Week 7 HW : Genetic Circuits Part II: Neuromorphic Circuits
PART 1: INTRACELLULAR ARTIFICIAL NEURAL NETWORKS
- Advantages of IANNs over Boolean Genetic Circuits
Intracellular Artificial Neural Networks (IANNs) provide key advantages over traditional Boolean logic-based genetic circuits. Conventional circuits produce discrete input-output relationships, limited to simple ON or OFF states, which restricts their information-processing power. In contrast, IANNs operate in an analog fashion, using molecular concentrations as continuous variables. This allows cells to generate nuanced, graded responses to fluctuating biological inputs.
These strengths make IANNs far more adaptable for handling the inherent complexity of biological systems, which rarely follow binary patterns. They also boost computational capacity inside cells by integrating multiple inputs at once, much like weighted summation in artificial neural networks. As a result, IANNs can support sophisticated functions, such as classifying biological signals or making multi-input decisions.
More specifically, IANNs offer:
• Continuous (analog) outputs, rather than just binary ON/OFF states
• Greater computational power through multi-input integration
• Robustness to biological noise, without relying on sharp thresholds
• Potential for multilayer network architectures
• More adaptive biological decision-making
Overall, IANNs offer a more realistic and advanced way to mimic how cells naturally process information.
- Applications of IANNs
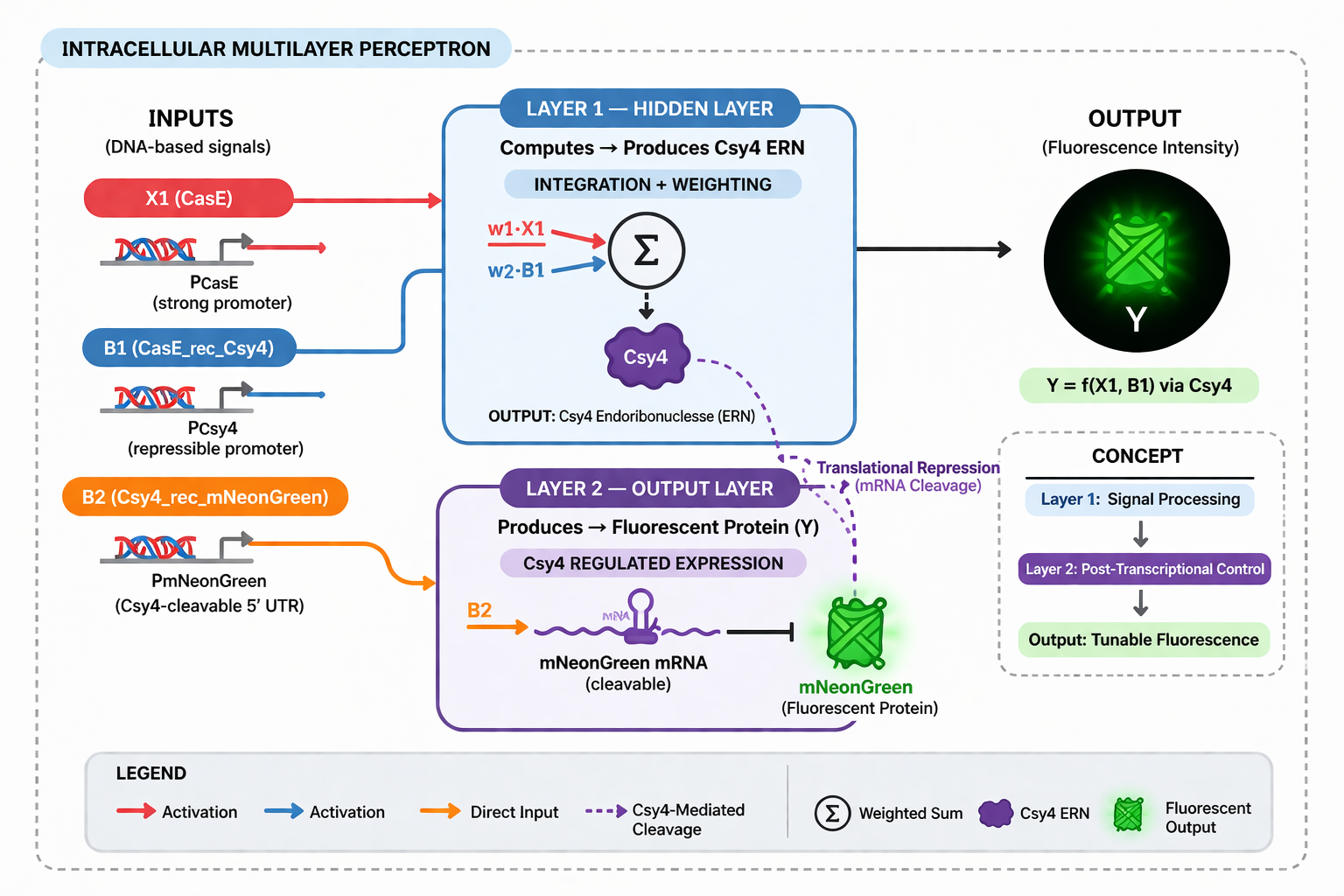
One of the most promising uses for Intracellular Artificial Neural Networks (IANNs) is as smart biosensors for detecting complex diseases like cancer, which often involve multiple biomarkers. In such a system, engineered cells take in various biological inputs—for instance, two protein biomarkers (X1 and X2)—each weighted according to its relevance to the disease state. The cell then integrates these signals through molecular mechanisms resembling a perceptron: contributions from each input are summed and passed through a biological activation function. If the integrated result exceeds a threshold, the cell triggers an output, such as:
• Expression of a fluorescent protein for detection
• Activation of an apoptosis pathway to eliminate target cells
This approach outshines traditional biosensors, which detect only single biomarkers, by recognizing more specific, multifaceted disease patterns. That said, deploying IANNs in biological systems comes with challenges, including:
• Biological noise that disrupts signal stability
• Limited dynamic range in gene expression
• Metabolic burden from extra components
• Cross-talk between regulatory pathways
• Difficulty in precisely tuning biological weights
