Subsections of Robert Feldman — HTGAA Spring 2026
Homework
Weekly homework submissions:
Week 1 HW: Principles and Practices
Tattoos are a popular form of body art, but a limitation is that they are permanent. I propose engineered microbial bio-tattoos. Human skin is populated with trillions of microbes. Engineering skin dwelling microbes to express skin-safe pigments and endow two dimensional multicellular organizations would allow for long-term body art that could be edited, moved, resized, or outright removed with a targeted topical anti-microbial agent. Living tattoos would be a considerable advancement of an ancient form of self-expression. Infection and toxicity risk Engineered microbes are a pathogenic risk. In standard tattooing, pigment is injected into the dermis and compartmentalized primarily by macrophages. If engineered microbes and the associated pigments need to be internalized by human cells, this presents risk of infection or toxicity.
Week 2 HW: DNA Read Write and Edit
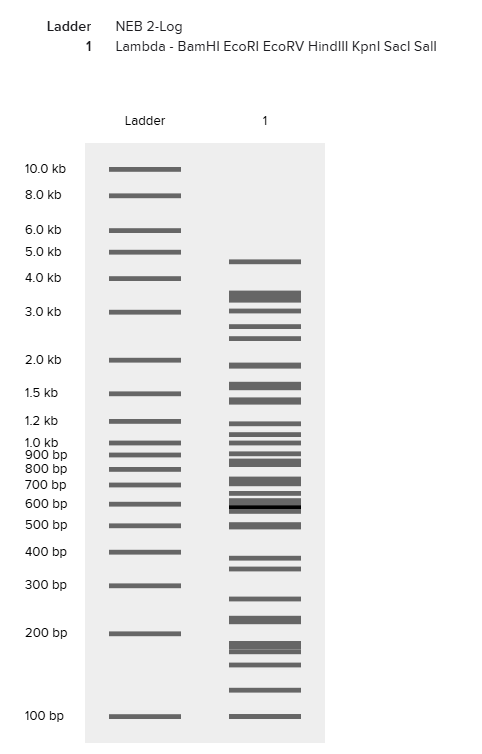
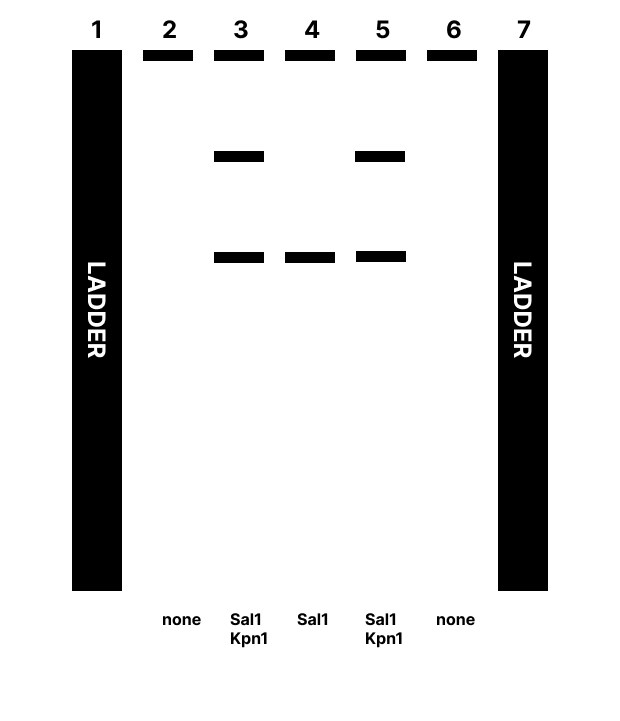
In silico digests Digestion with restriction enzymes yields DNA fragments of varied size. Running gel electrophoresis with a selection of enzymes can produce artwork. DNA design challenge 3.1) I have selected YugO, a bacterial potassium channel involved in metabolically mediated electrical signaling in B. subtilis. I am interested in this protein as it relates to my work exploring engineered electrical signaling behaviors in bacteria. Per UniProt, the sequence for this protein is : MKSNRIFISWLRWPLFIRIGVIILCLILLFGQIIYILEPKQFTSVFEGIWWAVVTVSTVGYGDYVPHTPLGQAAGILLILSGASFVTAYFATLSAAAFSRQHRYIEGKVAYKGRDHIILIGWNEKTNRLLKDLQLAAPSKTVVLIDESLTEGPLIENVHFIRGHAADDGTLKRANITEAESVMITADQYKSETDADMLSVLTLLSVKGLNPLAYCIVEILTDRFVTNAERAGANQIIGTSEFISRAMLQHYQVKLRPSKQQNGIKLTLDQHVELLAVPDELKGAAYKTCVLYFLDHNTTIIGIQKKEGPMLSPPLTYKVLETDQFLAI
Subsections of Homework
Week 1 HW: Principles and Practices
Tattoos are a popular form of body art, but a limitation is that they are permanent. I propose engineered microbial bio-tattoos. Human skin is populated with trillions of microbes. Engineering skin dwelling microbes to express skin-safe pigments and endow two dimensional multicellular organizations would allow for long-term body art that could be edited, moved, resized, or outright removed with a targeted topical anti-microbial agent. Living tattoos would be a considerable advancement of an ancient form of self-expression.
