside other goals, like promoting constructive uses, but you could propose other goals, for example, those relating to equity or autonomy.
Prevention of physical and psychological harm during experimental stages, non-malfeasance.
- Strengthen diagnostic protocols for canine hypothyroidism before giving the individuals any medical procedure or medicine.
- Limit the access and the quantity of samples extracted to each individual.
- Establish rules and strong selection criteria for canine individuals that will be participating in the experimental stage.
Fair and free access to information
- Establish clear, accessible, and transparent information for caregivers regarding the potential health risks that the research and experimental stages pose.
- Ensure that the results and information obtained in experimental stages and results are transferred to students and professionals in specializations, with emphasis on ethical and responsible usages.
- In the event of a successful result in canines and the intention to use this research on humans, any prior required animal experimentation must be reviewed and regulated by an official regulatory authority, ensuring fairness, accountability, and ethical continuity across species.
Next, describe at least three different potential governance “actions” by considering the four aspects below (Purpose, Design, Assumptions, Risks of Failure & “Success”). Try to outline a mix of actions (e.g. a new requirement/rule, incentive, or technical strategy) pursued by different “actors” (e.g. academic researchers, companies, federal regulators, law enforcement, etc). Draw upon your existing knowledge and a little additional digging, and feel free to use analogies to other domains (e.g. 3D printing, drones, financial systems, etc.).
- Purpose: What is done now and what changes are you proposing?
- Design: What is needed to make it “work”? (including the actor(s) involved - who must opt-in, fund, approve, or implement, etc)
- Assumptions: What could you have wrong (incorrect assumptions, uncertainties)?
- Risks of Failure & “Success”: How might this fail, including any unintended consequences of the “success” of your proposed actions?
A. Review of individual cases based on the evaluation of diagnostics made by an external entity and professional
To obtain a verified hypotheroid diagnosis, there must be an external entity to review and confirm these evaluations. This review must be done by physical and chemical tests that provide consistent and standardized results. This conclusion would accept or deny the participation of each subject. For this action to be applied, collaboration between institutions and external professionals must be done. These entities and actors must be accredited and affiliated by the governement Ecuador, in this case. External evaluation must be a mandatory step before initiating any procedure, because it will ensure that participants are correctly diagnosed and that budgets, time, and assets are well used.
For these actions to happen, it is assumed that external professionals will be qualified and available to be part of the program, that diagnostic criteria will be consistent in every case, and that these reviews will reduce misdiagnosis. The risks that are on the table could be clinical disagreements between actors involved, internal vs. external, and there is a high risk of corruption among professionals and institutions, which could dramatically lower the chances of succeeding with ethical and good treatment values.
B. Unannounced auditor´s inspections regarding research developments and animals´ health and care
Monitoring research progress should be audited by some unannounced visits in order to have all the information regarding the project clear. This action would be led by an external inspector of the research institutions, as well as the research team. This would ensure the supervision of ethical practices in the lab or clinic, as well as the real progress of the experimental project. The external inspector would be accredited by external academic institutions that are renowned for their knowledge in the field and their ethical practices. It is important that this actor can have the authority to propose adjustments when needed, and to stop or allow the research to continue when the conditions are met.
This would be a successful action assuming that academic institutions would have this type of professional under their wing, and that they have the capacity to support these mechanisms of control. The risks that could arise in this policy are that frequent inspections could provoke a hostile environment, affecting the performance of researchers and, therefore, the success of the research. Administrative burdens could slow the speed of the process, and rigidity could cloud the creative and precise environment needed in this type of research.
C. Mandatory and transparency in educational programs
With mandatory education for all actors involved and transparent communication pathways ensures that experimental procedures, ethical considerations, and long-term care requirements are responsibly applied beyond the research setting. This action would be implemented by educational programs given by the academic institutions involved in this research. It would have to be open for specialized students, professionals, and caregivers who will be part of the program. Also, accessible and clear training sessions would be provided, focusing on care requirements and ethical responsibilities.
Assuming that the information is being passed with transparency and it is being understood by all the attendants, it could lead to more ethical decision-making and supervision within all the actors involved. On the other side, these actions could fail if the educational content is not accessible for all participants, making it overly technical or poorly communicated. Also, an important thing to keep in mind is that there will be an emotional and subjective matter that caregivers will experience throughout the experimentation lapse; this can lead to inadvertent or sudden dopouts putting at risk both the research process and the animal’s life.
