Week 1 HW: Principles and Practices
Homework Week 1
Biological Engineering Application
First, describe a biological engineering application or tool you want to develop and why. This could be inspired by an idea for your HTGAA class project and/or something for which you are already doing in your research, or something you are just curious about.
A biological engineering application I want to develop is a bioactive skin‑repair sunscreen that not only blocks ultraviolet (UV) radiation but also repairs UV‑induced intracellular damage in skin cells. The goal is to prevent skin cancer more effectively than traditional sunscreens, which mainly work by absorbing or reflecting UV light rather than actively reversing oxidative damage. I am particularly motivated by living in high‑altitude, high‑UV environments (Colorado and Utah), where cumulative sun exposure and sunburn risk are significant. Traditional sunscreens can be underused, improperly applied, or raise environmental concerns, especially regarding some organic UV filters and marine ecosystems. A bioactive formulation that incorporates antioxidant enzymes and intracellular delivery peptides could help address these limitations by scavenging reactive oxygen species (ROS) generated after UV exposure. Implementing this vision would require advances in protein engineering, biomanufacturing, formulation science, and automated biodesign.
Reference: Chen, X. et al. (2016) “Topical application of superoxide dismutase mediated by HIV-TAT peptide attenuates UVB-induced damages in human skin,” European Journal of Pharmaceutics and Biopharmaceutics, 107, pp. 286–294. Available at: https://doi.org/10.1016/j.ejpb.2016.07.023.
Here is a potential biomanufacturing workflow I have designed for a simplistic version of this bioactive sunscreen.
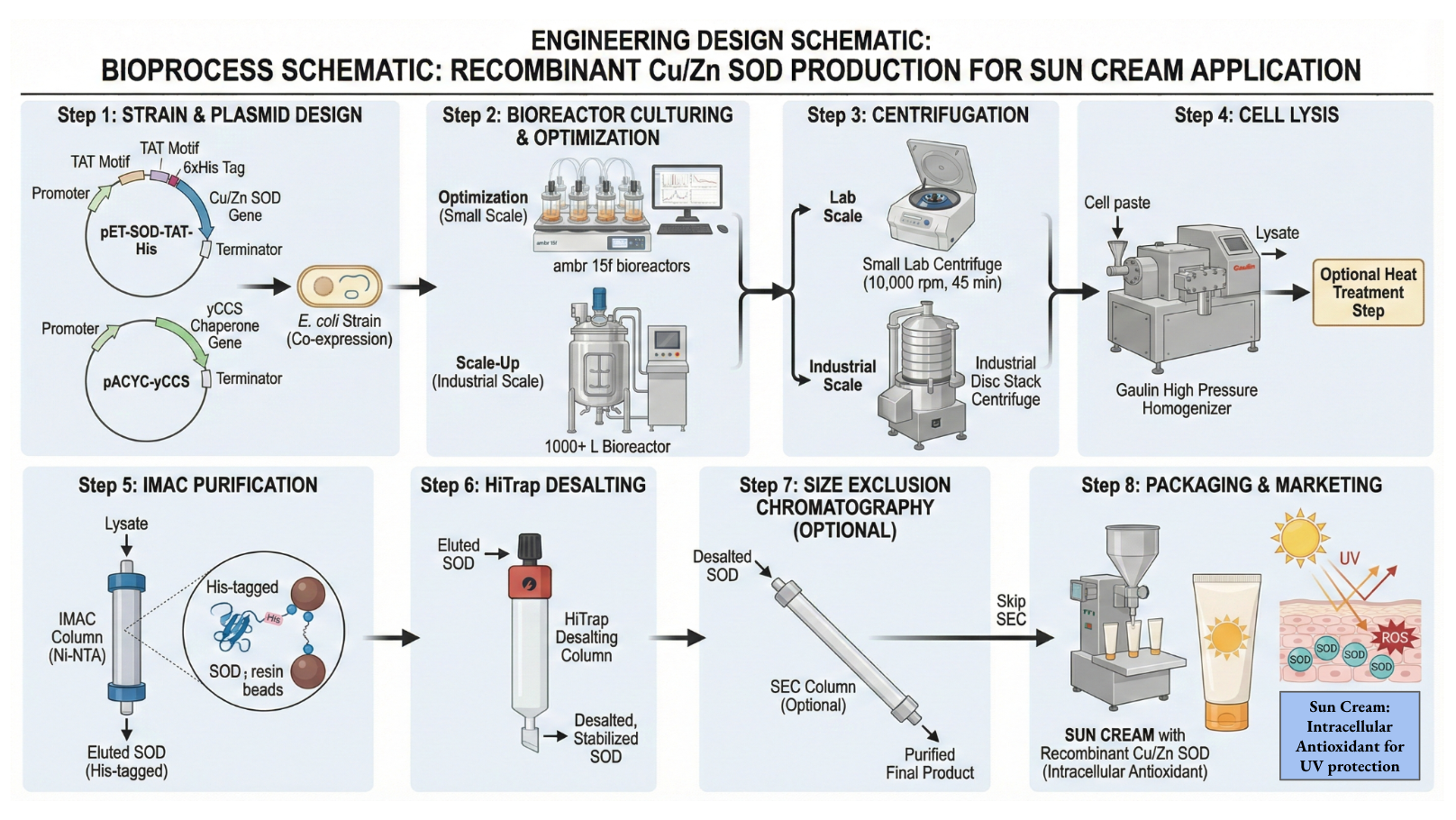
 Briefly, the workflow involves: recombinant expression of a therapeutic enzyme such as superoxide dismutase (SOD), purification and quality control, fusion or formulation with a cell‑penetrating peptide (for example, TAT), and incorporation into a stable topical vehicle compatible with large‑scale manufacturing and regulatory requirements.
