Individual Final Project
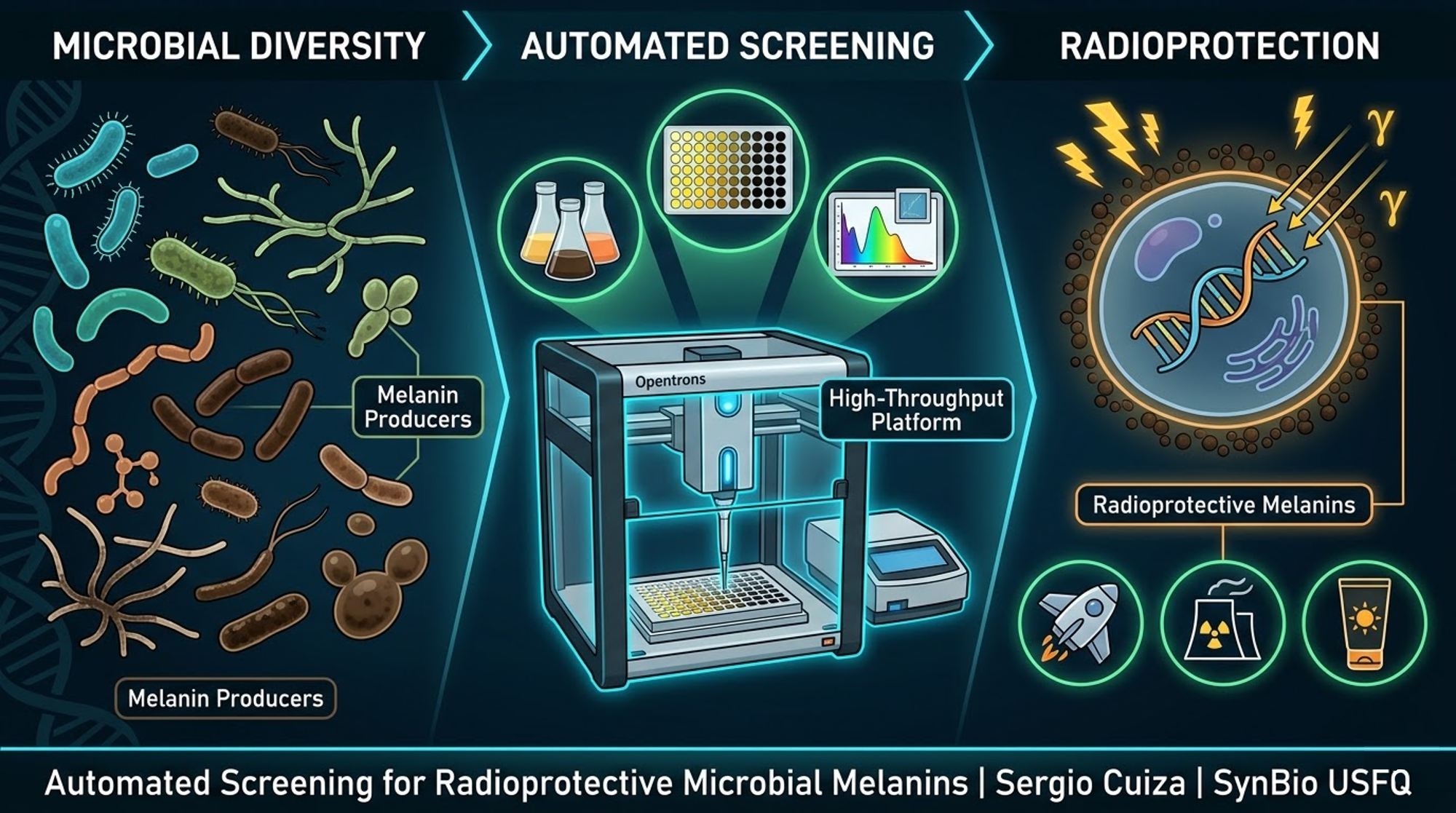

Automated High-Throughput Screening Platform for Radioprotective Microbial Melanins
ABSTRACT Ionizing radiation poses significant challenges for space exploration, nuclear facilities, medical applications, and industrial safety. Current radiation shielding materials are often synthetic, heavy, and environmentally problematic. Microbial melanins — natural biopolymers produced by bacteria, yeast, and other microorganisms — have demonstrated remarkable capacity to absorb various forms of radiation including UV, gamma, and X-rays. However, discovering and optimizing microbial strains that produce radioprotective melanins remains a slow, manual process. This project proposes the design of an automated high-throughput screening platform using Opentrons liquid handling robotics to systematically test diverse microorganisms for melanin production and radiation absorption capabilities. The workflow includes automated inoculation, controlled radiation exposure, spectrophotometric quantification, and functional radioprotection assays. The platform is designed for remote execution via cloud laboratories, enabling scalability and accessibility even without local wet-lab infrastructure. The expected outcomes include a validated automated protocol, identification of high-performance melanin-producing strains, and characterized melanin samples with demonstrated radioprotective properties for biotechnological applications.
