Individual Final Project-COVALENT ORGANIC FRAMEWORK (COF) BASED APTAMER CLOSED LOOP SYSTEM TO COMBAT ANTIBACTERIAL RESISTANCE
PROJECT PROPOSALS





I am selecting the project 2 idea out of the three because I feel the antimicrobial resistance crisis is hot topic when healthcare is concerned.

COVALENT ORGANIC FRAMEWORK (COF) BASED APTAMER CLOSED LOOP SYSTEM TO COMBAT ANTIBACTERIAL RESISTANCE
ABSTRACT
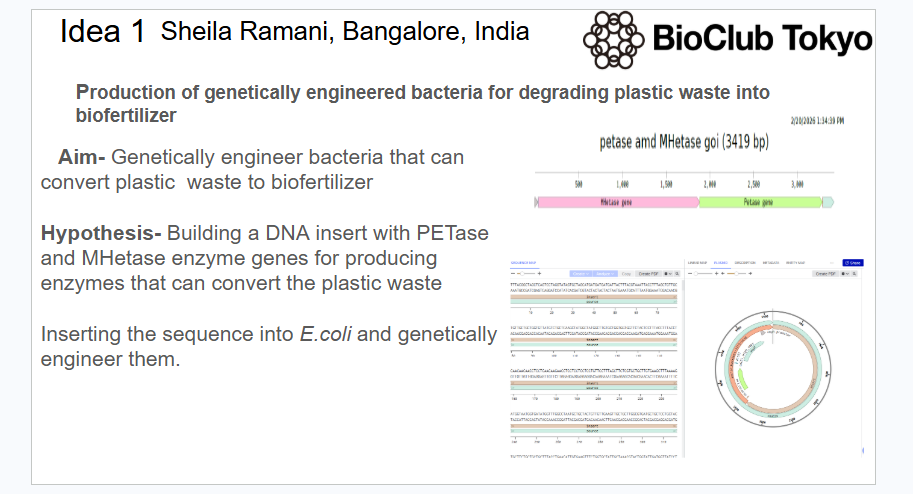
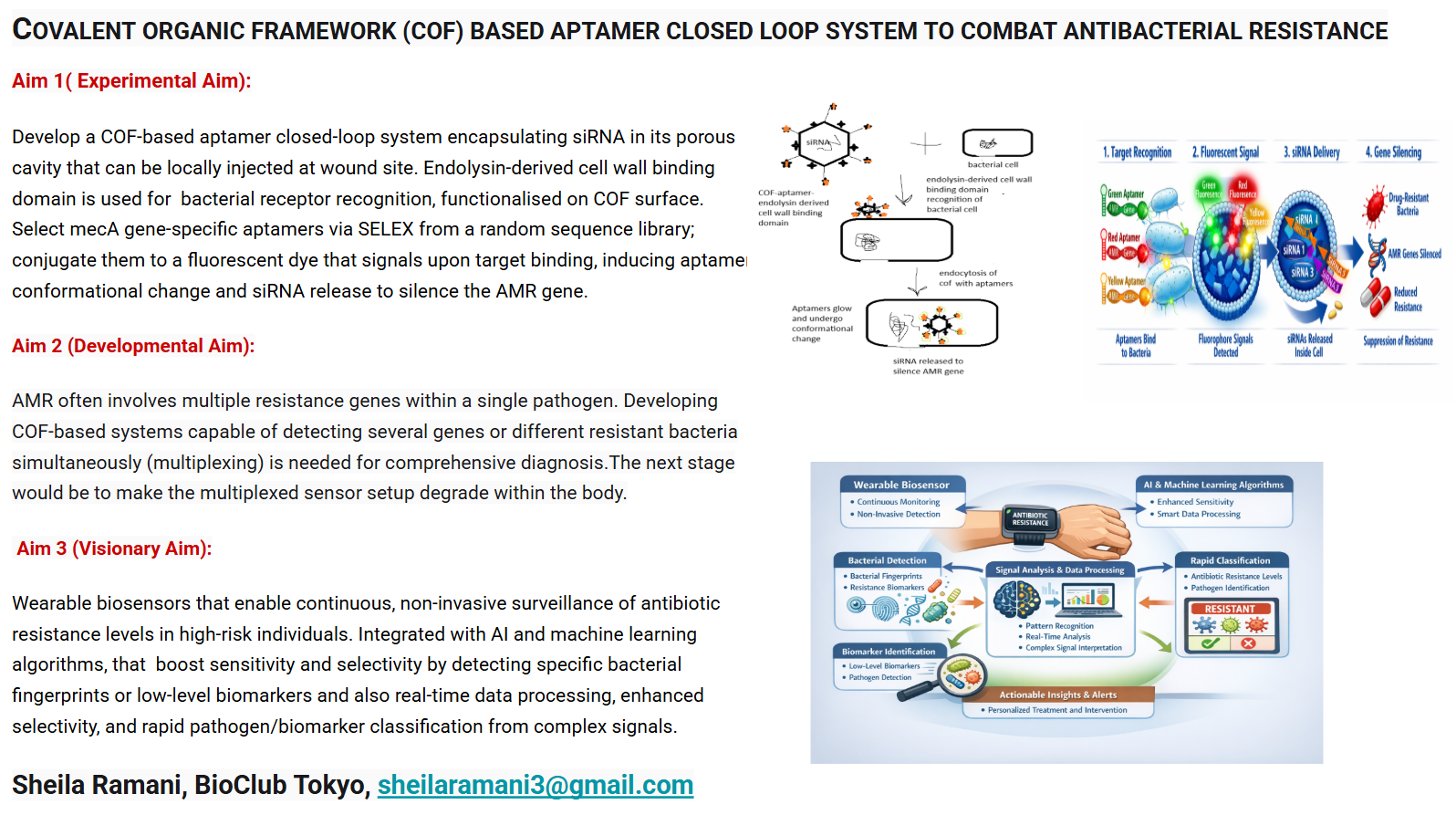
The antimicrobial resistance (AMR) is a global crisis in terms of healthcare that needs immediate attention. The misuse and overuse of antibiotics for various purposes has led to teh organsims developing resistance to it. Recently, biosensor mediated drug delivery systems have come into foreplay to combat AMR, also called as closed loop systems. These systems detects the presence of AMR-producing bacteria, proteins or AMR genes itself in a sample (e.g., wastewater or clinical sample, site of wound) but also triggers a response—such as releasing a neutralizing agent or drug or activating a downstream warning system. In this study a covalent organic framework(COF) functionalised with endolysin derived cell wall binding domain (CBD) binding to receptors and aptamers specific for the mecA genes(S.aureus) of AMR bacteria and siRNA within its cavity based biosensor is injected at the site of infection. The aptamer conjugated to a fluorescent dye fluoresceses on target binding and aptamer conformational change. This triggers the COF to release the siRNA that neutralizes the AMR gene. The biosensor aims to generate a highly sensitive, specific, rapidly detecting system that not only detects the biomarkers but also instantaneously produces a feedback treatment mechanism. The system can be used to detect proteins and toxins produced by the resistant bacteria by replacing the specific aptamers for the specific targets.
PROJECT AIMS
Aim 1: Experimental Aim (this project):
The main aim of this project is to create a COF based aptamer closed loop system that senses the receptor of bacteria by endolysin-derived cell wall binding domain on COF, mec A genes in the bacteria detected by fluorescence aptamer at the site of infection and release the siRNA enclosed within the COF and silence it.The device created would be injected at the wound site.
The CBD is functionalized on COF to make it target the bacterial receptor.The random sequence library can be commercially ordered and PCR reactions for the enriched sequences can be carried out by opentrons.Aptamer selection by SELEX(Systematic Evolution of Ligands by Exponential Enrichment) and its conversion to a fluorescent aptasensor can be done by opentrons automated systems. The functionalization of COF surface with functional groups for immobilization of aptamers and immbolization can also be done using liquid handling robots to minimise human errors.
In Vitro Transcription (IVT) is a highly efficient used for generating small interfering RNA (siRNA) designed to target antimicrobial resistance (AMR) genes in bacteria, these are encapsulated in the porous cavity of COF.DNA templates, usually derived from PCR products encoding the AMR target is used This is a cell free alternative to chemical synthesis and cost effective.COF is stabilized with a biocompatible coating (e.g., PEGylation) to improve dispersibility and reduce immune clearance, especially for intravenous injection.The functionalized COF-aptamer-siRNA complex is injected locally to the infection site, allowing the aptamers to bind to the targets and the COF to deliver the siRNA payload.
Aim 2: Development Aim:
COFs are quite robust but biological systems contain complex substances that can cause biofouling of the biosensor surfaces. these can lead to false positives, can be reduced by antifouling coatings.These could be silicone based polymers or fluoropolymer coatings.
AMR often involves multiple resistance genes within a single pathogen. Developing COF-based systems capable of detecting several genes or different resistant bacteria simultaneously (multiplexing) is needed for comprehensive diagnosis.The next stage would be to amke the sensor setup degrade within the body.
Aptamers that are more stable to nucleases should be created to prevent their degradation in vivo. These can eb done by backbone modifications by phosphothioate, conjugation with polyethylene glycol.
Most sensors are tested in laboratory buffers or spiked samples. Extensive validation in real, large-scale patient cohorts is necessary to meet regulatory standards.
Aim 3: Visionary Aim:
Wearable biosensors can provide continuous, non-invasive surveillance of antibiotic resistance levels in high-risk individuals.These biosensors can be integrated with AI and machine learning based algorithms.AI improves sensitivity and selectivity by detecting specific bacterial fingerprints or low-level biomarkers.Enables real-time data processing, enhanced selectivity, and rapid pathogen/biomarker classification by interpreting complex signals.Enhancing the antifouling properties of the COF surface is essential to ensure consistent performance in clinical samples.Developing cost-effective and scalable synthesis methods for COFs is necessary for commercial adoption.Most of these biosensors are in very nascent stage they have to undergo validation in real complex biological matrices.

