Week 1 HW: Principles and Practices
1. Early diagnostics trough microfluidics
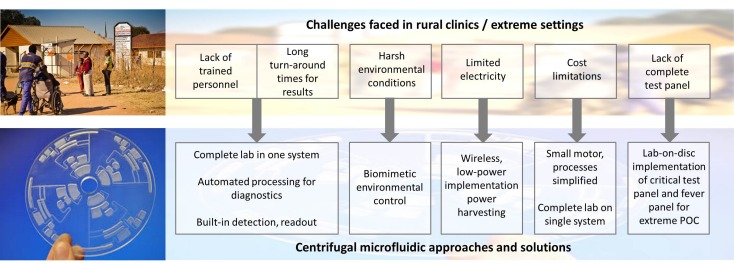
In current medical practice, prevention and early intervention are among the most effective strategies for improving health outcomes. However, in underserved regions such as the Peruvian Amazon, limited access to primary healthcare often leads to an accumulation of disease burden. Patients frequently seek medical attention only when symptoms have progressed to advanced or critical stages. In the region of Loreto, Perú, for example, widespread dependence on untreated river water exposes communities to persistent threats from pathogens, which can contribute to chronic malnutrition in children under five. Additionally, in remote and rural areas, the advanced laboratory infrastructure is scarce. In these contexts, delayed diagnosis is not just an inconvenience—it can be the difference between early treatment and a life-threatening outcome. Fast diagnostics save time, resources, and, most importantly, lives.


Motivated by this reality, my project focuses on developing field-deployable diagnostic systems using centrifugal microfluidics, also known as Lab-on-a-Disc technology. This approach has the potential to bring essential diagnostic capabilities directly to communities that are far from centralized laboratories. A single disc can be designed to perform a critical panel of tests, including diagnostics for malaria, tuberculosis (TB), hepatitis, and waterborne pathogens, enabling earlier intervention for diseases that significantly impact maternal and child mortality.
To make diagnostics viable in extreme and resource-limited environments, this platform leverages centrifugal pseudo-forces to replace bulky and expensive laboratory equipment. By simply rotating a disc, the system can function as a centrifuge, mixer, and mechanical lyser within a compact format. Importantly, this design is well suited for regions with unreliable electricity, as it requires only a small spindle motor and can even be powered wirelessly using standards such as Qi induction charging.

For molecular detection, the platform can integrate Loop-Mediated Isothermal Amplification (LAMP), a technique capable of amplifying detectable DNA products in under 40 minutes at a constant temperature. This eliminates the need for complex thermal cycling and significantly reduces system complexity. Finally, when combined with smartphone-based imaging and analysis, the Lab-on-a-Disc platform offers a sensitive, specific, user-friendly, rapid, and practical pathway so that communities often left behind by modern healthcare systems are no longer excluded from timely and lifesaving diagnostics.
References
Smith, S., Mager, D., Perebikovsky, A., Shamloo, E., Kinahan, D., Mishra, R., Torres Delgado, S. M., Kido, H., Saha, S., Ducrée, J., Madou, M., Land, K., & Korvink, J. G. (2016). CD-Based Microfluidics for Primary Care in Extreme Point-of-Care Settings. Micromachines, 7(2), 22. https://doi.org/10.3390/mi7020022
Wu, C., Cheng, J., Zhang, Y., & Yao, P. (2025). A LAMP Detection System Based on a Microfluidic Chip for Pyricularia grisea. Sensors, 25(8), 2511. https://doi.org/10.3390/s25082511
Zhang, J., Fu, Y., Fong, C. Y., Hua, H., Li, W., & Khoo, B. L. (2025). Advancements in microfluidic technology for rapid bacterial detection and inflammation-driven diseases. Lab on a Chip, 25(16), 3348–3375. https://doi.org/10.1039/d4lc00795f
2. Governance and Policy Goals
The Lab-on-a-Disc diagnostic platform is designed for deployment in remote, high-biodiversity regions such as the Peruvian Amazon, where healthcare infrastructure is limited and ecological and cultural contexts differ significantly from those of centralized laboratories. Governance goals must therefore address not only safety and performance but also the ecological, social, and interpretive risks that may arise when advanced diagnostic technologies are introduced into sensitive environments.
To guide safe and responsible use, five refined governance goals are aligned with the four overarching principles: Goals 1 and 2 support Safety and Non-Malfeasance; Goal 3 supports Responsible Use; Goal 4 supports Trust and Transparency; and Goal 5 supports Equity and Access. This hierarchy shows how each goal translates broader principles into practical guidance.
Governance Goal 1: Prevent Clinical and Interpretive Harm in Low-Resource Settings
In environments where diagnostic results may directly influence treatment decisions without access to confirmatory testing, incorrect or misunderstood results can cause immediate harm.
Sub-goals:
- Reduce the risk of false positives or negatives leading to inappropriate treatment, delayed care, or unnecessary referral.
- Ensure results are interpretable by non-specialist users, minimizing reliance on implicit technical knowledge.