Briefly, the workflow involves: recombinant expression of a therapeutic enzyme such as superoxide dismutase (SOD), purification and quality control, fusion or formulation with a cell‑penetrating peptide (for example, TAT), and incorporation into a stable topical vehicle compatible with large‑scale manufacturing and regulatory requirements.
Governance and policy goals
Next, describe one or more governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future, like ensuring non-malfeasance (preventing harm). Break big goals down into two or more specific sub-goals.
My overarching governance goal is to ensure that a bioactive sunscreen is safe, trusted, and equitably accessible, rather than becoming a niche or risky cosmetic for a privileged few. In particular, I would prioritize:
- Bioliteracy and trust: The public should understand, at a basic level, how the product works (e.g., enzyme‑mediated ROS scavenging and intracellular delivery) and why it is considered safe, to avoid both hype and backlash.
- Robust safety and regulatory oversight: Regulatory review (e.g., FDA or equivalent) and clinical trials should evaluate not only acute irritation and erythema, but also systemic exposure, long‑term effects, and off‑label use (eyes, mucous membranes, damaged skin).
- Equitable access and affordability: Pricing and distribution policies should ensure that high‑risk populations (rural communities, outdoor workers, high‑UV regions) can obtain the product at cost parity with or below conventional sunscreens.
Governance Actions
Next, describe at least three different potential governance “actions” by considering the four aspects below (Purpose, Design, Assumptions, Risks of Failure & “Success”). Try to outline a mix of actions (e.g. a new requirement/rule, incentive, or technical strategy) pursued by different “actors” (e.g. academic researchers, companies, federal regulators, law enforcement, etc). Draw upon your existing knowledge and a little additional digging, and feel free to use analogies to other domains (e.g. 3D printing, drones, financial systems, etc.).
Purpose: What is done now and what changes are you proposing?
Traditional sunscreens are regulated primarily on their ability to block UV and their short‑term safety, yet there are still gaps in evidence for cancer prevention and long‑term systemic safety. For a bioactive sunscreen, I propose strengthening peer‑reviewed evidence and clinical trials that specifically evaluate reduction in UVB‑induced skin damage, biomarker changes, and long‑term cancer risk proxies.
Design: What is needed to make it “work”? (including the actor(s) involved - who must opt-in, fund, approve, or implement, etc)
Preclinical and formulation phase
- Academic and industrial labs characterize enzyme stability, activity after formulation, skin penetration, and reduction of UV‑induced oxidative damage in ex vivo and small in vivo models.
- Formulation scientists optimize the topical vehicle to control enzyme release, minimize systemic absorption, and ensure compatibility with existing UV filters.
- Research funders (public agencies, foundations) support comparative studies that benchmark bioactive sunscreen against high‑quality conventional products, not substandard controls.
Early‑stage human studies
- Clinical research centers conduct small randomized trials that measure biomarkers such as erythema, sunburn cells, DNA damage indicators, and local inflammation in human volunteers, similar in scale to existing sunscreen and nanoparticle‑based sunscreen studies.
- These trials include participants with a range of skin types and ages, and track short‑term tolerability, sensitization, and signs of systemic exposure.
- Independent ethics boards oversee study design, with clear informed consent that explains the bioengineered components and remaining uncertainties.
Pivotal trials and regulatory review
- Larger, longer trials follow people over months or seasons of real‑world use, combining controlled UV‑challenge tests with everyday conditions.
- Regulators treat the bioactive sunscreen as a drug‑like product rather than a simple cosmetic, requiring data on chronic use, systemic exposure, and environmental release, in line with recent calls to modernize sunscreen regulation.
- Results and protocols are published and deposited in open repositories, with pre‑registration of trials to reduce publication bias and allow independent reanalysis.
