Week 1 Homework: Principles and Practices
Exchangeable Fabs
As an undergraduate student, I worked on a project that required the use of multiple antibodies to bind specific epitopes and stain them. Nonetheless, antibodies are not cheap; they take a long time to get to Latin American laboratories, and they usually suffer from thawing and unthawing processes, causing them to lose efficacy. On another note, commercial antibodies are not fully characterized; therefore, other protein interactions are not screened beforehand, leading to cross-reactivity, which ultimately leads to less reliable results [1].
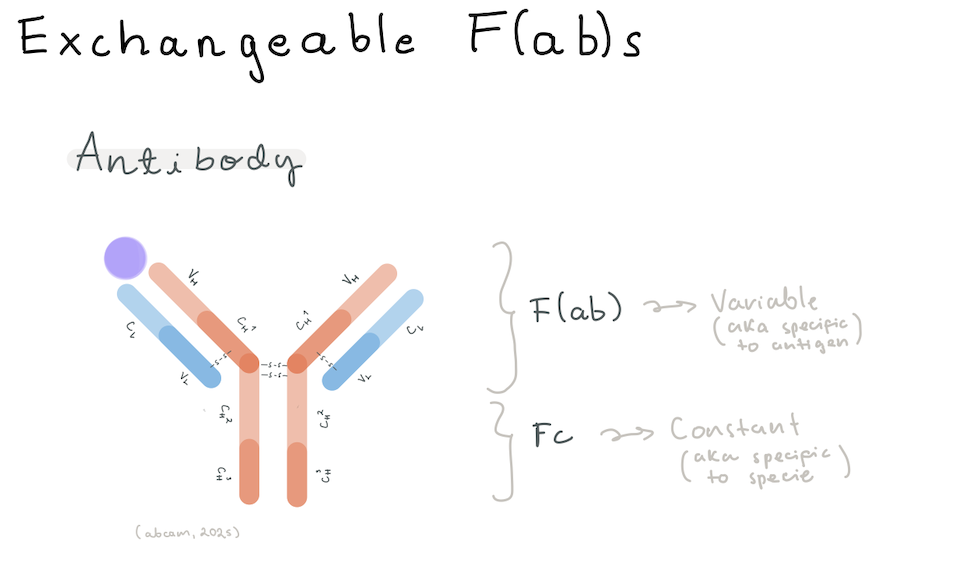
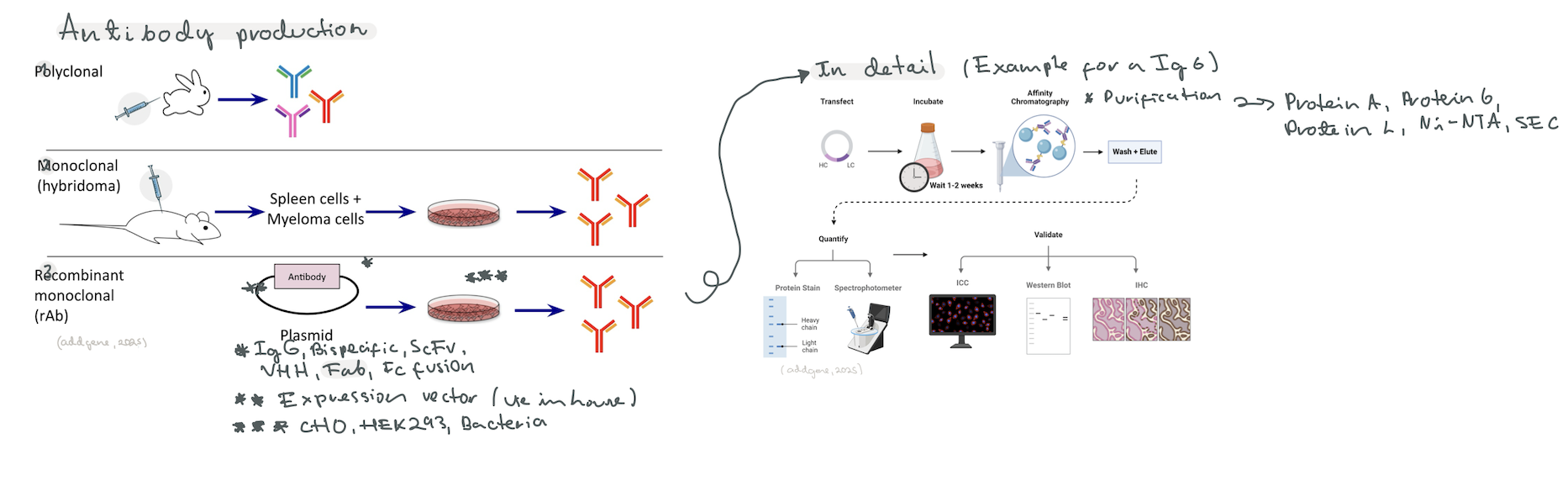
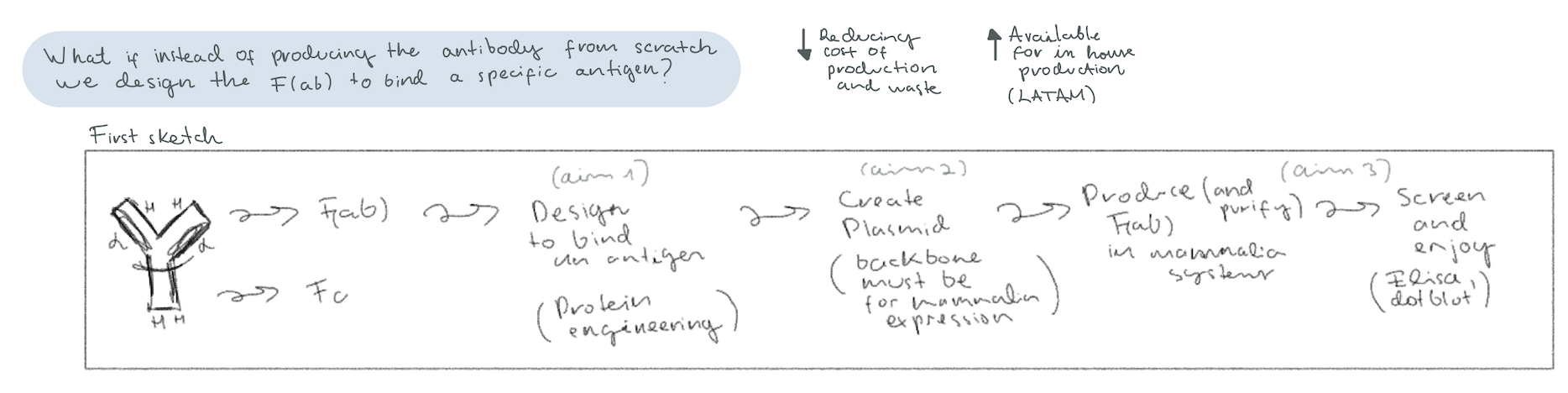
Taking inspiration from research done at the UW Institute for Protein Design [2], I decided to delve into a synthetic biology proposal for the protein engineering of “exchangeable Fabs”. Antibodies have two parts, the Fab (Fragment antigen-binding) region at the arms for antigen recognition, and the Fc (Fragment crystallizable) region at the stem for specific species immune system interaction [3]. The idea is to create a protocol that allows for Top-Down design of the Fab region, where you design a Fab that binds an epitope in silico, and then you go on to produce a plasmid to produce it in-house. I called it “exchangeable Fabs” because the idea in the long run would be to be able to select an epitope and then get a plasmid system that codes for the Fab region that you assemble in your own laboratory with an already acquired plasmid for the Fc region.
Governance Goals and Proposed Actions
Considering what I learned in class and drawing on Baker and Church’s Protein Design Meets Biosecurity, I define a high-level governance goal and propose three complementary governance actions.

High-Level Goal
Ensure that computationally designed, exchangeable Fab fragments are developed and deployed in ways that maximize scientific reliability, equity, and safety while minimizing risks of misuse, experimental harm, and environmental burden.
This overarching goal can be broken down into the following sub-goals:
Non-malfeasance and Biosecurity
- Prevent the misuse of antibody design pipelines to generate harmful or deceptive biological tools.
- Reduce the risk of poorly characterized binding agents entering experimental or clinical workflows.
Laboratory Safety and Scientific Integrity
- Ensure that designed Fab fragments are traceable, validated, and reproducible.
- Reduce experimental artifacts caused by cross-reactivity or undocumented binding behavior.
Equitable Access and Global Scientific Inclusion
- Lower structural barriers for under-resourced laboratories (e.g., in Latin America) to access reliable affinity reagents.
- Avoid creating new dependencies on proprietary platforms or centralized institutions.
