Week 7 HW: Genetic Circuits Part II: Neuromorphic Circuits
Assignment Part 1: Intracellular Artificial Neural Networks (IANNs)
1. What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions?
| Characteristic | Intracellular Artificial Neural Networks | Traditional Genetic Circuits (that use Boolean functions) |
|---|---|---|
| Input-output mapping | Continuous logic that can sum multiple inputs with determined importance or “weights”. This allows for classification of complex patterns. | Discrete simple logic (AND, OR, NAND) with ON/OFF behaviors. |
| Vulnerability to noise | Since they rely on graded responses, they can average across inputs. This makes them less vulnerable to change output when exposed to noise. | Sensitive to noise around thresholds. If there are small fluctuations the ON/OFF gate can be flipped. |
| Decision-making | They classify inputs into categories at once and produce signals to different “effector modules” (also called “winner-take-all decisions” in mammalian cells, as mentioned in Chen et al., 2024). This also allows for higher adaptive behavior. | They often produce a single binary output per circuit. This makes them less adaptable. |
Table created using information taken from:
Chen, Z., Linton, J. M., Xia, S., Fan, X., Yu, D., Wang, J., … & Elowitz, M. B. (2024). A synthetic protein-level neural network in mammalian cells. Science, 386(6727), 1243-1250.
Gentili PL, Stano P. Chemical Neural Networks Inside Synthetic Cells? A Proposal for Their Realization and Modeling. Front Bioeng Biotechnol. 2022 Jun 6;10:927110. doi: 10.3389/fbioe.2022.927110. PMID: 35733531; PMCID: PMC9208290.
2. Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
Microbial biosensors to detect mercury in water already exist and have been successful at being an eco-friendly and cost-effective alternative to other methods (Zevallos-Aliaga et al., 2024 & Roointan et al., 2015). They tend to use MerR transcription factor and its cognate promoter (Pmer) to drive a fluorescent or luminescent reporter in response to Hg 2+ (Zevallos-Aliaga et al., 2024), which means that the sensor works by a single input → single output. A useful application could be to turn this into an intracellular neural‑network‑like classifier that can receive several inputs inside the same cell to determine if a particular food matrix is likely above a regulatory mercury given limit.
The inputs to the intracellular neural network would be analog signals whose expression levels vary with concentration:
Pmer drives the expression of a regulator, protein A (for example, MerR fused to an activation domain), in response to bioavailable Hg2+.
A separate metal‑induced stress promoter activated by oxidative stress generated during mercury exposure drives regulator B, capturing more general toxicity associated with high mercury levels rather than Hg 2+ alone.
The network using protein A and regulator B would involve creating two layers. The IANN uses A and B as the outputs of two input neurons, which feed into a final decision node. A synthetic promoter (let’s call it Poutput) is designed with binding sites for both protein A and regulator B. Poutput could approximate a weighted sum of variables and concentrations and a threshold, where it would only activate a fluorescent protein when detecting that the combined levels of A and B exceed a defined level. This corresponds to the network classifying the sample as “above the safe mercury limit,” whereas lower or unbalanced inputs keep the output near baseline.
In practice, you could incubate a small portion of a fish fillet (or any other food matrix) with the engineered bacteria. If the intracellular ANN determines that the pattern of Hg 2+ and stress response lies in the “unsafe” region of its input space, the cells switch ON the fluorescent color, indicating that the food sample likely exceeds regulatory mercury limits. If not, the reporter remains OFF, indicating the sample is probably within safe limits to consume.
This design could face an important limitation within metal‑responsive regulators. Sometimes, they show cross‑reactivity with other metals, which may lead to false positives. Also, since response times are affected by transcription, translation, and protein synthesis, an answer to the test could take a long time (more in the hours than in the minutes range), which could be complicated when testing large amounts of samples for human consumption.
References:
Zevallos-Aliaga D, De Graeve S, Obando-Chávez P, Vaccari NA, Gao Y, Peeters T, Guerra DG. Highly Sensitive Whole-Cell Mercury Biosensors for Environmental Monitoring. Biosensors (Basel). 2024 May 13;14(5):246. doi: 10.3390/bios14050246. PMID: 38785720; PMCID: PMC11117708.