BACKGROUND
The project is novel as it involves the use of COFs with DNA aptamers specific for the AMR genes itself.The integration of COFs with aptamers to construct fluorescence sensors is a fast-growing area focused on overcoming limitations of previous generation biosensors (like low sensitivity or high background noise).The components (aptamers and COFs) are not new, their combination in biosensors is a recent, rapid, and developing (“emerging”) trend that offers “novel” sensing capabilities.These systems shorten the detection time from days (conventional culture-based techniques) to hours.
Diagnostics and therapy are separate steps in existing paradigm. The COFs act as a “theranostic” platform, where the system not only detects AMR genes (diagnosis) but can be engineered to deliver drugs or siRNA to the site of infection (therapy). This “closed-loop” system means the detection can immediately initiate a treatment, potentially reversing resistance.Modification of COFs to release drugs upon binding, the closed-loop system acts only in the presence of the resistance gene, minimizing off-target effects and reducing the risk of further resistance development.
In summary, the COF-based aptamer system moves the field from existing labor-intensive, slow phenotypic tests towards an automated, rapid, and precise molecular diagnostic and potential treatment modality.
PROJECT IMPACT
The project attempts to solve the growing global crisis of antimicrobial resistance due to misuse and overuse of antibiotics in humans and animals and agriculture. This has led to the development of drug resistance organisms or “superbugs”. This is making it difficult to treat cancer, infections and surgeries. Rapid, on-site diagnostics (Point-of-Care) reduce the reliance on broad-spectrum antibiotic therapy, directly combating the over-prescription that leads to the emergence of superbugs.The implementation of rapid diagnostics can decrease hospital stays, minimize the need for expensive second-line therapies, and reduce overall healthcare burdens.They allow faster identification of AMR and immediate treatment can be given reducing mortality rates.
The COF and aptamer based closed loop systems being very specific to AMR genes may identify low-abundance AMR genescontributing to our overall knowledge of the “resistome”. The suystem allows scientists to analyse the expression of AMR genes in real-time within complex samples.
A Covalent Organic Framework (COF)-based aptamer closed-loop system for antimicrobial resistance (AMR) genes promises a paradigm shift from traditional, slow diagnostic methods to rapid, on-site, and actionable “sample-to-answer” platforms.In summary, this technology changes the approach to AMR from reactive, slow identification to proactive, rapid, and tailored intervention, enhancing “One Health” approaches spanning clinical care, food safety, and environmental surveillance.
ETHICAL IMPLICATIONS
COFs are innovative, but their long-term biocompatibility and biodegradation in human or environmental systems are still being investigated. A false positive could lead to unnecessary use of antibiotics (speeding up resistance), while a false negative could lead to inadequate treatment and death. Rigorous safety testing must ensure that the COF carrier does not cause toxic effects in patients. It also mandates that the automated system has built-in checks to prevent wrongful or lethal treatment protocols, particularly if the sensor makes a mistake.The technology should be developed with a strategy for equitable distribution and affordability to ensure that benefits are not limited to wealthy populations, as AMR is a global crisis.
Developing stable, biocompatible COFs that can effectively immobilize aptamers. This often involves chemical processes to ensure the aptamers remain attached and functional on the framework’s surface.Establishing rigorous protocols for the containment and disposal of synthetic materials used in sensing to prevent environmental contamination. Targeted detection and subsequent localized treatment strategies must be carefully managed to avoid putting selective pressure on bacterial populations, which could inadvertently lead to the emergence of new resistance mechanisms.In clinical or environmental applications, the accumulation of organic matter on the sensor surface (biofouling) can interfere with the aptamer’s ability to bind to its target, reducing the system’s reliability over time.Portable “closed-loop” detection system (combining sensing with potential automated intervention) is essential for limiting the emergence and spread of AMR,
The assumption that the presence of an AMR gene (detected by the system) does not always equate to phenotypic resistance, as the gene might be inactive, damaged, or not expressed. This could lead to unnecessary or incorrect therapeutic decisions.The altrrnative proposals, if an AMR gene is silent, using a combination approach with weaker antibiotic and membrane permeabiliser can be effective.
EXPERIMENTAL DESIGN, TECHNIQUES, TOOLS, AND TECHNOLOGY
Aim 1 — DNA construct for mec A mRNA
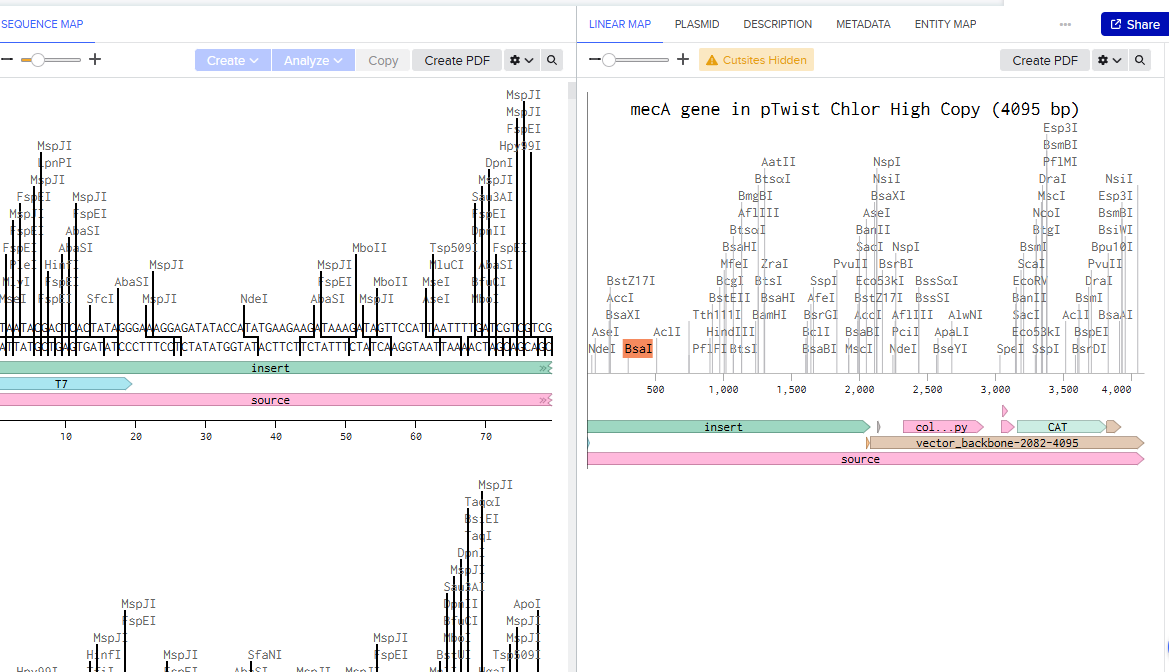