- INTRODUCTION 1.1 The Radiation Problem Radiation exposure is a fundamental challenge across multiple domains. Astronauts on deep space missions face chronic exposure to galactic cosmic radiation. Nuclear facility workers require protection during routine operations and emergency responses. Patients undergoing radiation therapy experience damage to healthy tissues surrounding tumors. Electronic equipment in high-radiation environments degrades prematurely. Each of these scenarios demands effective radiation protection.
Current solutions have significant limitations. Lead shielding is heavy and toxic. Polymer-based materials offer limited protection. Synthetic additives may degrade or release harmful compounds. The need for lightweight, biocompatible, and sustainable radioprotective materials is urgent and growing.
1.2 Melanin as a Radioprotective Material Melanin is a complex biopolymer found across many life forms. It provides pigmentation, but more importantly, it offers protection against environmental stresses including radiation. Research following the Chernobyl disaster revealed that certain fungi not only survived high radiation environments but actually thrived, with melanin playing a central role in their radiotolerance.
The mechanism involves multiple physical and chemical properties. Melanin absorbs electromagnetic radiation across a broad spectrum. It scavenges free radicals generated by radiation exposure. It can undergo reversible oxidation-reduction cycles that may even allow energy conversion. These properties make melanin exceptionally promising as a biological radioprotector.
Importantly, melanin is not limited to fungi. Many bacteria produce melanin, often with different structural characteristics that may confer unique radioprotective properties. Actinomycetes like Streptomyces species produce dark pigments. Pseudomonas aeruginosa produces pyomelanin under specific conditions. Bacillus species synthesize melanin-like compounds. Each of these represents a potential source of radioprotective material.
1.3 The Screening Challenge The fundamental problem this project addresses is discovery. How do we find the best microbial melanin producers among thousands of possibilities? How do we determine which growth conditions maximize production of the most radioprotective forms? How do we test whether a particular melanin actually protects against radiation?
Traditional approaches are manual, slow, and low-throughput. A researcher might test one organism at a time, one condition at a time, measuring one parameter at a time. This approach cannot explore the full landscape of microbial diversity and cultivation variables.
Automation offers a solution. Liquid handling robots can prepare dozens of media formulations, inoculate hundreds of microbial strains, and monitor thousands of samples over time. Plate readers can measure optical density, pigment absorbance, and functional assays automatically. The combination enables high-throughput experimentation that would be impossible manually.
1.4 Project Objectives This project aims to design an automated platform specifically for discovering microbial melanins with radioprotective properties. The specific objectives are:
First, to develop a robotic workflow using Opentrons for culturing diverse melanin-producing microorganisms in 96-well plate format with controlled media variations.
Second, to integrate methods for controlled radiation exposure and subsequent measurement of melanin production and microbial survival.
Third, to implement functional assays that directly test whether extracted melanins can protect reporter cells from radiation damage.
Fourth, to validate the platform through proof-of-concept experiments and deliver a protocol suitable for remote execution in cloud laboratories.
- BIOLOGICAL FOUNDATIONS 2.1 Melanin Types and Their Properties Melanin is not a single compound but a family of related biopolymers with shared characteristics and important distinctions.