ROOINTAN, A, SHABAB, N, KARIMI, J, RAHMANI, A, ALIKHANI, M. Y, & SAIDIJAM, M (2015). Designing a bacterial biosensor for detection of mercury in water solutions. Turkish Journal of Biology 39 (4): 550-555. https://doi.org/10.3906/biy-1411-49
3. Below is a diagram depicting an intracellular single-layer perceptron where the X1 input is DNA encoding for the Csy4 endoribonuclease and the X2 input is DNA encoding for a fluorescent protein output whose mRNA is regulated by Csy4. Tx: transcription; Tl: translation.
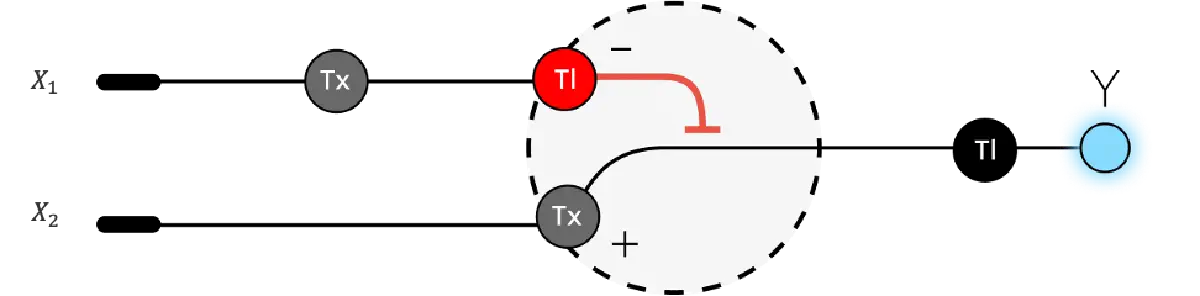

Draw a diagram for an intracellular multilayer perceptron where layer 1 outputs an endoribonuclease that regulates a fluorescent protein output in layer 2.
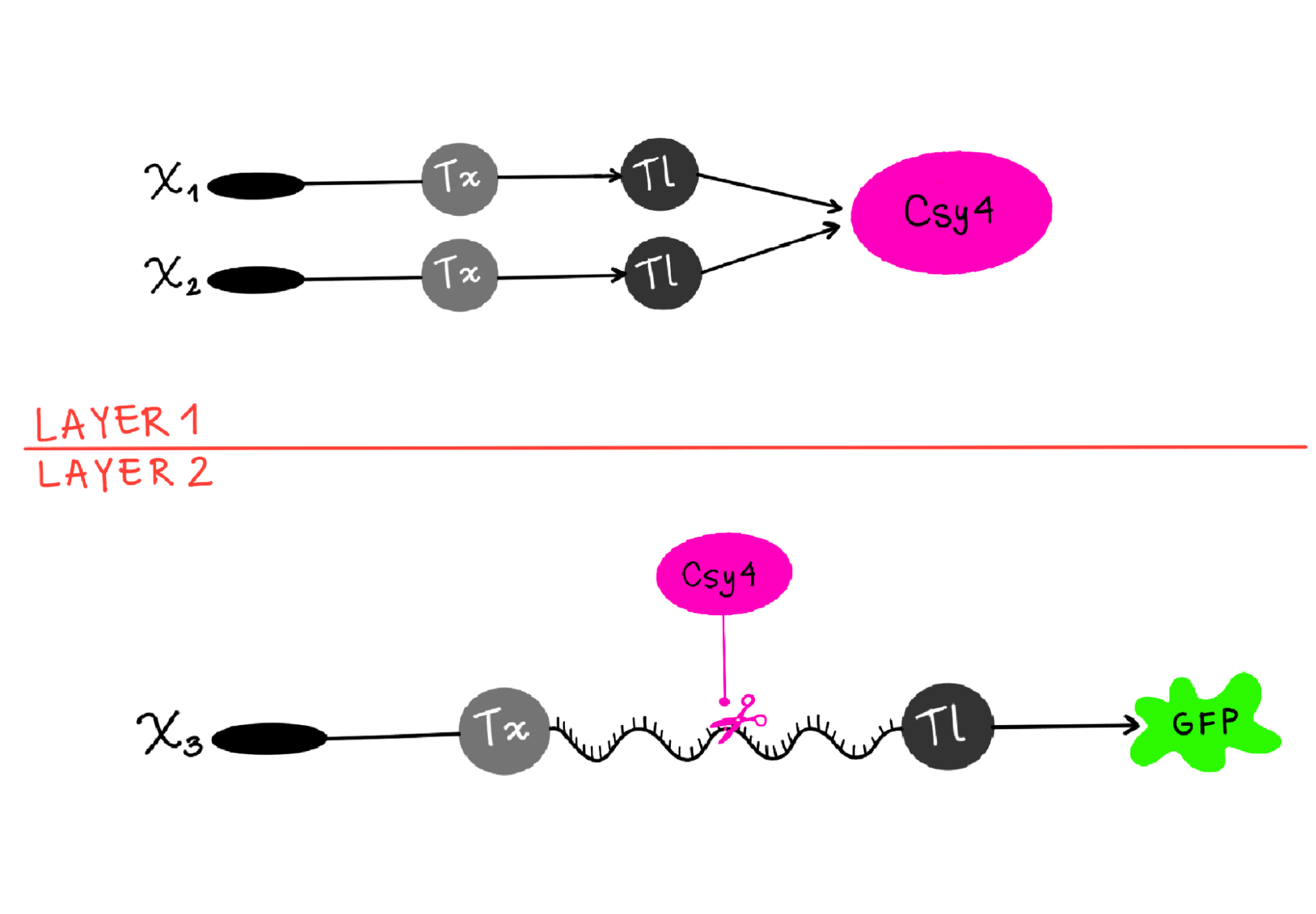

- I’m not sure this diagram is correct, but I tried to show how X1 and X2 are genes, which go through transcription (Tx) and translation (Tl) to then produce the