Benchling screenshot of the mec A gene.
This DNA construct is used for the next round of SELEX for aptamer selection, used as bait and also in in vitro validation of the system.

Benchling screenshot of the pTwist plasmid with mec A gene
mecA gene sequence: S. aureus MRSA252 (GenBank BX571856)
mecA CDS region used for siRNA target selection (nt 1–2700 of 2070 nt CDS)
Source: NCBI NC_002952.2, mecA locus
ATGAAAAAGATAAAAATTGTTCCACTTATTTTAATAGTTGTAGTTGTCGGGTTTGGTATATAAAATATT" “TCTAAATTTCAATTAGATTCTATTATTTTTAACAACGATTTTTCTAGCATGTGCAAAGAATATTTCTAC” “AGGAAATAAAGATGTTTATGAAGGTACTGAGCTATTTCCAAATGGTGTTTATAAAGATTTAGAAGAAAT” “GAATGCAGAAATTAGAGTTGTTTATCCAGATAAAGTTGTTAATGGTATAGATGCAACTACAAATGCTGT” “TGTTAAAAATGTTGATTATAAAGATAATGACAAAGACTTAAAAACAGCTATTAATGTTCCAACAAATAC” “AGCTCCTGTTCAAGCTGCTAAAGCTGAAAGCGATCTTGAAAATTTAACAACAGCTAATAATAAAGTTCC” “AGCTCAAGCTGCAAAAGCTGAAAGCGATCTTGAAAATTTAACAACAGCTAATAATAAAGTTCCAGCTCA”
Timeline-Order from Twist Bioscience (twistbioscience.com) as a synthetic gene fragment. Turnaround 7–10 days.
Pricing- Academic pricing ~£40–50
Aim 2 — Make mecA mRNA by In Vitro Transcription (IVT)
Resuspend DNA construct at 100 ng/uL in 10 mM Tris pH 8.0 (RNase-free).
Set up HiScribe T7 High Yield RNA Synthesis reaction (NEB E2040S, 20 uL): 500 ng DNA + 2 uL each NTP (ATP, CTP, GTP, UTP) + 2 uL T7 RNA Polymerase Mix + RNase-free water to 20 uL. Incubate 37°C, 2 h.
Add 2 uL TURBO DNase; incubate 37°C, 15 min to remove DNA template.
LiCl precipitation: add supplied LiCl solution; mix; -20°C, 30 min; centrifuge 12,000g, 15 min, 4°C; discard supernatant; wash pellet with ice-cold 70% ethanol; air-dry 5 min; resuspend in 20 uL RNase-free water.
Quantify: NanoDrop (A260/A280 >1.8). Integrity: denaturing 1% agarose gel — single band at ~600 nt. Yield: 50–100 ug. Aliquot 5 uL; store -80°C. Do not freeze-thaw.
Timeline- 1 day
Expected Result- Appearance: A sharp, distinct single band at the ~600 nt mark.
Pass Criteria: There should be no visible “smearing” below the band (which would indicate RNase degradation) and no high-molecular-weight bands at the top (which would indicate incomplete DNase digestion of the template). A clear to white, translucent pellet at the bottom of the tube is observed.
Aim 3 — Select mecA RNA-Specific Aptamer by SELEX
In silico mec A RNA specific aptamer
The link below is the colab notebook documentation I used to generate aptamers specific to mec A RNA target. https://colab.research.google.com/drive/12EsVyc_clGmh9xX_YXl2sWMD3ba6tXsB?usp=drive_link
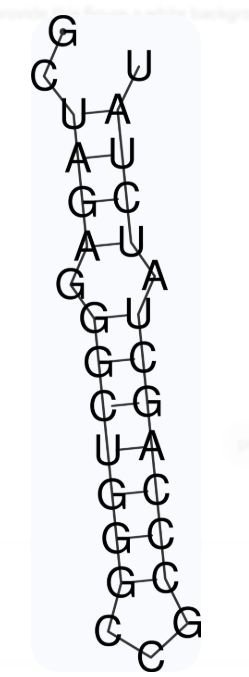
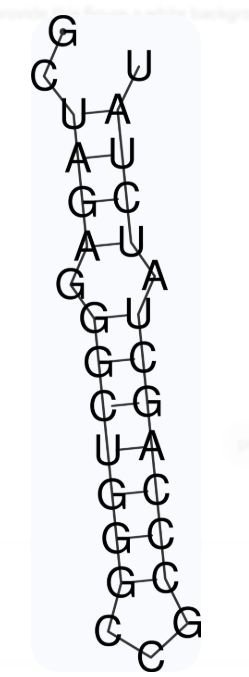
This is the best aptamer candidate prediction
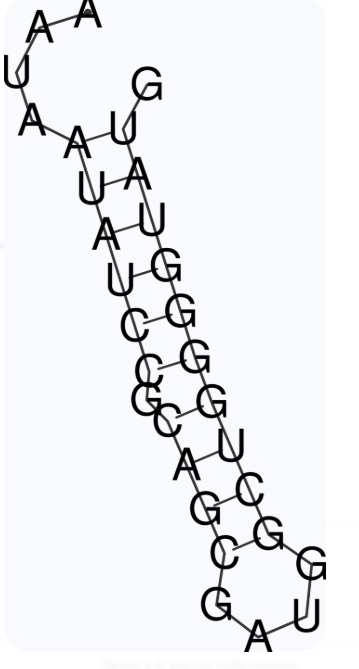