Eumelanin is the most common form, producing black or brown pigmentation. It is polymerized from dihydroxyindole precursors and exhibits broadband absorption from UV through visible light. Eumelanin conducts electricity, chelates metals, and shows remarkable stability. Bacteria in the genus Streptomyces produce eumelanin, as do many fungi.
Pheomelanin produces yellow to red-brown colors and incorporates sulfur into its structure. It is less photostable than eumelanin but offers different antioxidant properties. Some Bacillus species produce pheomelanin-like pigments.
Pyomelanin is produced via a different pathway involving homogentisic acid polymerization. Pseudomonas aeruginosa produces pyomelanin under certain conditions, and this form shows particular effectiveness at metal binding and potentially unique radiation interactions.
Allomelanins represent a diverse category of nitrogen-free melanins produced by various fungi and bacteria, with structures adapted to specific ecological niches.
Each melanin type may offer different advantages for radioprotection. Broadband absorption suggests eumelanin might be most effective across radiation types. Metal chelation could enable secondary protective mechanisms. Conductivity raises questions about energy dissipation pathways. The automated platform must be capable of detecting and distinguishing these various forms.
2.2 Microbial Producers The microbial world offers enormous diversity of melanin producers suitable for screening.
Among bacteria, Streptomyces species are well-documented eumelanin producers. Streptomyces glaucescens produces dark pigments during sporulation. Streptomyces antibioticus synthesizes melanin in response to specific nutrients. These bacteria are safe, fast-growing, and genetically tractable.
Pseudomonas aeruginosa produces pyomelanin when the tyrosine degradation pathway is active. While pathogenic strains exist, environmental isolates can be handled safely with standard precautions. The pyomelanin of Pseudomonas has been studied for metal bioremediation and shows distinct properties from eumelanin.
Bacillus species including Bacillus subtilis can produce melanin-like pigments under stress conditions. Their rapid growth and established laboratory protocols make them attractive screening candidates.
Marine bacteria represent an underexplored resource. Deep-sea isolates adapted to high pressure and darkness may produce melanins with unique properties. Extreme environments often yield extreme biochemistry.
Yeast including Cryptococcus species produce melanin that has been directly implicated in radiation resistance. While the Chernobyl fungi captured public attention, the underlying biochemistry exists across diverse yeast genera.
The screening platform must accommodate this diversity, using culture conditions appropriate for different microbial types while maintaining standardized measurement protocols.
2.3 Radiation Biology and Melanin Function Understanding how melanin protects against radiation requires understanding what radiation does to living systems.
Ionizing radiation creates reactive oxygen species that damage DNA, proteins, and membranes. The primary biological damage is often indirect, mediated by these free radicals rather than direct molecular hits.
Melanin provides protection through multiple mechanisms. First, it physically absorbs and scatters radiation, reducing the dose reaching sensitive cellular targets. Second, it acts as a free radical sink, neutralizing reactive species before they cause damage. Third, it may participate in electron transfer processes that dissipate energy harmlessly.
Studies on Cryptococcus neoformans showed that melanized cells survived significantly higher radiation doses than non-melanized controls. The effect was not merely passive shielding; melanin appeared to be actively involved in cellular recovery processes.
Importantly, melanin extracted from cells retains radioprotective properties. This means purified melanin could be incorporated into materials, coatings, or formulations independent of living organisms. The screening platform must therefore assess not only melanin production but also the functional performance of extracted material.
- AUTOMATION PLATFORM DESIGN 3.1 Hardware Components The platform centers on the Opentrons OT-2 liquid handling robot. This open-source automated pipetting system offers precision, programmability, and accessibility. It can accommodate 96-well plates, reagent reservoirs, and custom labware on its deck. Python-based protocol development enables complex experimental designs and remote execution.
A plate reader capable of absorbance measurements across UV-visible spectrum is essential. For melanin quantification, measurements at 405 nm correlate with pigment concentration, while full spectral scans from 350 to 700 nm provide information about melanin type and quality. Some plate readers also offer fluorescence and luminescence modes that could expand functional assay capabilities.