endoribonuclease (Csy4 like in the example) as output in layer 1. And then in Layer 2, Csy4 recognizes the endoribonuclease site in the mRNA of the fluorescent protein (GFP) and cuts it, so when it goes through Translation, the output is a regulated GFP.
Assignment Part 2: Fungal Materials
1. What are some examples of existing fungal materials and what are they used for? What are their advantages and disadvantages over traditional counterparts?
Fungal materials are becoming very popular very fast. I have seen many different uses: construction materials, clothing and jewelry, household objects… But I wanted to find other possibilities, maybe less common ones. These are some I found particularly interesting:
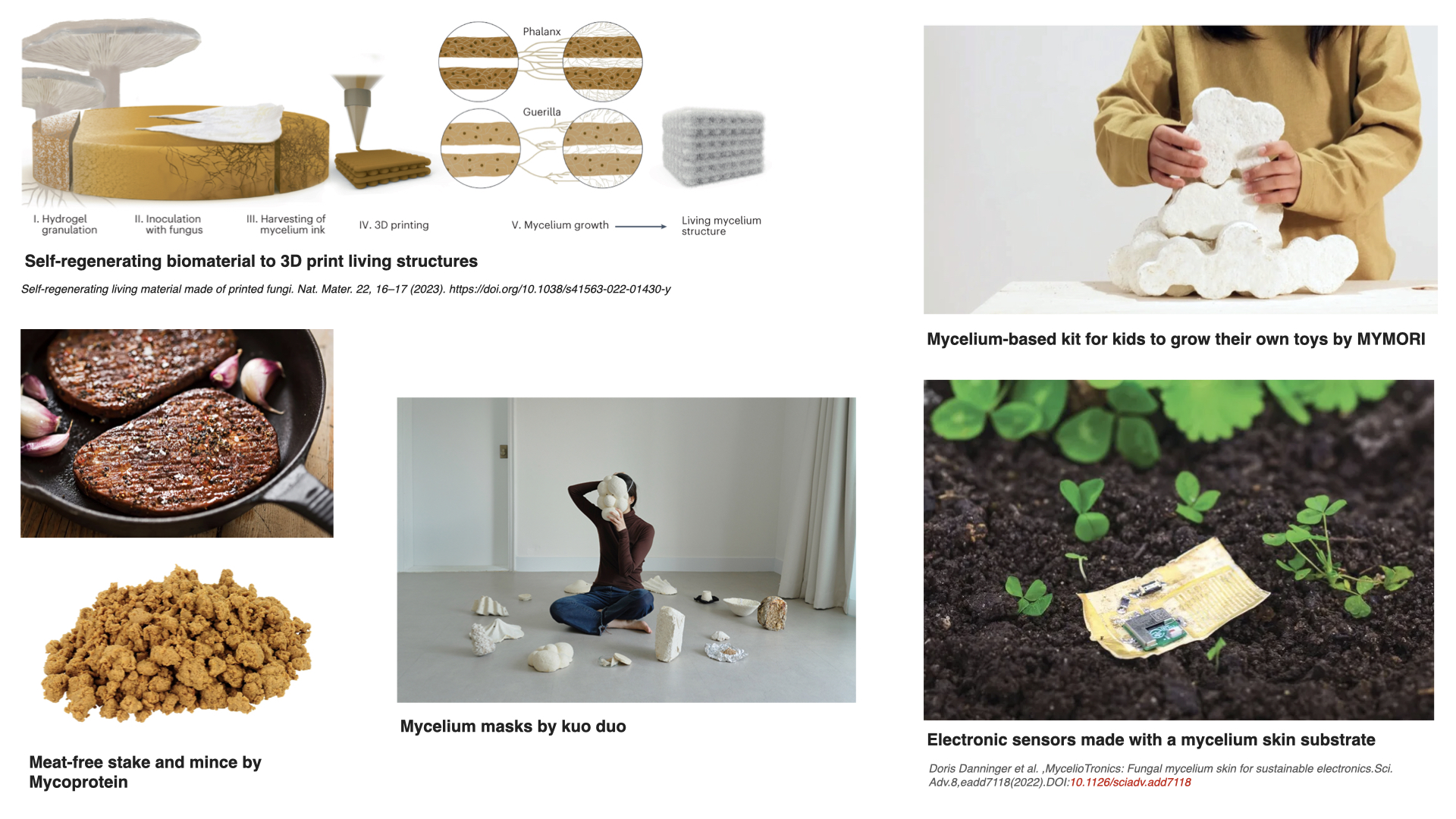

In most examples, ranging from construction materials to meat-free food alternatives, fungi are used because they are easily grown by using cheap plant waste (straw, rye, cardboard, paper, or others), relatively little energy, and more sustainable than the more common industrial alternatives (plastics, animal leather, polystyrene foams, etc.). Also, because their mycelium can easily grow into complex 2D or 3D shapes, bind loose materials into solid composites, and form flexible sheets. In short, its advantages are: renewability, biodegradability, and the ability to be grown with specific textures and forms. Nevertheless, many of these mycelium products need improvements. Some show lower mechanical performance, more vulnerability to moisture, and limited long‑term durability compared to their industrial counterparts. This makes them a bit niche in the beginning and difficulties their direct implementation.
2. What might you want to genetically engineer fungi to do and why? What are the advantages of doing synthetic biology in fungi as opposed to bacteria?