Next, score (from 1-3 with, 1 as the best, or n/a) each of your governance actions against your rubric of policy goals. The following is one framework but feel free to make your own:
| Does the option: | Option 1 | Option 2 | Option 3 |
|---|---|---|---|
| Enhance Biosecurity | |||
| • By preventing incidents | 1 | 1 | 1 |
| • By helping respond | 3 | 2 | 2 |
| Foster Lab Safety | |||
| • By preventing incident | 2 | 1 | 1 |
| • By helping respond | 3 | 2 | 2 |
| Protect the environment | |||
| • By preventing incidents | 3 | 2 | 3 |
| • By helping respond | 3 | 2 | 3 |
| Other considerations | |||
| • Animal wellfare and intervention | 1 | 1 | 1 |
| • Diagnostic accuracy and animal inclusion | 1 | 2 | 1 |
| • Ethical inclusion and transparency | 2 | 1 | 2 |
| • Equity in access to knowledge and care | 2 | 3 | 1 |
| • Minimizing costs and burdens to stakeholders | 1 | 2 | 3 |
| • Feasibility? | 1 | 2 | 2 |
| • Not impede research | 2 | 3 | 2 |
| • Promote constructive applications | 3 | 1 | 1 |
Last, drawing upon this scoring, describe which governance option, or combination of options, you would prioritize, and why. Outline any trade-offs you considered as well as assumptions and uncertainties. For this, you can choose one or more relevant audiences for your recommendation, which could range from the very local (e.g. to MIT leadership or Cambridge Mayoral Office) to the national (e.g. to President Biden or the head of a Federal Agency) to the international (e.g. to the United Nations Office of the Secretary-General, or the leadership of a multinational firm or industry consortia). These could also be one of the “actor” groups in your matrix.
The fundamental governance option to be prioritized is “Review of individual cases based on the evaluation of diagnostics made by an external entity and professional”. As it acts on the foundations of the experimental phases, it can prevent misdiagnosis or weakly supported clinical assumptions before the animal is in the trial. This option reduces the risk of ethical procedures, minimizes harm, protects animal welfare, and strengthens the scientific validity of the research foundation. The trade-off to be considered here is that this extra evaluation may slow down, at the beginning, and limit the number of participants, as well as the development of the research, but in the future are preventive in many aspects.
As complementary governance options are Option n.2 and Option n.3. These actions ensure accountability within all the actors involved by making knowledge a regulatory and independent system. The prevent the misuse of information and empowers informed decision-making throughout the research cycle. External regulatory actors are also essential because they can verify if clinical, laboratory, and welfare standards are applied before, during, and after experimentation. The trade-off here is that research institutions might perceive these actors as intruders and as potential uncertainty symbols of the research development. But, on the other hand, the combination of early prevention, continuous oversight, and broad education creates a distributed responsibility, which reduces the ethical failures to go unnoticed or unaddressed.
Reflecting on what you learned and did in class this week, outline any ethical concerns that arose, especially any that were new to you. Then propose any governance actions you think might be appropriate to address those issues. This should be included on your class page for this week.
As an experimental research that needs to be done in live animals, specifically canines, it is important to keep very clear the limit between ethical procedures and unethical ones. The fact that we do not communicate in the same way might be an important barrier between researchers and subjects. They might end up seen as numbers rather than brave and important living beings who are giving their lives to the experiment. It has arisen to me the question whether the experiment is worth the many health discomforts that the research individual will undergo to gain a major medical advancement?
Another question to keep in mind is whether we could develop a nanoscanner able to read the gland’s condition and avoid any invasive procedure, preventing from misdiagnostics and giving a much more accurate reading of the situation inside the animal´s body.
Some bibliography found about thyroid organoids for humans:
Kariyawasam, D., Stoupa, A., Nguyen Quoc, A., Pimentel Dantas, I., Polak, M., & Carré, A. (2025). From stem cells to organoids in thyroid: Useful tools or a step for cell therapy? La Presse Médicale, 54(4), 104301. https://doi.org/10.1016/j.lpm.2025.104301
ZHANG, Y., FU, M., WANG, H., & SUN, H. (2023). Advances in the Construction and Application of Thyroid Organoids. Physiological Research, 72(5), 557–564. https://doi.org/10.33549/physiolres.935102