The second best aptamer candidate prediction
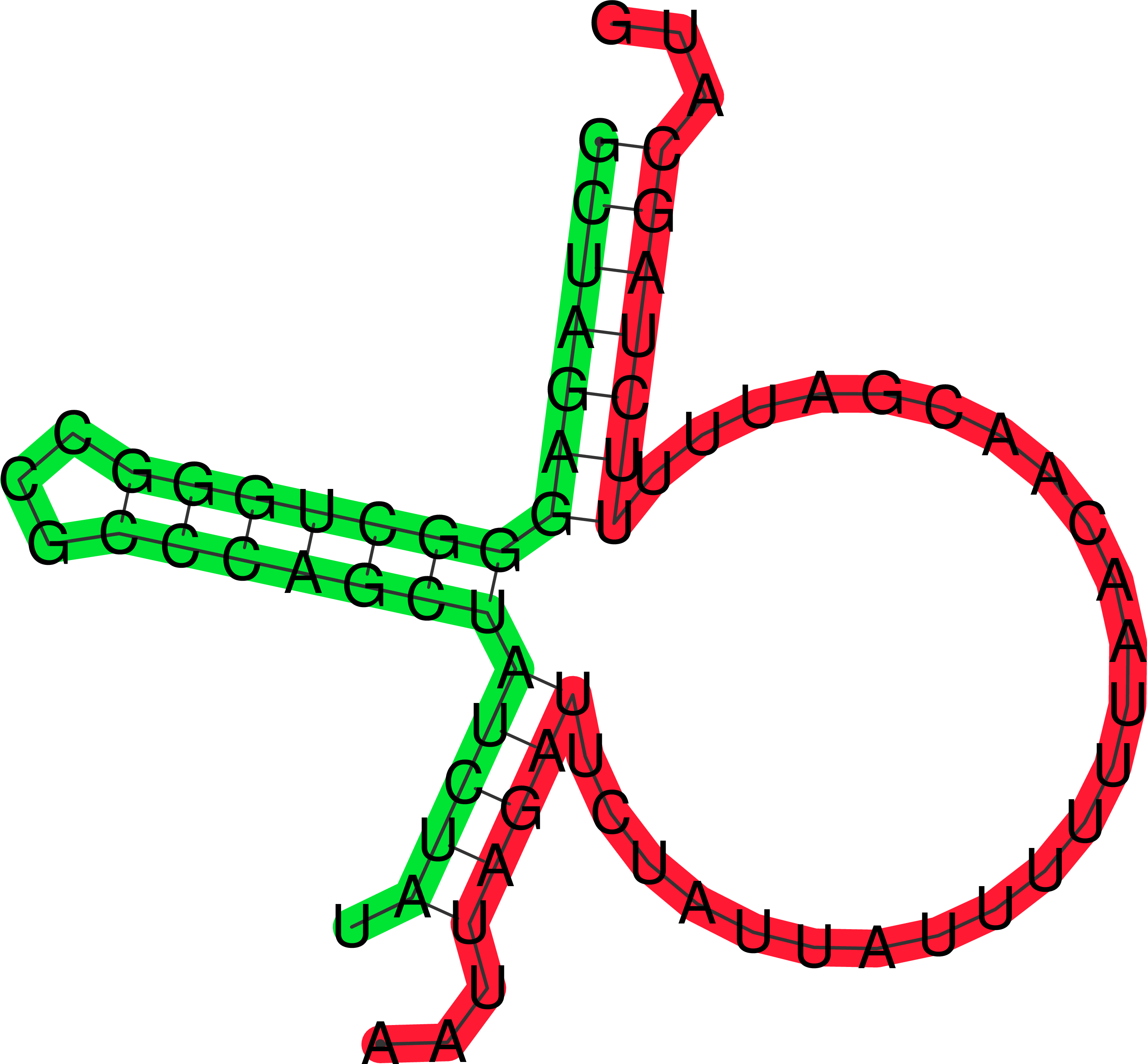