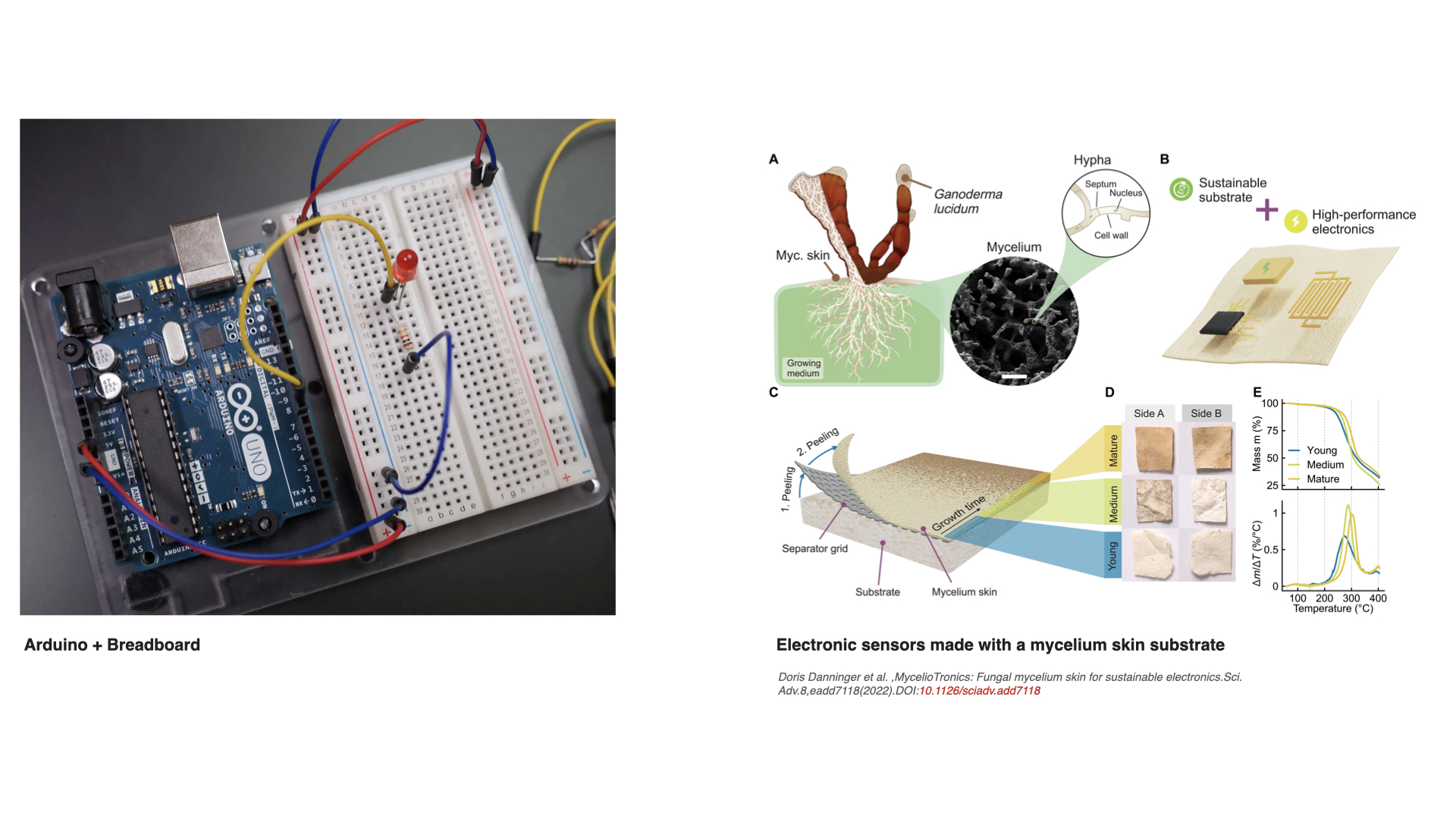

I find the electronic sensors made with mycelium skin substrate by MycelioTronics very interesting, and I immediately thought of the infamous Arduino breadboards. I would engineer fungi to grow mycelium skins that act as biodegradable Arduino‑style breadboards, where the mycelium itself helps carry electrical signals between components. The engineered mycelium could make more conductive substances (for example, metal‑binding molecules) along certain growth paths in the breadboard, so parts of the sheet become “tracks” that electricity can flow through, while the rest stays more insulating and structural. This way, the mycelium acts as the material that supports the components connected on the breadboard but also acts as the network that helps connect the different holes and contact points.
For this idea, fungi are more advantageous than bacteria because they naturally grow continuous 2D and 3D structures, they can be grown in cheaper substrates and sometimes under more simple conditions, and as eukaryotes they are better at more complex engineering. Engineering their DNA could potentially change how the grown sheet behaves as a structural material and as a bioelectric material.