Incubation with shaking maintains cultures under controlled conditions. Temperature control between 20 and 37 degrees Celsius accommodates diverse microbial types. Shaking ensures oxygenation and prevents settling.
For radiation exposure, several approaches are possible. UV lamps can be integrated into the platform for UV-B and UV-C exposure experiments. For gamma or X-ray simulation, chemical radiation mimetics or external irradiation facilities would be required. The platform design must accommodate transfer of plates to and from radiation sources while maintaining sterility and tracking.
3.2 Software and Protocol Architecture The experimental logic is implemented in Python scripts controlling the Opentrons robot. The protocol architecture includes modular components that can be combined flexibly.
The inoculation module handles preparation of seed cultures, normalization of cell density, and distribution to experimental plates. Different microbial types may require different inoculation strategies, accommodated through conditional logic.
The media preparation module combines stock solutions to create defined media variations. Nutrient gradients, stress inducers, and melanin precursors can be varied systematically across plates.
The sampling module transfers aliquots from culture plates to measurement plates at defined timepoints. This enables kinetic measurements of melanin production without disturbing the main culture.
The data collection module coordinates with the plate reader, triggering measurements and storing results in structured formats for downstream analysis.
3.3 Experimental Workflow The complete experimental workflow proceeds through defined stages.
Stage one involves preparation of microbial libraries. Individual strains are grown, verified, and stored in glycerol stocks. Working plates are prepared with standardized inocula.
Stage two is media formulation. Stock solutions of carbon sources, nitrogen sources, trace elements, and melanin precursors are prepared. The robot combines these in varying ratios across 96-well plates according to experimental design matrices.
Stage three is inoculation. The robot transfers standardized inoculum to each well, seals plates with gas-permeable membranes, and initiates incubation.
Stage four involves monitoring. At scheduled intervals, the robot samples each well, transfers samples to measurement plates, and the plate reader records absorbance spectra. Data is automatically logged to cloud storage.
Stage five is harvest. At the end of the growth period, cultures are processed to extract melanin. Centrifugation separates cells from supernatant. Melanin is precipitated, washed, and resuspended for downstream assays.
Stage six is functional testing. Purified melanin samples are applied to reporter cells, exposed to radiation, and survival is measured. This directly tests radioprotective efficacy.
3.4 Quality Control and Reproducibility Automation eliminates many sources of human error but introduces its own quality considerations.
Positional accuracy is verified using calibration plates and dye distribution assays. The robot’s pipetting precision is checked regularly.
Evaporation control is critical for long-term experiments. Plate seals, humidified incubation, and edge effect compensation are implemented.
Cross-contamination between wells is prevented through tip usage protocols. Fresh tips for each transfer, appropriate blowout procedures, and randomized plate layouts minimize interference.
Biological replicates and technical replicates are built into experimental designs. Statistical power is calculated to ensure meaningful hit identification.
- ASSAY DEVELOPMENT 4.1 Melanin Quantification Quantifying melanin production requires distinguishing pigment from cell density and media components.
The primary measurement is absorbance at 405 nm, which correlates with melanin concentration across types. However, this measurement is confounded by light scattering from cells and absorbance by media components.
The solution is differential measurement. Wells are measured before and after centrifugation or filtration to separate cells and pigment. Alternatively, media-only controls enable background subtraction.
Spectral scanning provides additional information. The shape of the absorbance curve from 350 to 700 nm differs between melanin types. Eumelanin shows monotonic decrease with increasing wavelength. Pyomelanin may show distinct features. Machine learning approaches could classify melanin type from spectral data.
For absolute quantification, melanin standards are required. Commercial melanin from Sepia officinalis provides a reference, but microbial melanins may have different extinction coefficients. Parallel gravimetric analysis on selected samples establishes conversion factors.
4.2 Radiation Exposure Assays Testing radiation response requires controlled exposure and survival measurement.
For UV exposure, LED arrays integrated into the platform deliver precise doses. Dose-response curves are generated by varying exposure time or intensity. Survival is measured by comparing growth rates post-exposure to unexposed controls.
