Week 6: Genetic Circuits - part I
Assignment: DNA Assembly
Answer these questions about the protocol in this week’s lab:
- What are some components in the Phusion High-Fidelity PCR Master Mix, and what is their purpose?
Elements:
- Pyrococcus-like enzyme fused with a processivity-enhancing domain that increases fidelity and speed.
- 5
3polymerase activity, 35exonuclease activity, and generates blunt-ended products. - Phusion DNA Polymerase, deoxynucleotides, and reaction buffer that has been optimized and includes MgCl2
- Requirements to be added: template, primers, and water.
Purpose:
- DNA needs to be correct after amplification
- Cloning and long or difficult amplicons
- What are some factors that determine primer annealing temperature during PCR?
Annealing temperature:
- The temperature used during the primer annealing step of a PCR, which depends on the primer melting temperature
- Usually lower than the melting temperature
- Too high Ta: the primer will not anneal to the template, and the amplification will not proceed
- Too low Ta: nonspecific binding of the primers to the template or each other can occur, causing:
- increased likelihood of nonspecific product formation
- decreased formation of the intended product due to inefficient reaction conditions.
Factors of primer annealing temperature:
- Base composition (proportion of ATCG nucleotides)
- Primer concentration
- Ionic reaction environment
- Two methods from this class create linear DNA fragments: PCR and restriction enzyme digestion. Compare and contrast these two methods, both in terms of protocol and in terms of when one may be preferable to the other.
PROTOCOLS PCR vs RESTRICTION ENZYME DIGESTION:
PCR is the technique for mainly DNA amplification.