Governance Goal 2: Minimize Ecological and Biological Spillover Risks
Deploying molecular diagnostics in regions of high biodiversity raises concerns beyond human health, including accidental environmental exposure and biological contamination.
Sub-goals:
- Prevent the release of amplified DNA, reagents, or biological samples into surrounding ecosystems.
- Ensure safe containment and disposal of consumables in settings without formal biohazard waste infrastructure.
- Avoid unintended ecological interactions, particularly in environments with poorly characterized microbial diversity.
Governance Goal 3: Prevent Misuse and Function Creep in Field Diagnostics
Even benign diagnostic tools can be repurposed or extended beyond their original intent, especially when deployed outside formal regulatory oversight.
Sub-goals:
- Limit use of the platform to predefined diagnostic assays, preventing unauthorized adaptation for unvalidated testing.
- Reduce the risk of informal surveillance, coercive testing, or testing without informed consent.
- Prevent the normalization of diagnostic data collection without appropriate ethical or institutional oversight.
Governance Goal 4: Ensure Cultural Legitimacy and Trust in Local Contexts
Introducing unfamiliar technologies into marginalized communities can generate distrust, resistance, or misuse if local values and perceptions are not respected.
Sub-goals:
- Avoid deployment models that frame the technology as an external or extractive intervention.
- Ensure diagnostic practices align with local understandings of illness, care, and authority.
- Support community engagement and transparency to prevent fear, stigma, or misinformation surrounding testing.
Governance Goal 5: Enable Equitable and Sustainable Access Without Dependency
Governance must ensure that access to diagnostics does not create long-term dependency on external suppliers or short-lived pilot programs.
Sub-goals:
- Prevent reliance on proprietary consumables that cannot be locally sourced or substituted.
- Support maintenance, repair, and adaptation at the local level.
- Ensure that withdrawal of the technology does not leave communities worse off than before deployment.
References
National Academies of Sciences, Engineering, and Medicine. (2006). Globalization, biosecurity, and the future of the life sciences. National Academies Press.
United Nations Office for Disarmament Affairs. (2022). Global guidance framework for the responsible use of the life sciences: Mitigating biorisks and governing dual-use research. United Nations.
3. Governance Actions
Based on the governance goals outlined above, the following actions propose concrete interventions at different points in the development and deployment of the Lab-on-a-Disc diagnostic platform. Each action targets a specific failure mode while preserving the technology’s ability to deliver public health benefits in remote, high-biodiversity settings.
Governance Action 1: Assay Locking and Use-Restriction by Design
Actors: Device developers, academic laboratories, manufacturing partners
Purpose
At present, microfluidic diagnostic platforms are often designed to be modular and adaptable. While this flexibility supports innovation, it also enables unvalidated or inappropriate use in the field. This action proposes restricting field-deployed devices to a predefined set of validated diagnostic assays.
Design
- Embed physical or firmware-based constraints that limit the disc and reagents to specific, pre-approved assays (e.g., malaria, TB, hepatitis).
- Require laboratory-level modification or authorization to expand assay capability.
- Maintain an open design at the research stage, but enforce restrictions at the deployment stage, similar to how drones limit flight modes or altitudes for consumer users.
Assumptions
- That misuse or function creep is more likely in the field than in controlled research environments.
- That technical restrictions can meaningfully reduce misuse without preventing legitimate diagnostics.
Risks of Failure & Success
- Restrictions may reduce adaptability to emerging pathogens.
- Overly rigid controls could push users toward unofficial modifications, increasing risk.
- Success could unintentionally slow emergency responses where rapid assay adaptation is needed.
Governance Action 2: Context-Specific Training and Certification for Field Use
Actors NGOs, public health agencies, academic partners, local health authorities
Purpose
Currently, diagnostic tools are often deployed with minimal user training, assuming intuitive use. This action introduces a requirement for short, context-specific certification before field deployment.
Design
- Develop localized training modules addressing sample handling, result interpretation, waste disposal, and cultural sensitivity.
- Certification tied to device access, similar to operator licensing for drones or medical imaging equipment.
- Training delivered in collaboration with local institutions to ensure cultural relevance.
Assumptions
- That misuse and misinterpretation are more likely due to lack of contextual understanding than malicious intent.
- That short, targeted training can significantly reduce error rates.
Risks of Failure & Success
- Certification requirements could slow deployment in urgent situations.
- Training programs may become symbolic rather than effective if poorly implemented.
- Successful certification systems may unintentionally exclude informal healthcare providers who play key roles in remote regions.
Governance Action 3: Embedded Waste Containment and Disposal Protocols
Actors: Device designers, manufacturers, environmental health regulators
Purpose
Standard biohazard disposal infrastructure is often absent in remote regions, increasing the risk of environmental contamination. This action proposes technical containment as a governance mechanism.
Design
- Design disposable discs with sealed reaction chambers that remain closed after use.
- Incorporate visual indicators confirming successful containment.
- Pair deployment with simple, low-tech disposal guidance suitable for rural settings, analogous to sharps containers in vaccination campaigns.