Infection and toxicity risk Engineered microbes are a pathogenic risk. In standard tattooing, pigment is injected into the dermis and compartmentalized primarily by macrophages. If engineered microbes and the associated pigments need to be internalized by human cells, this presents risk of infection or toxicity.
Effect on existing microbial communities Balanced microbial communities are essential for healthy skin. Careful consideration of community-wide effects are required to ensure bio-body art does not cause dysbiosis and potential bodily harm.
Safe administration practices Tattoo artists are already required to undergo considerable training to safely tattoo. New body art paradigms will require the development of extensive bio-safety training practices to protect patients, and tattoo shop “labs” will need to be regulated to limit unintended environmental exposures, similar to any research lab that generates potential biohazards.
Governance
- Eliminate pathogen, immunogenic, and pigment toxicity risk
Purpose - Engineered microbial tattoos could infect healthy skin, or be contagious via skin to skin contact. Introduction of new foreign organisms could induce an immune response and lead to undesirable or dangerous autoimmune/allergic reactions. The pigment generated by engineered microbes could be toxic to healthy skin. These risks will be considered and addressed to limit health concerns.
Design - Microbes will be engineered with specific sensitivies to a targeted topical/systemic antimicrobial that can be used to eliminate synthetic tattoo microbes. A non pathogenic strain will be chosen, and any pathogenic mechanisms will be deactivated (unless pathogenicity is necessary for effective tattooing???) Only nontoxic pigments will be selected, analogous to those currently used in tattooing practice, and expression levels will be carefully modulated via engineering strategies, including promotoer strength, inducible systems, etc. Implementing these approaches may involve the CDC, FDA, and local and state health departments.
Risks - In convential tattooing ink is injected into the dermis and internalized by cells. Do microbial tattoos need to be pathogenic to be functional? Can they produce pigments that are internalized by cells, and consistently replenish pigments to ensure tattoo longevity? What if microbes aquire pathogenicity naturally after injection into the skin? What if foreign microbes/pigments cause autoimmunity or cancer?
- Minimize effects on existing skin-dwelling microbial communities, control proliferation and distribution
Purpose - The aim of this governance is to limit skin microbiome dysbiosis and imbalance. This includes strategies for selective distribution and pigment deposition, controlling growth rates to avoid microbial overgrowth, and considering effects on the skin ecosystem to protect healthy skin function.
Design - Existing skin microbes may be chosen as chasis to avoid introducing new species. A species with an appropriate proliferation rate can be chosen, or this rate could be engineered. Ecosystem modeling dynamics, in vitro experiments, and in vivo experiments in appropriate human skin models will be conducted to evaluate risks. Again, the CDC, FDA, and local health departments would be involved.
Risks - Even with careful ecological considerations, engineered microbes might acquire mutations that allow them to outcompete existing commensal/mutualistic strains. Would changes in environments or seasonality (ie wet vs dry climates) effect communities? Do people with different ethnicities/skin pigment expressions react differently? What if someone changes their soap/the fabrics they wear/etc? Given the limited understanding around the role of the microbiome in human health, will existing knowledge allow us to fully understand the longterm effects of introducing engineered species?
- Develop bio-safety training for bio-tattoo artists and regulate tattoo shop facilities
Purpose - Tattoo artists and shops are already heavily regulated to ensure safe tattooing. The advent of engineered microbial tattoos would require significant new training and regulation. Artists must be trained to handle potenitally biohazardous samples and introduce them to human subjects. Shops facilities must be built similar to bio-labs so environemental risks are minimal.
Design - Tattoo artists already undergo apprenticeships followed by licensing and blood-borne pathogen training. This would be expanded with the inclusion of biosafety training, synthetic/recombinant nucleic acids training, etc. Biology backgrounds for artists would be desirable. Shops have to follow strict policies on sterilization, instrument preparation, etc. Shops would need to be outfitted with lab equipment and environmental controls including potentially biosafety cabinets, incubators, etc. This regulation would largely fall to local and state health organizations, and other actors include individual shops, artist gilds, universities and organizations which might create training/certificate programs, etc.