Public acceptance and uptake
- Honest media and marketing that explain mechanism, benefits, risks, and limits
- Coordination with clinicians and public‑health agencies so it is framed as a preventive health tool
- Take lessons from GLP‑1 drugs: strong evidence plus clear communication and clinician endorsement to build trust and awareness
Assumptions: What could you have wrong (incorrect assumptions, uncertainties)?
This plan assumes that the bioactive sunscreen will ultimately prove cost‑effective enough that payers and governments are willing to fund subsidies, which depends on strong trial data and realistic cost projections. It assumes that manufacturers can scale production without dramatic price spikes or supply bottlenecks. It also assumes that providing the product at low or no cost will translate into consistent use, which may not hold without ongoing education and cultural alignment. The effectiveness and mechanisms of action, outlined in Chen et al. 2016, could be found to be not accurate and overestimated in different skin types or groups of people. This product could always fail in clinical trials or be found unsafe. Public acceptance could be tricky, the TAT peptides being sourced from HIV could create public backlash and getting ahead of such a media storm and sourcing another TAT peptide that is not associated with a disease would be ideal.
Reference: Chen, X. et al. (2016) “Topical application of superoxide dismutase mediated by HIV-TAT peptide attenuates UVB-induced damages in human skin,” European Journal of Pharmaceutics and Biopharmaceutics, 107, pp. 286–294. Available at: https://doi.org/10.1016/j.ejpb.2016.07.023.
Risks of Failure & “Success”: How might this fail, including any unintended consequences of the “success” of your proposed actions?
Failures: This product could always fail in clinical trials or be found unsafe. If it’s not actually feasible to generate a bioactive and protective sunscreen that outperforms traditional sunscreens and is cost competitive that could cause development of this product to fail or hit roadblocks.
Consequences: There could be long term health consequences for such a product not caught in clinical trials. Public acceptance of such a biotechnology product could lead to the acceptance of other more dangerous products.
Ideally, the product would work and a bioactive sunscreen would reduce skin cancer rates drastically and equitably.
3 governance actions:
Action 1: Peer review and clinical trials
- Purpose: Traditional sunscreens are regulated primarily on their ability to block UV and their short‑term safety, yet there are still gaps in evidence for cancer prevention and long‑term systemic safety. For a bioactive sunscreen, I propose strengthening peer‑reviewed evidence and clinical trials that specifically evaluate reduction in UVB‑induced skin damage, biomarker changes, and long‑term cancer risk proxies.
- Design:
- Academic and industrial researchers design studies that characterize enzyme stability, skin penetration, ROS reduction, and DNA damage markers in human skin.
- Clinical investigators run randomized controlled trials to compare the bioactive sunscreen with standard SPF‑matched sunscreens across diverse skin types and age groups.
- Regulators (e.g., FDA or international counterparts) require data on systemic absorption, endocrine disruption potential, and chronic exposure, consistent with current concerns about conventional actives.
- Independent review boards and journals enforce transparency, data sharing, and conflict‑of‑interest disclosures.
- Assumptions: This action assumes that mechanisms seen in early clinical and ex vivo studies (e.g., TAT‑SOD decreasing UVB‑induced damage) will translate to large, diverse populations and real‑world use. It also assumes that trial endpoints (e.g., sunburn cells, oxidative stress biomarkers) are valid proxies for long‑term cancer risk.
- Risks of failure and “success”:
- Failure risks: Trials could reveal limited benefit over high‑quality traditional sunscreens, unacceptable safety issues, or strong inter‑individual variability.
- Success risks: Demonstrating strong protection might create risk compensation, where users rely solely on the product and ignore complementary behaviors like shade or protective clothing.
Action 2: Media, marketing, and science communication
Purpose: I propose a coordinated science communication strategy to build informed trust rather than hype, ensuring the product is understood as one part of a broader sun‑safety toolkit.
Design (actors and implementation):
Science communicators, dermatologists, and public health agencies jointly develop clear, accessible messaging about how the bioactive sunscreen works, what is known, and what remains uncertain.
Marketing campaigns emphasize evidence‑based benefits, appropriate use, and limitations, avoiding sensational claims about being “sunburn‑proof” or “cancer‑proof.”
Community health workers and local organizations co‑design outreach in high‑risk communities, emphasizing culturally relevant framing and local representation, echoing the importance of trust described by George Shultz.
Assumptions: This action assumes that transparent, co‑created communication can overcome distrust linked to “biotech,” “engineered enzymes,” or associations with HIV‑derived TAT peptides. It also assumes that industry actors will accept constraints on exaggerated marketing.
Risks of failure and “success”:
- Failure risks: Poorly framed messages could trigger fear (e.g., focus on “HIV‑TAT”) or be dismissed as corporate spin.
- Success risks: Extremely successful marketing might oversell benefits and normalize dependence on a proprietary product instead of promoting broader sun‑safety and environmental protections.