Assumptions
- That ecological spillover risks are driven more by disposal practices than by the diagnostic process itself.
- That technical containment can compensate for limited waste infrastructure.
Risks of Failure & Success
- Increased manufacturing complexity and cost.
- Improper disposal may still occur despite containment.
- Widespread success could increase plastic waste if sustainability is not addressed in parallel.
Governance Action 4: Community Engagement and Consent-Based Deployment Models
Actors: NGOs, local governments, public health organizations, community leaders
Purpose
Introducing unfamiliar diagnostic technologies without community engagement risks mistrust, resistance, or misuse. This action formalizes community involvement as a governance requirement rather than an optional add-on.
Design
- Require community-level consultation before deployment.
- Co-develop communication strategies explaining what the diagnostics can and cannot do.
- Integrate informed consent practices adapted to local cultural norms, similar to participatory models used in environmental monitoring.
Assumptions
- That cultural misalignment, rather than technical failure, is a major barrier to adoption.
- That trust increases correct and ethical use.
Risks of Failure & Success
- Engagement processes may slow deployment timelines.
- Community leaders may not represent all stakeholders.
- Successful engagement could create expectations of long-term support that exceed project resources.
4. Scoring of Governance Options
Scale: 1 = best, 3 = weakest, n/a = not applicable
| Criteria | Assay Locking | Training & Certification | Waste Containment | Community Engagement |
|---|---|---|---|---|
| Enhance Biosecurity | ||||
| • By preventing incidents | 1 | 2 | n/a | 2 |
| • By helping respond | 3 | 2 | n/a | 2 |
| Foster Lab / Field Safety | ||||
| • By preventing incidents | 2 | 1 | 1 | 2 |
| • By helping respond | 3 | 2 | 2 | 2 |
| Protect the environment | ||||
| • By preventing incidents | n/a | 2 | 1 | 2 |
| • By helping respond | n/a | 3 | 2 | 2 |
| Other considerations | ||||
| • Minimizing costs and burdens | 2 | 2 | 3 | 2 |
| • Feasibility | 2 | 1 | 2 | 2 |
| • Not impede research | 3 | 2 | 1 | 1 |
| • Promote constructive applications | 2 | 1 | 2 | 1 |
5. Governance and Prioritization Recommendations
A layered governance approach focused on Training and Certification, Embedded Waste Containment, and Community Engagement and Consent-Based Deployment should be prioritized, according to the above scoring. The most common failure modes in low-resource environments—misinterpretation of results, inappropriate handling of biological samples, and an excessive dependence on automated diagnostics—are directly addressed by training and certification. Deployment in high-biodiversity environments, where ecological spillover risks are significant and traditional biohazard disposal infrastructure is lacking, requires embedded waste containment. By ensuring that the technology is not viewed as an extractive or external intervention, community engagement promotes long-term sustainability, appropriate use, and trust. Despite offering robust protection against abuse, Assay Locking was not given priority as a primary mechanism because it may restrict adaptability in responding to emerging health threats. However, it remains a valuable complementary measure in high-risk or highly decentralized deployment contexts.
Uncertainties, Assumptions, and Trade-offs
Since training and community involvement may slow initial rollout, this approach accepts trade-offs between deployment speed and long-term legitimacy. Additionally, it makes the assumption that there is enough institutional capacity to support community consultation and certification programs. Political continuity, long-term funding, and scalability beyond pilot deployments are still uncertain, which emphasizes the need for adaptable, multi-layered governance as opposed to relying solely on one control mechanism.
In order to address health issues that stem from structural inequality and ecological vulnerability, this recommendation is mainly addressed to the Peruvian government, in particular public health and environmental authorities, as well as the United Nations, whose role in norm-setting, coordination, and support is crucial.


Reflection: Ethical Considerations and Governance Lessons from Week 1
Reflecting on this week’s lectures and discussions in How to Grow (Almost) Anything, one ethical concern that became particularly salient to me was the idea that engineering solutions can generate harm not through malicious intent but through misalignment with social, cultural, or ecological contexts. Prior to this week, I primarily associated ethical risk in biotechnology with dual-use or deliberate misuse. What was new to me was how easily harm can emerge from well-intentioned technologies when governance is treated as an afterthought rather than a design requirement.
In the context of my project, this raised concerns about how diagnostic technologies might alter decision-making in communities with limited access to follow-up care or how biological waste and amplified DNA could unintentionally interact with ecosystems characterized by high and poorly understood biodiversity. I also became more aware of the ethical implications of trust and authority: who is believed when a diagnostic result is produced and how that authority is perceived when the technology originates outside the community.
To address these concerns, I believe governance actions must extend beyond technical safeguards. Context-specific training, community engagement, and clear communication about the limitations of diagnostic tools are as important as analytical accuracy. Embedding governance into the lifecycle of the technology—from design to deployment—helps ensure that innovation supports, rather than overrides, local knowledge and autonomy.
This week reinforced for me that ethical biotechnology is not defined solely by what a technology can do, but by how, where, and for whom it is used, and that governance is a critical component of responsible bioengineering practice.