Week 1 HW: Principles and Practices
ARM-Net: Alzheimer’s Recovery Micro-TNT Network
1.Biological Engineering Application
I am developing ARM-Net, a synthetic biology tool designed to overexpress CCT4 specifically in microglia. Unlike the full TRiC/CCT complex, CCT4 can function independently or as homo-oligomers to promote microtubule synthesis. By boosting these chaperonins, I aim to induce the formation of Tunneling Nanotubes (TNTs) between microglia, creating an “Intercellular Care Network” (ICN)
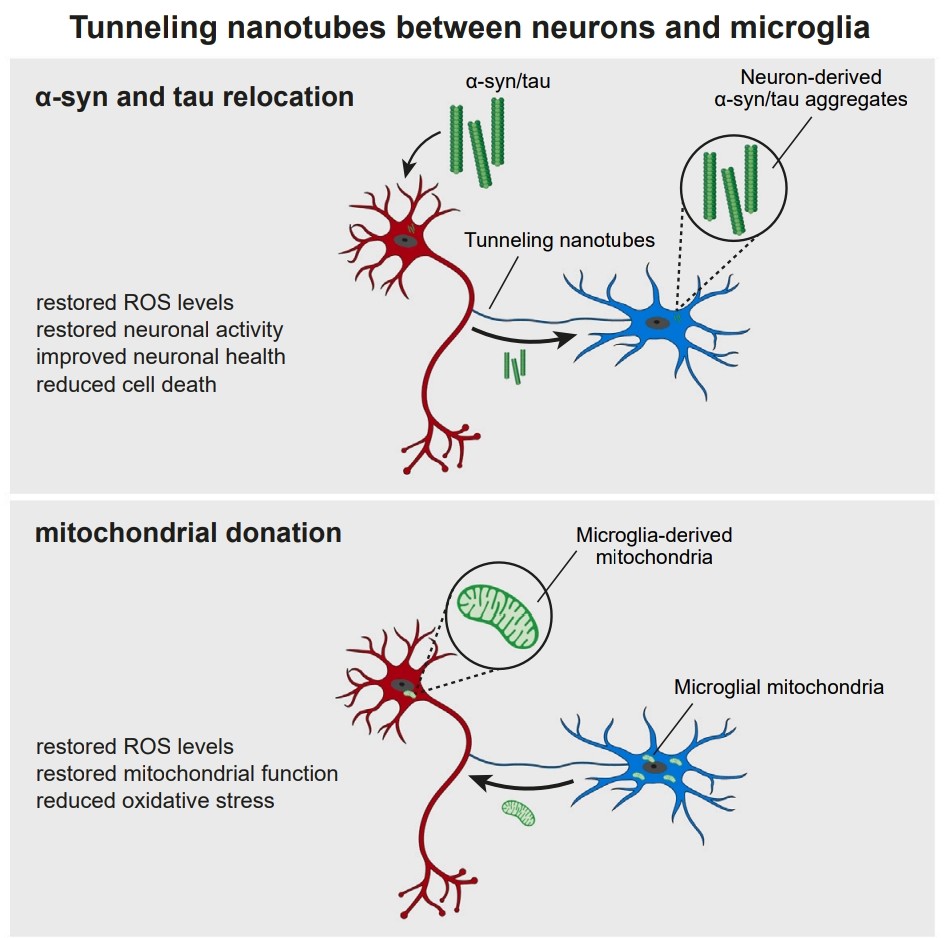
 Source: Nuntaprut et al. (2024), “Microglia rescue neurons from aggregate-induced neuronal dysfunction,” Science.
Source: Nuntaprut et al. (2024), “Microglia rescue neurons from aggregate-induced neuronal dysfunction,” Science.
Why: Current Alzheimer’s Disease (AD) treatments focus on slowing progression. ARM-Net aims for recovery. By networking microglia, we can distribute the overwhelming proteotoxic load (Amyloid-β,Tau) across a cellular collective for efficient degradation and allow healthy cells to “rescue” damaged ones by transferring mitochondria and resources via TNTs.
2. Governance and Policy Goals
Primary Goal: Ensuring Non-malfeasance and Biological Integrity
•The overarching goal is to ensure that the induction of TNTs (Tunneling Nanotubes) via CCT4 overexpression promotes brain recovery without introducing new pathological risks or irreversible side effects.