For ionizing radiation, external sources are required. Collaboration with facilities having X-ray or gamma sources enables this testing. Plates are irradiated in batches and returned to the platform for post-exposure monitoring.
An alternative approach uses chemical radiation mimetics. Compounds like bleomycin or hydrogen peroxide produce DNA damage similar to ionizing radiation. While not identical to true radiation exposure, these enable high-throughput screening without specialized facilities.
The key output is the radiation protection factor: the ratio of survival with melanin to survival without melanin under identical exposure conditions.
4.3 Functional Protection Assays The ultimate test of radioprotective melanin is whether it protects living cells from radiation damage.
The assay uses reporter cells, typically E. coli or yeast, that are easy to culture and measure. Melanin samples are added to reporter cell cultures, which are then exposed to radiation. Survival is measured by colony formation or growth rate.
Controls include no melanin, known protective compounds, and melanin from reference strains.
The assay must account for direct effects of melanin on reporter cells independent of radiation. Toxicity controls ensure that observed protection is not confounded by growth stimulation or inhibition.
For melanins that cannot be easily purified, co-culture assays can test whether producer cells protect non-producer reporters through shared melanin in the medium.
4.4 Bioprospecting Logic The screening strategy balances breadth with depth.
Primary screening tests many strains under standardized conditions to identify producers. This is qualitative or semi-quantitative, aimed at candidate selection.
Secondary screening tests selected strains under varied conditions to optimize production. Media components, inducers, and environmental parameters are systematically varied.
Tertiary screening performs functional radiation protection assays on purified melanins from top candidates. This identifies strains producing not just abundant melanin, but melanin with genuine radioprotective efficacy.
Final characterization includes spectral analysis, stability testing, and preliminary material property assessment.
- EXPECTED OUTCOMES AND APPLICATIONS 5.1 Deliverables The project will produce multiple concrete outputs.
First, a validated automated protocol for melanin screening, fully documented and open-source, enabling replication by other researchers.
Second, a database of microbial strains with quantified melanin production under defined conditions, including spectral profiles and growth characteristics.
Third, identification of top candidate strains producing melanin with demonstrated radioprotective activity, preserved as viable cultures and genomic DNA.
Fourth, characterized melanin samples from top candidates, with spectral data, stability profiles, and functional protection metrics.
Fifth, a roadmap for scaling from discovery to production, including genetic optimization strategies and fermentation development pathways.
5.2 Biotechnological Applications The discovered melanins have multiple potential applications.
In space exploration, melanin-based coatings could protect spacecraft surfaces and equipment from cosmic radiation. Lightweight and flexible, they offer advantages over metal shielding. Incorporated into fabrics, they could protect astronauts during spacewalks.
In nuclear facilities, melanin additives in paints and sealants could provide supplemental protection for workers and equipment. Biodegradable melanin films could simplify waste management.
In medicine, melanin formulations could be applied topically to protect skin during radiation therapy. Systemic delivery might protect healthy tissues during cancer treatment, though this requires extensive safety testing.
In consumer products, melanin could replace synthetic UV absorbers in sunscreens. Natural, biodegradable, and potentially more effective, melanin-based sun protection aligns with consumer demand for sustainable ingredients.
In electronics, melanin’s conductivity and radiation stability might enable novel components for extreme environments.
5.3 Scientific Contributions Beyond applications, the project advances fundamental science.
Understanding how melanin structure relates to radioprotective function guides rational discovery. By correlating spectral, chemical, and functional data across diverse melanins, we may identify which molecular features matter most.
The automated platform itself contributes methodology. Open-source protocols enable others to conduct similar screens, accelerating discovery across the field.
Characterizing melanins from underexplored microbial groups expands knowledge of natural diversity. Each new melanin type reveals evolutionary solutions to environmental challenges.
5.4 Commercial Potential The project has clear commercialization pathways.
Licensing top-producing strains to biotechnology companies for melanin production. Fermentation scale-up could supply material for multiple applications.
Developing melanin formulations for specific markets: sunscreens, industrial coatings, medical devices. Each application requires formulation optimization and regulatory approval.