RESTRICTION ENZYME DIGESTION is the technique for cutting specific sections of DNA.
Both act in different temperatures and procedures, but the final applications are varied: for PCR, there is DNA amplification, cloning, sequencing, etc, while for Restriction enzyme digestion, there are DNA-specific cutting, cloning, etc.
In the table below, the comparison between protocols can be better understood:
- How can you ensure that the DNA sequences that you have digested and PCR-ed will be appropriate for Gibson cloning?
- How does the plasmid DNA enter the E. coli cells during transformation?
- Describe another assembly method in detail (such as Golden Gate Assembly)
- Explain the other method in 5 - 7 sentences plus diagrams (either handmade or online).
- Model this assembly method with Benchling or Asimov Kernel!
Assignment: Asimov Kernel
- Create a Repository for your work
- Create a blank Notebook entry to document the homework and save it to that Repository
- Explore the devices in the Bacterial Demos Repo to understand how the parts work together by running the Simulator on various examples, following the instructions for the simulator found in the “Info” panel (click the “i” icon on the right to open the Info panel)
- Create a blank Construct and save it to your Repository
- Recreate the Repressilator in that empty Construct by using parts from the Characterized Bacterial Parts repository
- Search the parts using the Search function in the right menu
- Drag and drop the parts into the Construct
- Confirm it works as expected by running the Simulator (“play” button) and compare your results with the Repressilator Construct found in the Bacterial Demos repository