The aptamer-mec A mRNA cofolded structure prediction.
These were generated using ViennaRNA and RNAfold and RNA cofold tools. These structures are not enough they need experimental validation that can be done by SELEX.
Aptamers are selected in vitro, eliminating the need for animals to produce antibodies reducing animal usage. Generate DNA aptamers specififc to the mec A RNA by SELEX and immobilize it onto the functionalized COFs.
Library generation: Order the single-stranded DNA (ssDNA) library from a reputable supplier (e.g., IDT, TriLink, Sigma).
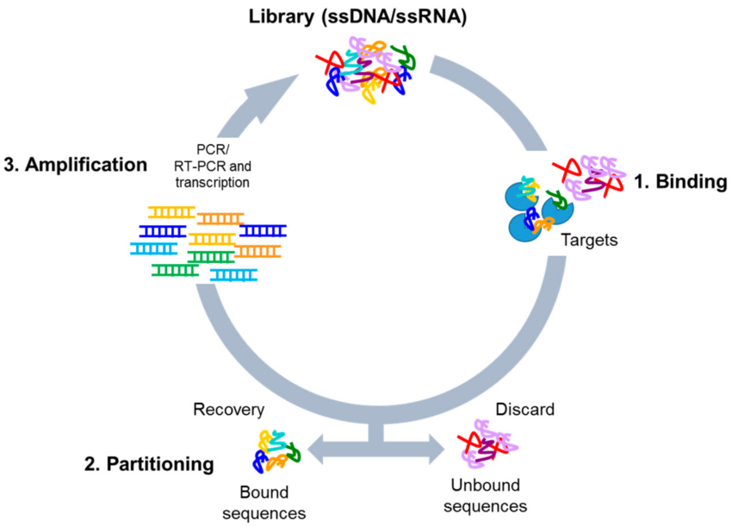

Aptamer Development (SELEX): A large library of random sequences is used to screen for specific binding to a target. This process can be carried out by Opentrons.Opentrons Flex and the older OT-2, are used to automate the SELEX (Systematic Evolution of Ligands by Exponential Enrichment) process, enabling high-throughput, reproducible, and automated generation of DNA/RNA aptamers. The automation covers critical steps such as incubation, washing, binding, and elution, reducing human error and increasing the speed of aptamer discovery. This increases binding affinity,improve specificity, removing sequences that bind to non-target components. PCR/RT-PCR amplification of the bound sequences is carried out automatically to generate the pool for the next round.Flex robot allows for 96-channel pipette usage (with partial tip pickup), allowing for faster processing of libraries compared to manual methods.
Fluorescent functionalization involves labeling these selected aptamers with fluorophores.
Expected Result
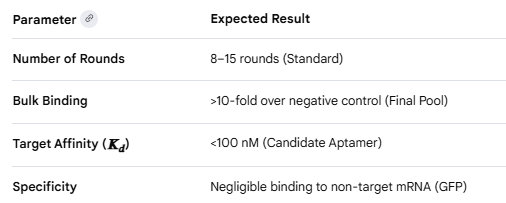

Aim 5 — Design and Order siRNA


siMecA-1 targets nt 312–332 in the early CDS — this region is consistently accessible (low predicted secondary structure) and scores 7/8 on Reynolds criteria. The 2’-OMe modifications at alternating positions on the antisense strand are important for your system specifically: wound fluid is rich in nucleases and will degrade unmodified siRNA within hours. The phosphorothioate overhangs add an extra layer of stability without interfering with RISC loading.
mecA CDS nt 312–332 (early coding sequence, accessible region)
Sense (guide): 5'—GGAAAUAAAGAUGUUUAUGAA—dTdT—3'
Antisense (pass): 5’—UUCAUAAACAUCUUUAUUUCC—dTdT—3'
Timeline- 1 week
Expected Result-The expected result for the Design and Order siRNA step is the acquisition of validated, high-purity duplexes ready for experimental use.
Aim 6 — Synthesise the COF Nanoparticle
Materials
• TAPB: 1,3,5-tris(4-aminophenyl)benzene — 0.1 mmol (Sigma 805874)
• BTCA: 1,3,5-benzenetricarbaldehyde — 0.15 mmol (Sigma 399663)
• 1,4-Dioxane (anhydrous) + 6 M acetic acid (catalyst); sealable Pyrex tube
Protocol
Dissolve TAPB + BTCA in 4.5 mL dioxane + 0.5 mL 6 M acetic acid. Degas: 3 freeze-pump-thaw cycles. Seal under vacuum.
Heat at 120°C, 72 h (no stirring). Yellow precipitate = COF forming.
Centrifuge 10,000g, 20 min. Wash: DMF (3×), THF (2×), acetone (2×), deionised water (3×). Dry at 80°C vacuum, 24 h.
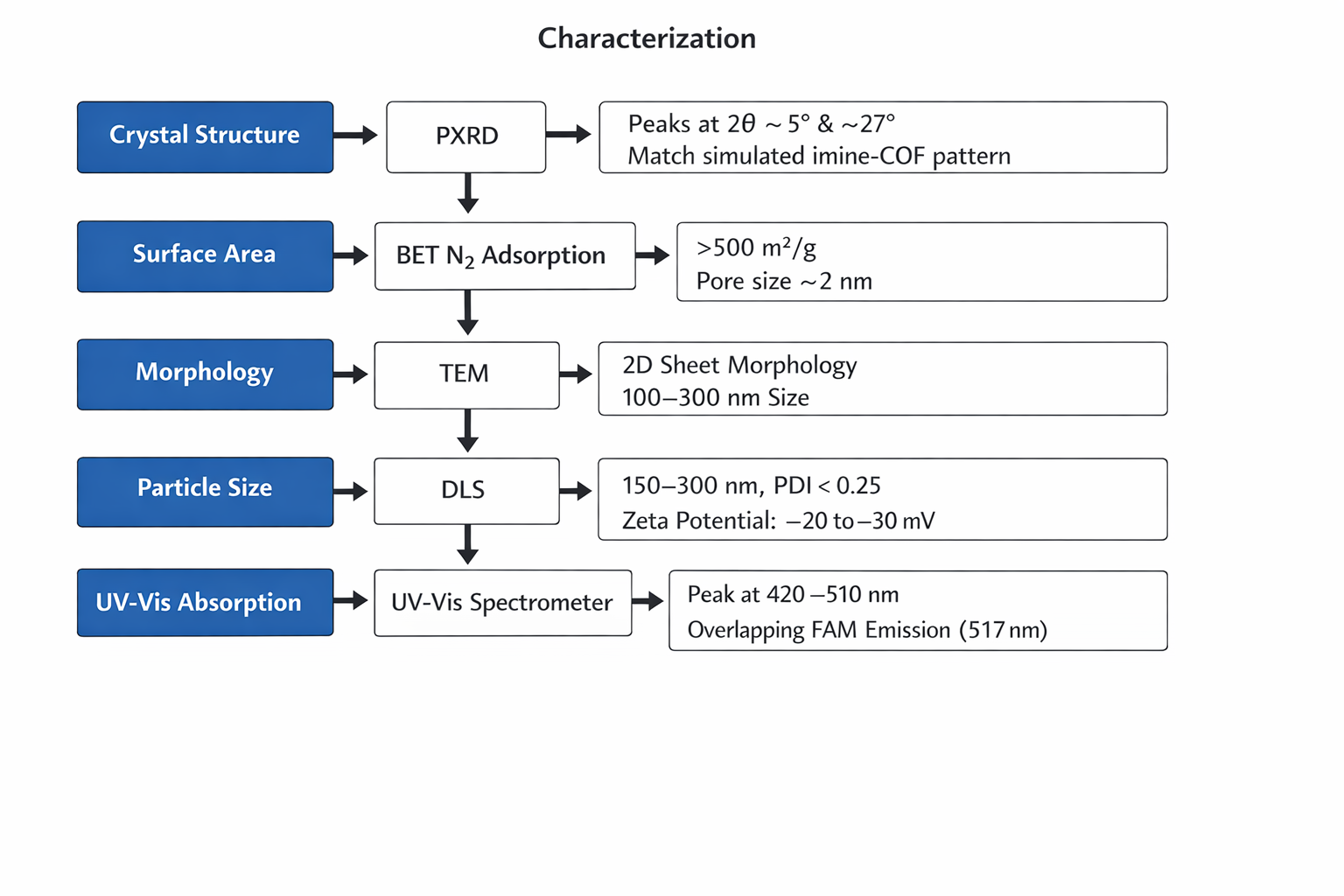