Offering screening services to companies seeking customized melanins for specific applications. The platform can test customer strains or conditions under contract.
Selling characterized melanin samples as research reagents. The growing interest in melanin biology creates demand for well-defined reference materials.
- IMPLEMENTATION PLAN 6.1 Phase One: Platform Development The initial phase focuses on building and validating the automated platform.
Opentrons protocols are developed for basic liquid handling tasks: media preparation, inoculation, sampling. Each module is tested individually with dye solutions to verify accuracy.
Integration with the plate reader is established. Communication protocols ensure synchronized measurements and data transfer.
Quality control procedures are implemented. Acceptance criteria for pipetting precision, evaporation rates, and cross-contamination are defined.
A small test set of known melanin producers validates the complete workflow. Expected results confirm that the platform detects melanin production as anticipated.
6.2 Phase Two: Library Assembly Microbial strains are assembled from multiple sources.
ATCC provides authenticated reference strains including Streptomyces, Pseudomonas, and Bacillus species. Environmental isolates from local sources add diversity. Collaborations with other laboratories expand the collection.
Strains are archived in standardized format with barcoded tubes for automated retrieval. Working plates are prepared for screening.
Genomic DNA is prepared from each strain for future reference. Partial sequencing confirms identity and enables phylogenetic analysis.
6.3 Phase Three: Primary Screening The assembled library is screened under standardized conditions.
Each strain is grown in base medium and melanin production is measured over time. Producers advance to secondary screening.
Data is collected automatically and stored in a structured database. Production kinetics, final yields, and spectral profiles are recorded.
Hits are defined as strains producing melanin above a threshold, with preference for rapid production and desirable spectral characteristics.
6.4 Phase Four: Secondary Optimization Hit strains enter optimization screening.
Media components are varied systematically. Carbon source, nitrogen source, trace elements, and melanin precursors are tested individually and in combinations.
Environmental parameters including temperature, pH, and aeration are varied within practical ranges.
Inducers including oxidative stress agents, metal ions, and light are evaluated.
For each condition, melanin production is quantified and compared to baseline. Optimal conditions for each strain are identified.
6.5 Phase Five: Functional Validation Melanin from top producers under optimal conditions is purified and tested for radioprotective activity.
Purification methods are standardized. Cell disruption, solvent extraction, and precipitation steps are optimized for yield and purity.
Purified melanin is characterized by UV-Vis spectroscopy, FTIR for chemical groups, and elemental analysis.
Radioprotection assays using reporter cells are performed under controlled exposure conditions. Protection factors are calculated.
The best performers advance to final characterization and archiving.
6.6 Timeline Phase One is completed in months one through three. Hardware setup, protocol development, and validation occur during this period.
Phase Two occupies months four and five. Strain acquisition, verification, and library assembly proceed in parallel with ongoing protocol refinement.
Phase Three runs from months six through eight. Primary screening of the assembled library generates initial hit lists.
Phase Four extends from months nine through twelve. Hit strains are optimized through systematic condition testing.
Phase Five completes the project in months thirteen through fifteen. Functional validation identifies the most promising candidates for application development.
- RESOURCE REQUIREMENTS 7.1 Equipment The core equipment requirements are well-defined.
An Opentrons OT-2 robot with associated accessories including pipettes, tip racks, and deck modules. The high-throughput configuration with temperature control is preferred.
A plate reader with UV-Vis absorbance capability. Monochromator-based instruments offer flexibility for spectral scanning. Filter-based instruments require appropriate filter sets for melanin quantification.
Incubation and shaking capacity for multiple plates. Stacked incubators maximize throughput within limited footprint.
Centrifugation capability for processing samples. A plate centrifuge enables in-plate processing without transfer steps.
Standard microbiology equipment including biosafety cabinets, incubators, and freezers for strain maintenance.
7.2 Consumables Consumable requirements include 96-well plates appropriate for microbial culture, with gas-permeable seals for long-term incubation. Deep-well plates for reagent storage and mixing. Pipette tips in bulk quantities.
Media components including carbon sources, nitrogen sources, salts, vitamins, and specialized precursors. Melanin standards for reference.