Action 3: Equitable distribution and subsidization
Purpose: I propose policies to treat bioactive sunscreen as a preventive health tool rather than a luxury cosmetic, aligning distribution with skin‑cancer prevention goals.
Design (actors and implementation):
Public health agencies and insurers consider covering the product for high‑risk groups (e.g., outdoor workers, immunocompromised patients, very fair skin types) as a preventive intervention.
Governments and payers negotiate pricing or volume guarantees to keep retail costs comparable to or lower than common sunscreens in pharmacies and supermarkets.
Community clinics and rural health programs distribute the product alongside educational materials on sun safety and skin‑cancer screening.
Assumptions: This action assumes that strong cost‑effectiveness arguments can be made (e.g., reduced future skin‑cancer treatment costs) and that manufacturing can scale without driving prices beyond what subsidies can realistically cover.
Risks of failure and “success”:
Failure risks: If manufacturing remains expensive, subsidies could be politically unpopular or unsustainable, reinforcing inequities.
Success risks: If access is expanded without strong safety and literacy measures, misuse or overreliance could spread broadly rather than being confined to early adopters.
Next, score (from 1-3 with, 1 as the best, or n/a) each of your governance actions against your rubric of policy goals. The following is one framework but feel free to make your own:
| Does the option: | Peer Review/Clinical Trials | Media and Marketing | Equitable Distribution |
|---|---|---|---|
| Enhance Biomedicine | 1 | n/a | 2 |
| • By preventing disease | 1 | n/a | 2 |
| • By helping treat illness | 1 | n/a | 2 |
| • Ensure safe bioproduct | 1 | n/a | n/a |
| Enhance Bioliteracy | 1 | 2 | 3 |
| • By educating the public | 2 | 1 | 3 |
| • By fostering trust in biotechnologies | 1 | 3 | 2 |
| Protect the environment | 3 | 2 | 1 |
| • By reducing other sunscreens | n/a | 1 | 2 |
| Other considerations | |||
| • Minimizing costs and burdens to stakeholders | 1 | 2 | 3 |
| • Feasibility? | 1 | 2 | 3 |
| • Promote constructive applications | 3 | 1 | 2 |
Last, drawing upon this scoring, describe which governance option, or combination of options, you would prioritize, and why. Outline any trade-offs you considered as well as assumptions and uncertainties. For this, you can choose one or more relevant audiences for your recommendation, which could range from the very local (e.g. to MIT leadership or Cambridge Mayoral Office) to the national (e.g. to President Biden or the head of a Federal Agency) to the international (e.g. to the United Nations Office of the Secretary-General, or the leadership of a multinational firm or industry consortia). These could also be one of the “actor” groups in your matrix.
I would prioritize a combined strategy that begins with rigorous peer review and clinical trials (Action 1), then layers on responsible science communication (Action 2), and finally builds toward equitable distribution (Action 3). Robust evidence generation is non‑negotiable; without it, any marketing or subsidization risks amplifying harms or wasting resources. Once a strong safety and efficacy case exists, coordinated communication can promote informed uptake while explicitly acknowledging uncertainties and avoiding overclaiming. Only after those foundations are in place would I advocate for large‑scale subsidization and integration into public health programs, given the political, economic, and logistical complexity of equitable distribution.
For a target audience, I would imagine this recommendation addressed to a national public‑health agency (a federal department of health or an NIH‑like funding body) that can coordinate research funding, regulatory pathways, and outreach partnerships. A key trade‑off is time: prioritizing careful trials and communication may slow wide deployment, but it helps preserve long‑term public trust and reduces the risk that an early failure poisons attitudes toward future bioactive preventives. My largest uncertainties involve real‑world cost‑effectiveness, long‑term systemic safety, environmental impact compared with current sunscreens, and how public attitudes toward engineered biologics will evolve.
Reflecting on what you learned and did in class this week, outline any ethical concerns that arose, especially any that were new to you. Then propose any governance actions you think might be appropriate to address those issues.
In class, the discussion of ethics, safety, and governance highlighted how quickly powerful biotechnologies can move from lab to market and how unevenly risks and benefits are distributed across communities. George P. Shultz’s essay “Trust Is the Coin of the Realm” reinforced that durable trust is built when communities see their own members involved in decision‑making, rather than having technologies imposed on them. For bioactive sunscreen and similar interventions, this suggests that governance should not only involve regulators and companies but also community representatives in high‑UV, high‑risk areas, including rural, low‑income, and historically marginalized groups.
Governance actions I see as appropriate include: funding community‑based participatory research around sun‑safety tools; requiring public comment and local advisory boards when introducing novel preventive biotechnologies; and supporting training programs that help people from affected communities become scientists, clinicians, and communicators. These steps could align the development of bioactive sunscreens with the broader ethical goal of biotechnology that is responsive to, and shaped by, the communities it is meant to serve