Figure created by copilot
Timeline- 4-5 days
Aim 7 — Produce and Purify LysK-CBD
LysK is the endolysin from staphylococcal phage K. Its C-terminal cell wall binding domain (CBD, ~120 aa) binds WTA (wall teichoic acid) on the S. aureus surface with high specificity. It does not bind Gram-negative bacteria, human cells, or most non-staphylococcal Gram-positive bacteria. Only the CBD is expressed here — not the lytic enzymatic domain — so the construct does not lyse bacteria, only binds them.
Aim 7.5— Express CBD in E. coli
Synthesise codon-optimised LysK-CBD gene (aa 479–593 of LysK, UniProt Q9KLD5) with N-terminal 6×His tag and TEV protease site.
Clone into pBR322(+) via (Golden Gate assembly). Transform into BL21(DE3) competent E. coli.
Express: grow at 37°C to OD600 = 0.6; add IPTG (0.5 mM); shift to 20°C; express overnight (16–18 h).
Lyse cells: French press or sonication in lysis buffer (50 mM NaH2PO4, 300 mM NaCl, 10 mM imidazole, pH 8.0) + protease inhibitors.
Ni-NTA affinity purification: load lysate onto Ni-NTA agarose; wash (20 mM imidazole); elute (250 mM imidazole). Dialyse into PBS pH 7.4. Concentrate to 5–10 mg/mL by Amicon Ultra 10 kDa.
Validate: SDS-PAGE (expected band: ~15 kDa). Western blot with anti-His antibody. Yield: 5–15 mg/L culture.

Figure created by copilot
CBD specificity validation (do before conjugation)
Coat wells of a 96-well plate with heat-killed MRSA (10^8 CFU/mL in PBS, 60°C, 1 h). Also coat with heat-killed MSSA, E. coli (negative control), and BSA (background control).
Add His-CBD (100 nM) to each well; incubate 1 h, RT; wash 3× PBS; detect with anti-His-HRP antibody (1:5000); TMB substrate; read OD450.
Pass: MRSA OD450 >5-fold above BSA background. E. coli signal <1.2-fold above BSA (species specificity confirmed).
Timeline- 1-2 weeks
Aim 8 — Conjugate CBD to COF Surface
Protocol
Activate CBD carboxyl groups: add EDC (2 mM) + NHS (5 mM) to CBD solution (1 mg/mL) in 50 mM MES pH 6.0. React 15 min, RT. Remove excess by Zeba desalting spin column (7 kDa MWCO).
Resuspend bare COF (2 mg/mL) in PBS pH 7.4. Add NHS-activated CBD at mass ratio 1:2 (CBD:COF). React 2 h, 4°C, gentle rotation.
Quench: ethanolamine to 10 mM final, 30 min RT.
Purify: centrifuge 12,000g, 15 min; wash 3× PBS to remove unbound CBD. Measure CBD in supernatant by BCA protein assay. Conjugation efficiency (CE%) = (CBD_added - CBD_in_supernatants) / CBD_added × 100. Target: >60%.
Validate CBD-COF binding to S. aureus
- Incubate CBD-COF (200 nM) with live MRSA (10^8 CFU/mL) vs E. coli (same CFU) in PBS for 30 min at 37°C.
- Centrifuge to pellet bacteria + bound COF. Wash 3× PBS. Measure FAM fluorescence of pellet vs supernatant. Higher FAM in MRSA pellet vs E. coli pellet confirms specific CBD-mediated binding.
- Also: flow cytometry or confocal imaging — COF-bound bacteria should show FAM fluorescence. E. coli should not.
Assembly order — critical
Correct assembly order: Step 8: CBD conjugated to COF FIRST (before siRNA loading) Step 9: siRNA loaded into CBD-COF pores Step 10: Aptamer conjugated to CBD-COF surface (after siRNA loading)
Reason: CBD conjugation (Step 8) uses EDC/NHS at pH 6 which can be harsh. Running this before siRNA loading prevents siRNA displacement. Aptamer conjugation (Step 10) is mild and safe after loading.
Chemistry CBD carboxyl groups (Asp/Glu residues) react with primary amines on the COF surface via EDC/NHS coupling, forming stable amide bonds. Use a site-directed approach if possible: activate CBD carboxyls in buffer away from the His-tag to preserve binding activity of the CBD domain.
Timeline- 2 days
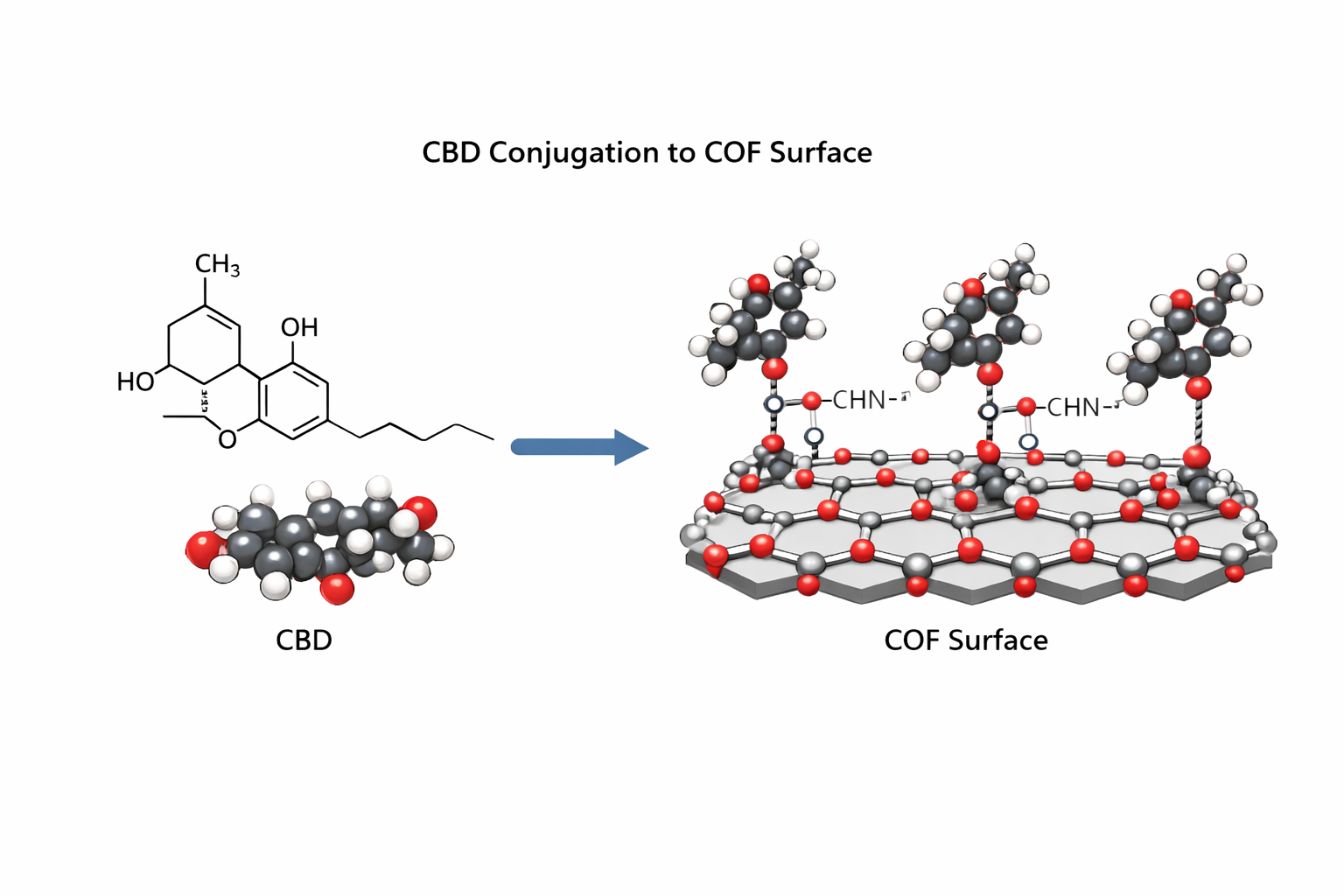