Environmental Sustainability
- Reduce waste associated with disposable commercial antibodies and repeated cold-chain shipping.
- Minimize redundant production of poorly validated reagents.
Governance Actions
I propose three complementary governance actions, pursued by different actors and using distinct mechanisms.
Option 1: Standardized Fab Design and Validation Protocols (Academic Setting)
Purpose
Currently, antibodies, especially commercial ones, are often under-characterized, with limited disclosure of off-target interactions. I propose the development of community-wide standards for the computational design, experimental validation, and reporting of exchangeable Fab fragments.
Design
Actors: Academic researchers, journals, funding agencies.
Requirements:
- Minimum in silico validation benchmarks (e.g., binding specificity screens).
- Mandatory disclosure of design parameters and validation assays.
- Journal and funder requirements for compliance as a condition of publication or funding.
Assumptions
- Computational predictions meaningfully correlate with in vitro behavior.
- Academic communities are willing to converge on shared standards.
Risks of Failure and “Success”
- Failure risk: Standards may be unevenly adopted or reduced to box-checking exercises.
- Success risk: Over-standardization could slow exploratory or unconventional research.
Option 2: Modular Licensing of Fab–Fc Systems (Incentive-Based Governance)
Purpose
To prevent misuse while promoting access, I propose a modular licensing framework in which validated Fc backbones are shared under open or semi-open licenses, while Fab designs are distributed with usage constraints.
Design
Actors: Universities, nonprofit consortia, biotechnology companies.
Mechanisms:
- Fc regions distributed as standardized, safety-validated plasmids.
- Fab designs tagged with metadata describing intended use, validation level, and constraints.
- Governance structures analogous to open-source software licenses with biological usage clauses.
Assumptions
- Laboratories will comply with licensing norms in the absence of strong enforcement mechanisms.
- Existing plasmid-sharing infrastructures can support metadata-rich distribution.
Risks of Failure and “Success”
- Failure risk: Informal sharing networks may bypass governance mechanisms.
- Success risk: Licensing complexity may discourage adoption by smaller or under-resourced laboratories.
Option 3: Technical Safeguards Embedded in Design Pipelines
Purpose
Rather than relying solely on rules or norms, this option embeds technical constraints directly into antibody design tools to reduce the likelihood of harm.
Design
Actors: Tool developers, computational biologists, funding agencies.
Features:
- Automated screening against databases of known toxin-related or pathogenic epitopes.
- Flagging or blocking of designs with high-risk similarity profiles.
- Audit logs for design requests, analogous to controls used in DNA synthesis.
Assumptions
- Reliable and up-to-date databases of hazardous epitopes exist.
- Researchers will tolerate a degree of friction in design workflows in exchange for increased safety.
Risks of Failure and “Success”
- Failure risk: False positives may block benign or exploratory research.
- Success risk: Users may migrate to unregulated or less transparent tools.
Scoring of Governance Options Against Policy Goals
| Policy Goal / Criterion | Option 1 | Option 2 | Option 3 |
|---|---|---|---|
| Enhance Biosecurity | 2 | 2 | 1 |
| • By preventing incidents | 2 | 2 | 1 |
| • By helping respond | 2 | 3 | 2 |
| Foster Laboratory Safety | 1 | 2 | 2 |
| • By preventing incidents | 1 | 2 | 2 |
| • By helping respond | 2 | 3 | 2 |
| Protect the Environment | 2 | 1 | 2 |
| • By preventing incidents | 2 | 1 | 2 |
| • By helping respond | n/a | n/a | n/a |
| Other Considerations | |||
| • Minimizing costs and burdens to stakeholders | 2 | 3 | 2 |
| • Feasibility | 1 | 2 | 2 |
| • Not impede research | 1 | 2 | 3 |
| • Promote constructive applications | 1 | 1 | 2 |
Scoring scale: 1 = best performance; 3 = weakest performance; n/a = not applicable.
References
[1] Baker, M. Reproducibility crisis: Blame it on the antibodies. Nature (2019).
https://www.nature.com/articles/d42473-019-00273-7
[2] Institute for Protein Design. Designing antibodies with RFdiffusion (2025).
https://www.bakerlab.org/2025/02/28/designing-antibodies-with-rfdiffusion/
[3] Janeway, C. Immunobiology. Garland Science.
https://www.ncbi.nlm.nih.gov/books/NBK27144/
AI assistance was used to organize, clarify, and refine explanations