Risks - Improper regulation, training, and environmental control could result in health risks for patients, pathogen outbreak risk, etc. Unlicensed tattoo artists already exist, this would just be adding an additional layer of risk to this.
| Does the option: | Option 1 | Option 2 | Option 3 |
|---|---|---|---|
| Enhance Biosecurity | |||
| • By preventing incidents | 1 | 3 | 2 |
| • By helping respond | 2 | 3 | 1 |
| Foster Lab Safety | |||
| • By preventing incident | 2 | 3 | 1 |
| • By helping respond | 2 | 3 | 1 |
| Protect the environment | |||
| • By preventing incidents | 2 | 3 | 1 |
| • By helping respond | 1 | 3 | 2 |
Prioritized governance - Eliminate pathogen, immunogenic, and pigment toxicity risk
Potential pathogenicity of engineered microbes introduced to the human skin presents such a high risk level that this governance action is paramount to successful technology development. Pathogens must not cause direct health risk to patients, including immunogenicity and risk of contagious transfer by skin-to-skin contact. While ecosystem wide effects and safe administration practices are also signifcant ny other governance considerations pale in comparison to this essential primary goal. The CDC and local and state level health agencies would be involved in any determinations of biohazard risk. The FDA is currently responsible for regulation safe pigments used in tattooing, and would also be involved.
Week 2 Lecture Prep
Professor Jacobson
- Nature’s machinery for copying DNA is called polymerase. What is the error rate of polymerase? How does this compare to the length of the human genome. How does biology deal with that discrepancy?
The error rate is 1:1x10^6. The length of the human genome is ~3 billion base pairs. Thus, biology has developed proofreading and repair systems to address errors.
- How many different ways are there to code (DNA nucleotide code) for an average human protein? In practice what are some of the reasons that all of these different codes don’t work to code for the protein of interest?
Dr. LeProust
- What’s the most commonly used method for oligo synthesis currently?
Phosphoramidite DNA Synthesis
Why is it difficult to make oligos longer than 200nt via direct synthesis?
Why can’t you make a 2000bp gene via direct oligo synthesis?
George Church
- What are the 10 essential amino acids in all animals and how does this affect your view of the “Lysine Contingency”?
The 10 essential ammino acids are Cys, His, Ile, Leu, Lys, Met, Phe, Thr, Trp, Tyr, and Val. Lysine is an essential amino acid, so the lysine contingency would work unless the dinos could find lycine in their diet. This makes me realize that controlling synthetic organisms is very hard.
Citations
Hou Y, Wu G. Nutritionally Essential Amino Acids. Adv Nutr. 2018;9(6):849-51. doi: 10.1093/advances/nmy054. PubMed PMID: 30239556; PMCID: PMC6247364.
Week 2 HW: DNA Read Write and Edit
In silico digests
Digestion with restriction enzymes yields DNA fragments of varied size. Running gel electrophoresis with a selection of enzymes can produce artwork.
DNA design challenge