Figure created by copilot
Aim 9 — Load siRNA into CBD-COF Pores
Protocol
Resuspend CBD-COF at 2 mg/mL in RNase-free PBS pH 7.4. Sonicate 10 min.
Anneal siMecA-1: mix sense + antisense (equal molar), 95°C 5 min, cool to RT over 1 h.
Add annealed siRNA to CBD-COF at mass ratio 1:10 (siRNA:COF). Incubate 4°C, 2 h, rotation.
Centrifuge 12,000g, 15 min. Measure siRNA in supernatant (UV-Vis, A260). Calculate EE% = (siRNA_added - siRNA_supernatant) / siRNA_added × 100. Target: >85%.
Wash pellet 2× PBS pH 7.4. Resuspend in RNase-free PBS at 2 mg/mL.
pH-triggered release validation
Incubate aliquots at PBS pH 7.4 vs acetate buffer pH 5.5, 37°C. Measure released siRNA at 1, 4, 8, 24 h
Pass: <15% release at pH 7.4; >70% release at pH 5.5 at 24 h. Release ratio >5:1.
Mechanism: imine (Schiff base) linkages in the COF backbone hydrolyse at pH 5.5 (infected wound acidity), expanding pore structure and releasing siRNA. Stable at pH 7.4. This is the pH safety gate — siRNA only releases where MRSA is actively infecting.
Timeline- 1 day
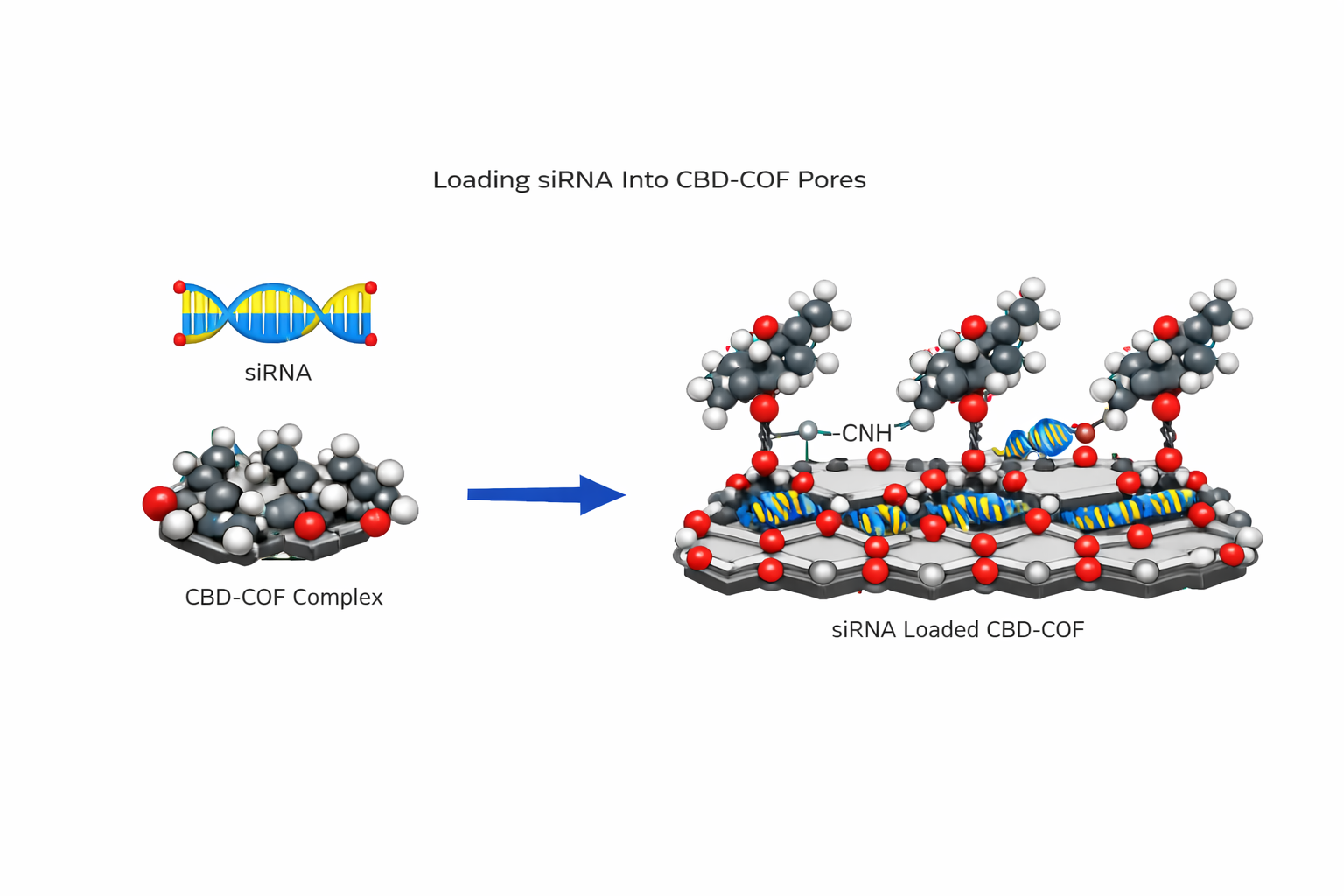