Reagents for melanin extraction and purification including solvents, acids, and bases.
Assay reagents for functional testing including reporter strain media, viability indicators, and control compounds.
7.3 Strains and Biological Materials The strain collection requires acquisition from multiple sources.
ATCC strains are purchased with appropriate licenses. Expected cost is several thousand dollars for a diverse collection.
Environmental isolates require isolation and characterization effort. Collaboration with local microbiology groups can accelerate this process.
Control strains with documented melanin production are essential for assay validation.
7.4 Computing Resources Data management requires structured storage for experimental results. Cloud-based solutions enable remote access and collaboration.
Analysis pipelines process plate reader data, calculate production metrics, and identify hits. Python scripts handle routine processing.
Protocol development uses Opentrons software and version control. Protocols are documented for reproducibility.
7.5 Collaboration and Expertise Successful execution requires diverse expertise.
Microbiology expertise for strain handling and verification. Local collaborators can provide guidance.
Automation expertise for Opentrons programming and troubleshooting. Online communities offer support.
Data analysis expertise for interpreting screening results. Statistical methods identify meaningful differences amid experimental variation.
Radiation biology expertise for designing and interpreting protection assays. Collaboration with medical physics or radiation safety groups enables proper exposure experiments.
- RISK ASSESSMENT AND MITIGATION 8.1 Technical Risks The platform may fail to detect melanin production in some strains. Pigment may be cell-associated rather than secreted, requiring different measurement approaches. Alternative protocols using whole-cell measurements address this risk.
Contamination may compromise long-term experiments. Strict aseptic technique, antibiotic supplementation where appropriate, and frequent monitoring mitigate this risk.
Strains may not grow under standardized conditions. Flexible protocols accommodate different growth requirements through conditional media formulation.
Radiation exposure assays may be logistically difficult. Chemical mimetics provide a high-throughput alternative while collaboration for true radiation exposure is established.
8.2 Biological Risks Some melanin-producing strains are opportunistic pathogens. Risk assessment and biosafety level determination precede work with each strain. Appropriate containment and handling procedures are implemented.
Environmental isolates may include unknown organisms. Initial characterization identifies potential hazards before scale-up.
Genetic modification, if pursued later, requires additional biosafety consideration. The initial project avoids modification, focusing on natural diversity.
8.3 Timeline Risks Strain acquisition may face delays. Multiple sources and backup suppliers are identified.
Equipment delivery and setup may take longer than anticipated. Parallel activities advance other project components during waiting periods.
Unexpected technical challenges in protocol development require troubleshooting time. Modular development allows some work to proceed while issues are resolved.
8.4 Mitigation Strategies All major risks have mitigation plans.
For technical risks, pilot experiments with known producers validate each protocol module before full screening begins. Early detection of issues prevents wasted effort.
For biological risks, tiered containment approaches match handling procedures to risk level. Most strains require only basic biosafety precautions.
For timeline risks, critical path analysis identifies activities that cannot be delayed. Parallel work and flexible sequencing maintain progress.
- CONCLUSIONS This project designs an automated platform to discover microbial melanins with genuine radioprotective properties. By combining liquid handling robotics, spectrophotometric measurement, and functional radiation assays, the platform enables systematic exploration of microbial diversity that would be impossible manually.
The focus on function rather than organism ensures that the best producers are identified regardless of their biological classification. Bacteria, yeast, and other microbes are evaluated equally based on melanin production and radioprotective efficacy.
Applications span multiple industries. Space exploration needs lightweight radiation shielding. Nuclear facilities require durable protective materials. Medical applications demand biocompatible radioprotectors. Consumer products benefit from natural, sustainable ingredients.
The platform is designed for remote execution, making it accessible to researchers without local wet-lab infrastructure. Cloud laboratory deployment through Ginkgo Bioworks enables the project to proceed regardless of physical location.
The expected outcomes include validated protocols, identified high-performance strains, and characterized melanin samples ready for application development. Each outcome contributes to the ultimate goal: harnessing microbial melanins to protect against radiation across the contexts where it matters most.