Sub-goal A: Prevention of Pathological Propagation
While TNTs are designed to facilitate the clearance of toxic proteins like Amyloid-β and Tau, they must not inadvertently act as a conduit for the spread of these very same pathogens or viral vectors across the brain.
•Actionable Metric:
Implement molecular “checkpoints” or filtering mechanisms within the TNT structure to ensure one-way or cargo-specific transport.
•Risk Mitigation:
Preventing spread of neurodegenerative diseases through the engineered network.
Sub-goal B: Safeguarding Cognitive Identity and Reversibility
Ensuring that the physical modification of the brain’s immune network does not alter the patient’s fundamental personality, memories, or self.
•Actionable Metric:
Integration of an inducible kill-switch or degradation domain that can dissolve the TNT network if behavioral side effects are observed.
•Ethical Standard:
Prioritizing the preservation of the brain’s natural cell death cycles. The tool must avoid creating a permanent “hyper-connected state” to maintain the ecological homeostasis of the neural environment.
3. Governance Actions
| Aspect | Action 1: Molecular Filtering (Technical) | Action 2: Phased Implementation & Kill-switches (Rule) | Action 3: Open-sour Safety Registry (Incentive) |
|---|---|---|---|
| Purpose | Implement a molecular “gate” to allow only authorized cargo through TNTs. | Require mandatory inducible “kill-switches” to dissolve the network if side effects occur. | Create a shared database of “failed” network architectures to prevent repeating risks. |
| Design | Researchers engineer CCT4 with cargo-specific filtering domains. | Federal Regulators (FDA/PMDA) mandate reversibility as a condition for clinical trials. | International Consortia provide funding bonuses for sharing negative safety data. |
| Assumptions | Assumes molecular gates won’t clog or block essential resource sharing. | Assumes the small-molecule trigger can reach all networked cells in the brain. | Assumes companies will prioritize collective safety over proprietary secrecy. |
| Risks | Failure: Evolution of “cloaked” pathogens. Success: High cost of customized filters. | Failure: Kill-switch mutation. Success: Slower innovation due to strict safety layers. | Failure: Poor data quality. Success: A “monopoly” on safety standards by large firms. |
4.Scoring Table
| Does the option: | Option 1 | Option 2 | Option 3 |
|---|---|---|---|
| Enhance Biosecurity | |||
| • By preventing incidents | 1 | 2 | 1 |
| • By helping respond | 3 | 1 | 2 |
| Foster Lab Safety | |||
| • By preventing incident | 1 | 2 | 2 |
| • By helping respond | 3 | 1 | 2 |
| Protect the environment | |||
| • By preventing incidents | 3 | n/a | 2 |
| • By helping respond | n/a | 2 | 1 |
| Other considerations | |||
| • Minimizing costs and burdens to stakeholders | 2 | 2 | 3 |
| • Feasibility? | 2 | 1 | 2 |
| • Not impede research | 3 | 3 | 1 |
| • Promote constructive applications | 1 | 2 | 1 |
5. Prioritization and Recommendation
Priority 1: Elimination of Pathogenic Propagation Risks
The absolute highest priority is ensuring that TNTs do not facilitate the spread of viruses or neurotoxic proteins. Unless this fundamental danger is completely eliminated through rigorous design, this project cannot even reach the starting line of clinical development. We must guarantee that our engineered “care network” does not inadvertently become a “superhighway” for the very pathology it aims to treat.
Priority 2: Maintenance of Biological Homeostasis
Clearing the requirement for systemic homeostasis is also a paramount necessity. The brain is a delicate ecosystem; any intervention that induces hyper-connectivity must not disrupt the natural metabolic balance, synaptic pruning, or the healthy turnover of cells.
Recommendation
prioritize a combination of Action 1 (Molecular Filtering) and Action 2 (Pulsatile Activation) as the primary strategy to address these critical hurdles.
•Technical Strategy:
By engineering CCT4 with cargo-specific filtering domains, we provide a physical barrier against pathogen spread.
•Regulatory Standard:
I recommend that national health agencies and the International Neuroethics Society mandate “Temporal Reversibility” as a non-negotiable standard. By ensuring the network is only active in “pulses,” we allow the brain to return to its natural homeostatic state between treatment cycles.