- Diagram Intracellular Multilayer Perceptron

PART 2: FUNGAL MATERIALS
- Examples, Uses, Advantages, and Disadvantages of Fungal Materials
Fungal materials represent an innovative class of biomaterials made by growing fungal mycelium into sustainable, eco-friendly products. These have already found applications across industries, including:
• Mycelium-based packaging as a styrofoam alternative
• Synthetic mycelium leather for fashion
• Fungal bricks or construction panels
Producers cultivate mycelium on organic substrates like agricultural waste, forming dense, customizable structures. Compared to traditional materials, fungal options offer standout benefits. They’re fully biodegradable, avoiding environmental pollution, and require far less energy since they’re “grown” rather than manufactured through energy-intensive processes. Plus, they repurpose organic waste, aligning perfectly with circular economy principles.
Still, challenges remain:
• Lower mechanical strength than synthetic counterparts
• Sensitivity to humidity and environmental conditions
• Variability in outcomes due to the biological nature of fungi
• Longer production times versus conventional manufacturing
- Genetic Engineering in Fungi and Its Advantages
I am interested in developing environmentally responsive mycelium materials (smart responsive mycelium), living substances that sense changes around them and respond functionally. Using synthetic biology, we can engineer fungi to express specific genes in reaction to targeted stimuli, like pollutants, humidity shifts, or mechanical stress.
This engineering aims to produce active materials that don’t just sit passively but “sense” and “react,” opening doors to applications such as:
• Environmental biosensors (detecting pollutants or toxins)
• Adaptive construction materials (responding to humidity or cracks)
• Biomaterial-based monitoring systems (visual indicators of conditions)
Unlike conventional materials, these integrate biological functions directly into their structure, adding substantial value. Compared to bacteria, fungi offer distinct advantages for synthetic biology-driven biomaterials. As multicellular organisms, they naturally form three-dimensional mycelial networks, yielding solid, structured materials without extra manufacturing steps. Bacteria, being mostly unicellular, excel at molecule production but fall short for building bulk materials. Fungi also boast advanced protein secretion and thrive on cheap lignocellulosic waste substrates, making them more efficient and sustainable.
Challenges include slower growth rates and trickier genetic manipulation than bacteria. Yet for living, responsive, and adaptive materials, fungi shine by seamlessly blending biological functions with physical form.