- Document all of this work in your Notebook entry - you can copy the glyph image and the simulator graphs, and paste them into your Notebook
- Build three of your own Constructs using the parts in the Characterized Bacterials Parts Repo
- Explain in the Notebook Entry how you think each of the Constructs should function
- Run the simulator and share your results in the Notebook Entry
- If the results don’t match your expectations, speculate on why and see if you can adjust the simulator settings to get the expected outcome
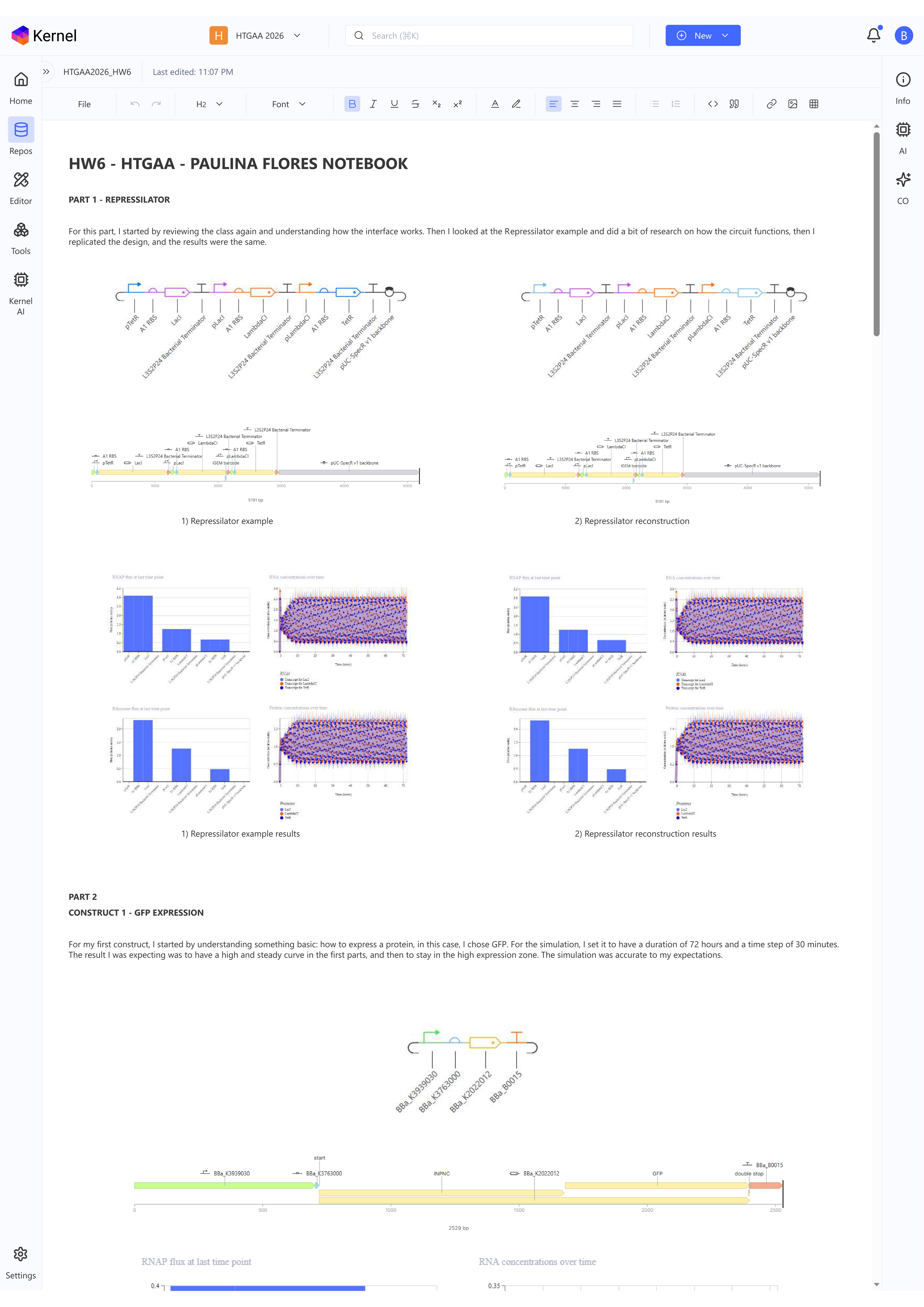
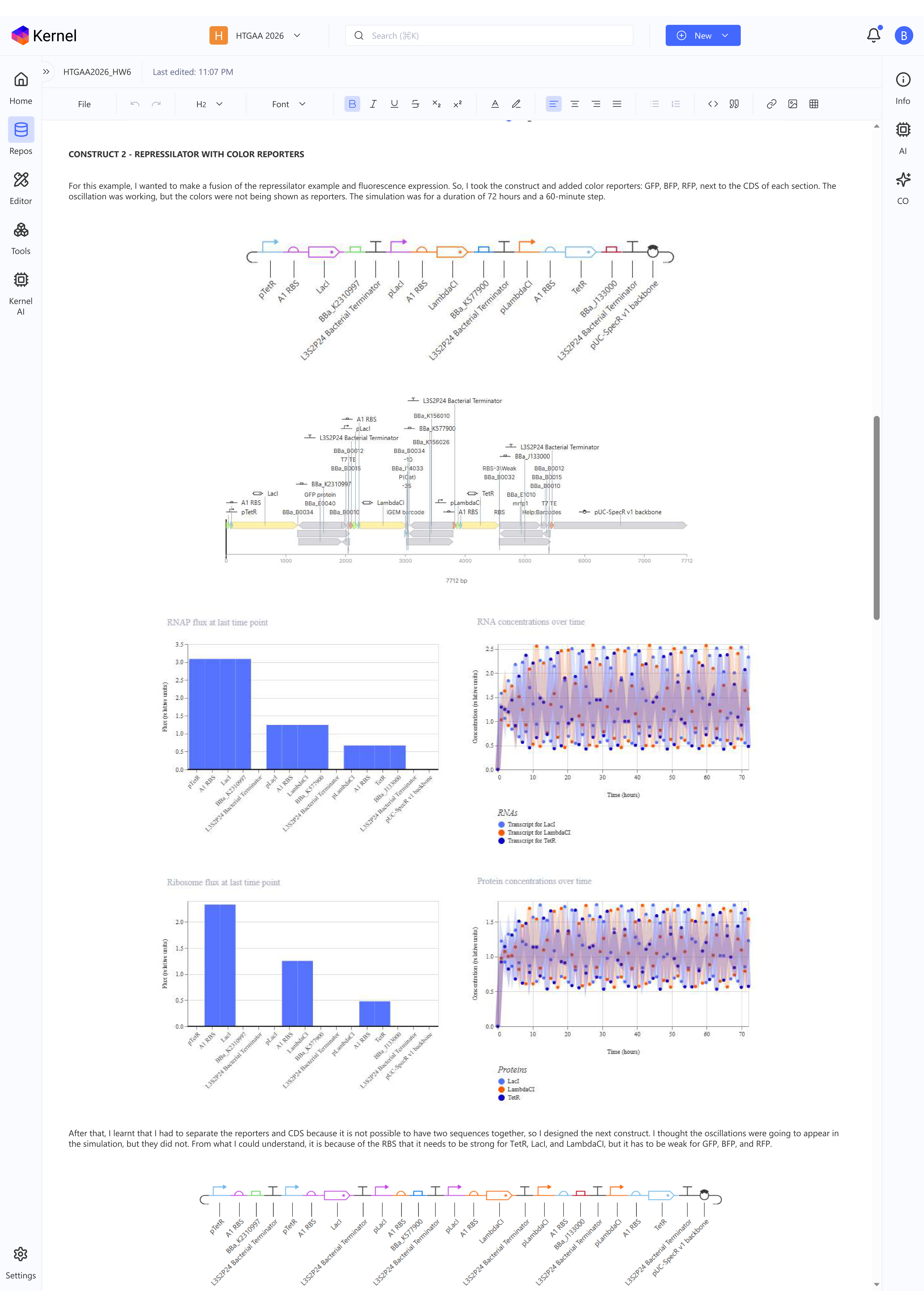
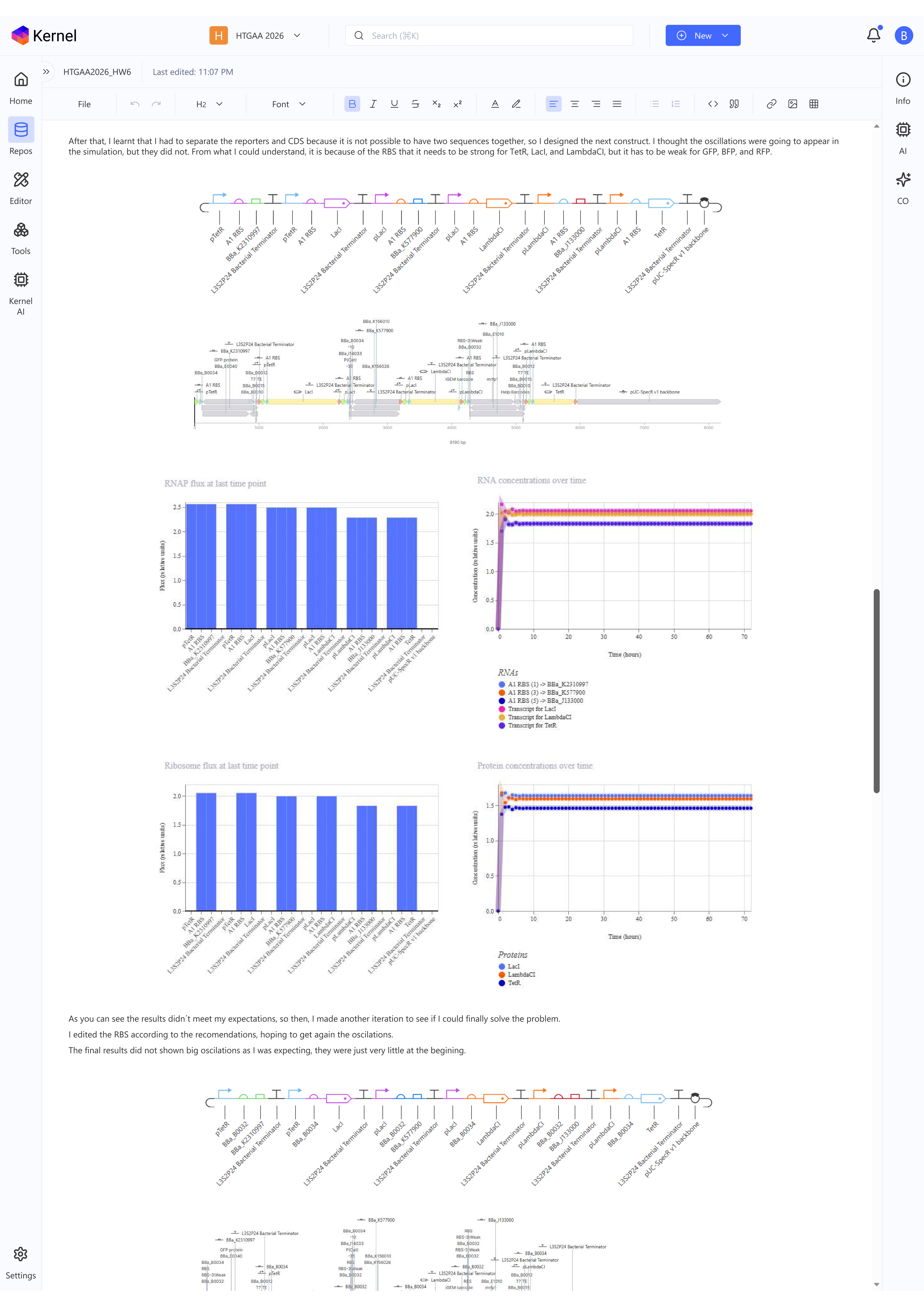
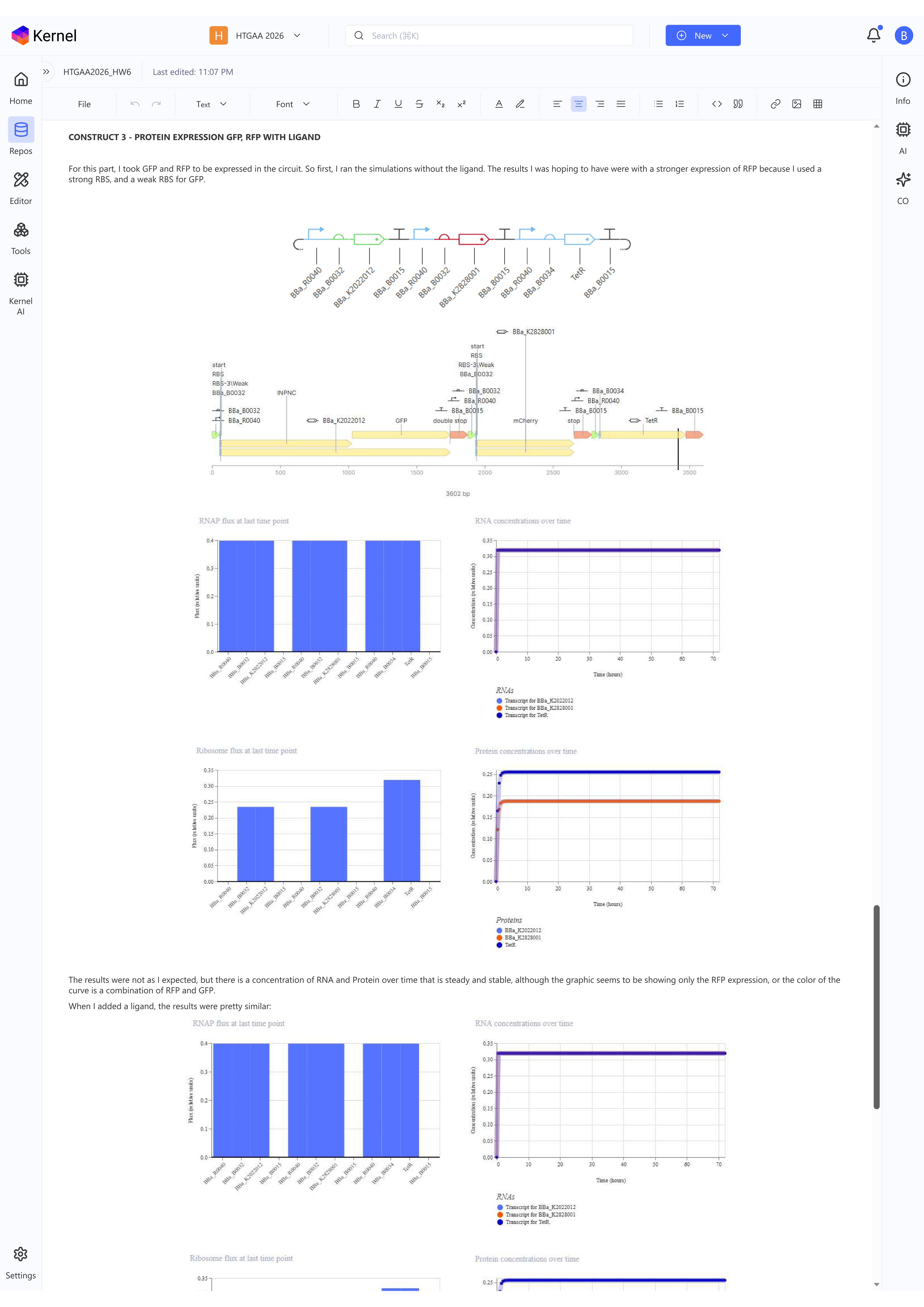
For this assignment, I took notes in the Notebook as suggested. Here are the images of it, in which I registered every step:
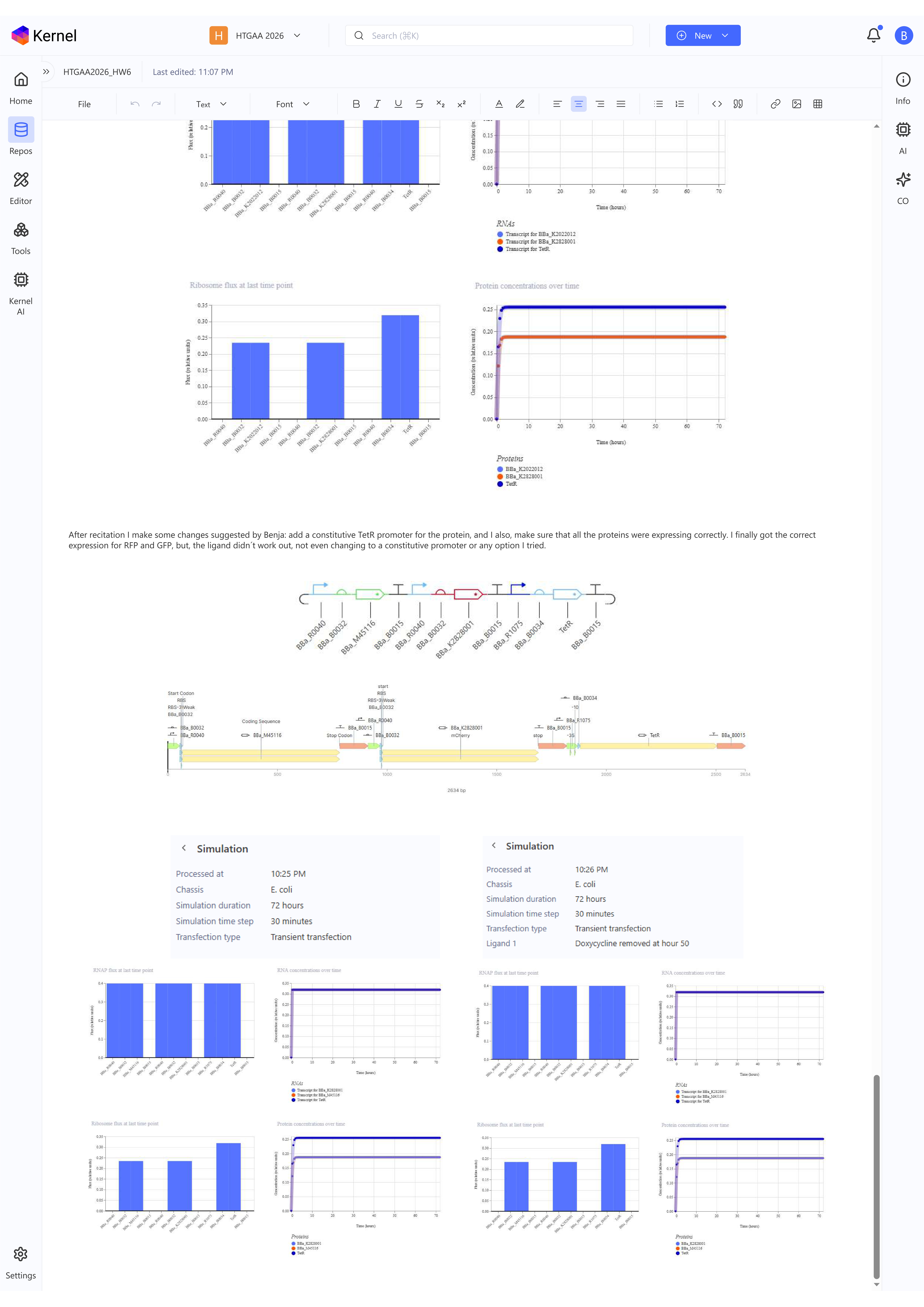
For the first part: represillator construct, the process was smooth, as well as the results compared to the reference. For the second part of constructs 1, 2, and 3. I designed a simple protein expression of GFP; the results were in a steady state. After this construct, I tried a repressilator using GFP, BFP, and RFP, the first simulation was pretty good, but I noticed that fluorescent proteins were not expressing, so I made a few changes. The result I obtained was not what I was expecting because the oscillations did not appear as steady as I thought they were going to. Instead, I obtained a few oscillations at the beginning of the simulations, and the expression was steady. For the last construct, I tried to use GFP and RFP with a TetR protein and a Ligand. The results of the simulation without the ligand were successful after a few iterations. Although the ligand did not work out, I tried many options.