3.1) I have selected YugO, a bacterial potassium channel involved in metabolically mediated electrical signaling in B. subtilis. I am interested in this protein as it relates to my work exploring engineered electrical signaling behaviors in bacteria. Per UniProt, the sequence for this protein is : MKSNRIFISWLRWPLFIRIGVIILCLILLFGQIIYILEPKQFTSVFEGIWWAVVTVSTVGYGDYVPHTPLGQAAGILLILSGASFVTAYFATLSAAAFSRQHRYIEGKVAYKGRDHIILIGWNEKTNRLLKDLQLAAPSKTVVLIDESLTEGPLIENVHFIRGHAADDGTLKRANITEAESVMITADQYKSETDADMLSVLTLLSVKGLNPLAYCIVEILTDRFVTNAERAGANQIIGTSEFISRAMLQHYQVKLRPSKQQNGIKLTLDQHVELLAVPDELKGAAYKTCVLYFLDHNTTIIGIQKKEGPMLSPPLTYKVLETDQFLAI
3.2) The nucleotide sequence for this protein is:
atgaaaagcaaccgcatttttattagctggctgcgctggccgctgtttattcgcattggc
gtgattattctgtgcctgattctgctgtttggccagattatttatattctggaaccgaaa
cagtttaccagcgtgtttgaaggcatttggtgggcggtggtgaccgtgagcaccgtgggc
tatggcgattatgtgccgcataccccgctgggccaggcggcgggcattctgctgattctg
agcggcgcgagctttgtgaccgcgtattttgcgaccctgagcgcggcggcgtttagccgc
cagcatcgctatattgaaggcaaagtggcgtataaaggccgcgatcatattattctgatt
ggctggaacgaaaaaaccaaccgcctgctgaaagatctgcagctggcggcgccgagcaaa
accgtggtgctgattgatgaaagcctgaccgaaggcccgctgattgaaaacgtgcatttt
attcgcggccatgcggcggatgatggcaccctgaaacgcgcgaacattaccgaagcggaa
agcgtgatgattaccgcggatcagtataaaagcgaaaccgatgcggatatgctgagcgtg
ctgaccctgctgagcgtgaaaggcctgaacccgctggcgtattgcattgtggaaattctg
accgatcgctttgtgaccaacgcggaacgcgcgggcgcgaaccagattattggcaccagc
gaatttattagccgcgcgatgctgcagcattatcaggtgaaactgcgcccgagcaaacag
cagaacggcattaaactgaccctggatcagcatgtggaactgctggcggtgccggatgaa
ctgaaaggcgcggcgtataaaacctgcgtgctgtattttctggatcataacaccaccatt
attggcattcagaaaaaagaaggcccgatgctgagcccgccgctgacctataaagtgctg
gaaaccgatcagtttctggcgatt
3.3) I need to optimize codon usage depending on the organism I will be expressing this protein in, because different organisms have different codon preferences. I have selected B. subtilis 168 as this is the organism I am working with in the lab.
Codon optimized sequence:
ATG AAA TCA AAC AGA ATC TTC ATC TCT TGG TTG CGT TGG CCG CTG TTT ATT CGG ATT GGA GTG ATC ATC CTG TGC CTG ATA TTG CTC TTT GGA CAA ATT ATC TAT ATC TTG GAG CCG AAA CAG TTT ACA AGC GTT TTC GAG GGG ATT TGG TGG GCC GTT GTC ACA GTG TCA ACA GTC GGG TAC GGG GAC TAT GTC CCT CAT ACC CCT TTA GGA CAG GCG GCC GGC ATT TTA CTT ATA CTT AGC GGG GCG TCA TTT GTG ACA GCA TAT TTC GCG ACG CTG TCT GCG GCT GCT TTC AGT CGG CAG CAT CGC TAC ATT GAA GGA AAA GTC GCA TAT AAG GGC AGA GAT CAT ATC ATC CTC ATA GGA TGG AAT GAG AAA ACC AAT CGG TTA CTT AAA GAC CTT CAA TTA GCG GCT CCT AGT AAA ACA GTA GTG CTT ATC GAT GAA TCA CTG ACG GAA GGC CCA CTG ATC GAG AAC GTG CAT TTC ATC CGG GGC CAT GCG GCG GAT GAT GGC ACG CTC AAA CGG GCG AAC ATT ACG GAA GCG GAG AGC GTT ATG ATT ACA GCT GAT CAG TAC AAA TCA GAA ACA GAT GCC GAT ATG CTT AGC GTA CTG ACA TTA CTC TCA GTG AAA GGC CTT AAT CCG TTA GCA TAC TGT ATT GTC GAG ATT CTC ACC GAT AGA TTT GTG ACG AAC GCC GAG AGA GCG GGC GCC AAC CAG ATT ATC GGC ACA TCT GAA TTT ATT TCC CGC GCT ATG CTT CAG CAT TAC CAA GTT AAA CTG CGT CCG AGC AAA CAA CAG AAC GGC ATT AAG TTG ACG CTG GAC CAA CAC GTT GAA TTA TTA GCG GTT CCG GAT GAA TTA AAA GGG GCG GCG TAT AAG ACG TGT GTC CTG TAT TTC CTC GAT CAT AAT ACA ACT ATT ATT GGG ATT CAA AAA AAA GAG GGT CCT ATG TTG AGC CCG CCA TTA ACC TAT AAA GTG TTG GAA ACA GAT CAA TTT CTT GCT ATT
3.4) In this case I am intending to express this as a functional protein in a living community (this particular sequence is already native to B. subtilis 168, I am intending to include chimeric components), thus a cell dependent approach will be taken. This sequence can be synthesized, cloned into a plasmid using enzyme digestion and Gibson assembly, and transformed in competent B. subtilis 168.
Twist DNA
5.1 DNA Read
i)
ii)
5.2 DNA Write
i)
ii)
5.3 DNA Edit
i)
ii)