Figure created by copilot
Aim 10 — Conjugate Aptamer to CBD-COF Surface
Protocol
Activate CBD-COF surface amines: EDC (2 mM) + NHS (5 mM) in 50 mM MES pH 6.0. React 15 min, RT, rotation.
Centrifuge 12,000g, 10 min; discard supernatant; resuspend in PBS pH 7.4.
Add mecA structure-switching aptamer (3’-C6-NH2) at molar ratio 1:200 (aptamer:COF surface sites). React 2 h, 4°C, rotation.
Quench: ethanolamine 10 mM, 30 min RT.
Purify: Amicon Ultra 100 kDa spin column (14,000g, 3× PBS washes). Free aptamer passes through; complete construct retained. Calculate CE% from aptamer in filtrate. Target: >70%.
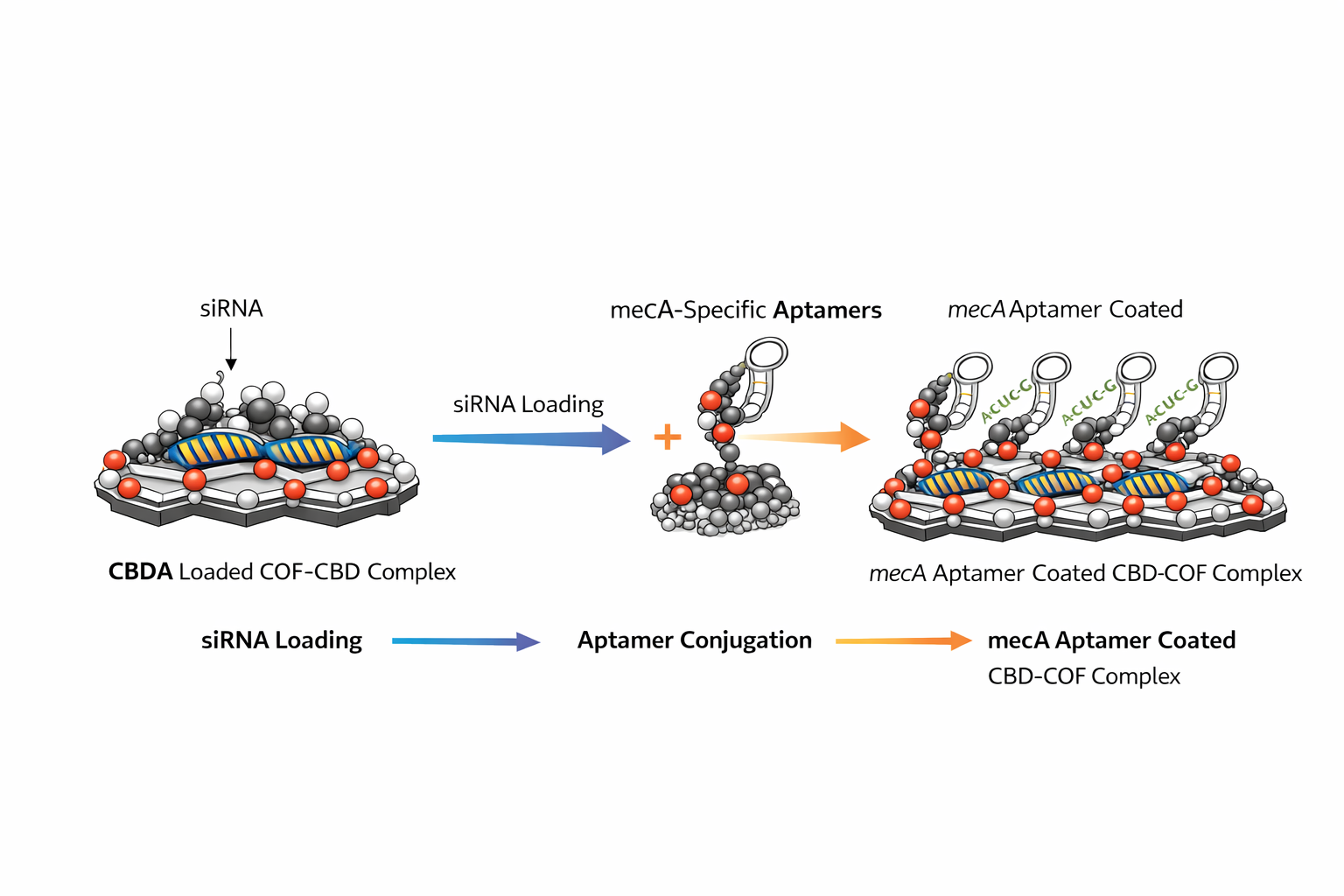

Figure created by copilot
Characterization:
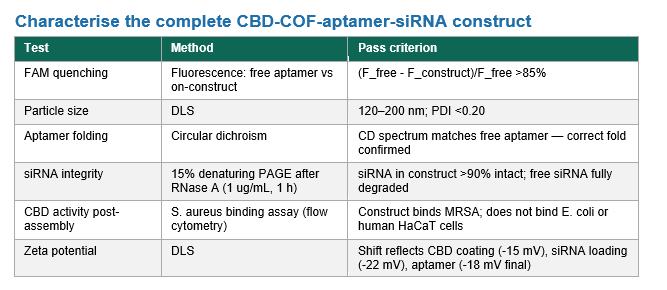

Timeline- 2 days
Aim-11 In vitro validation
11a — Fluorescence switching assay
96-well black plate. 100 nM complete construct in selection buffer, 37°C.
Add IVT mecA mRNA at 0, 0.1, 1, 10, 100, 500 nM. Incubate 37°C, 1 h.
Measure FAM fluorescence every 15 min (Ex 492, Em 517 nm).
Controls: non-mecA mRNA (GFP IVT); non-targeting aptamer construct + mecA mRNA; blank.
Pass: >5-fold signal increase at 10 nM mecA mRNA. <1.5-fold with non-mecA mRNA.
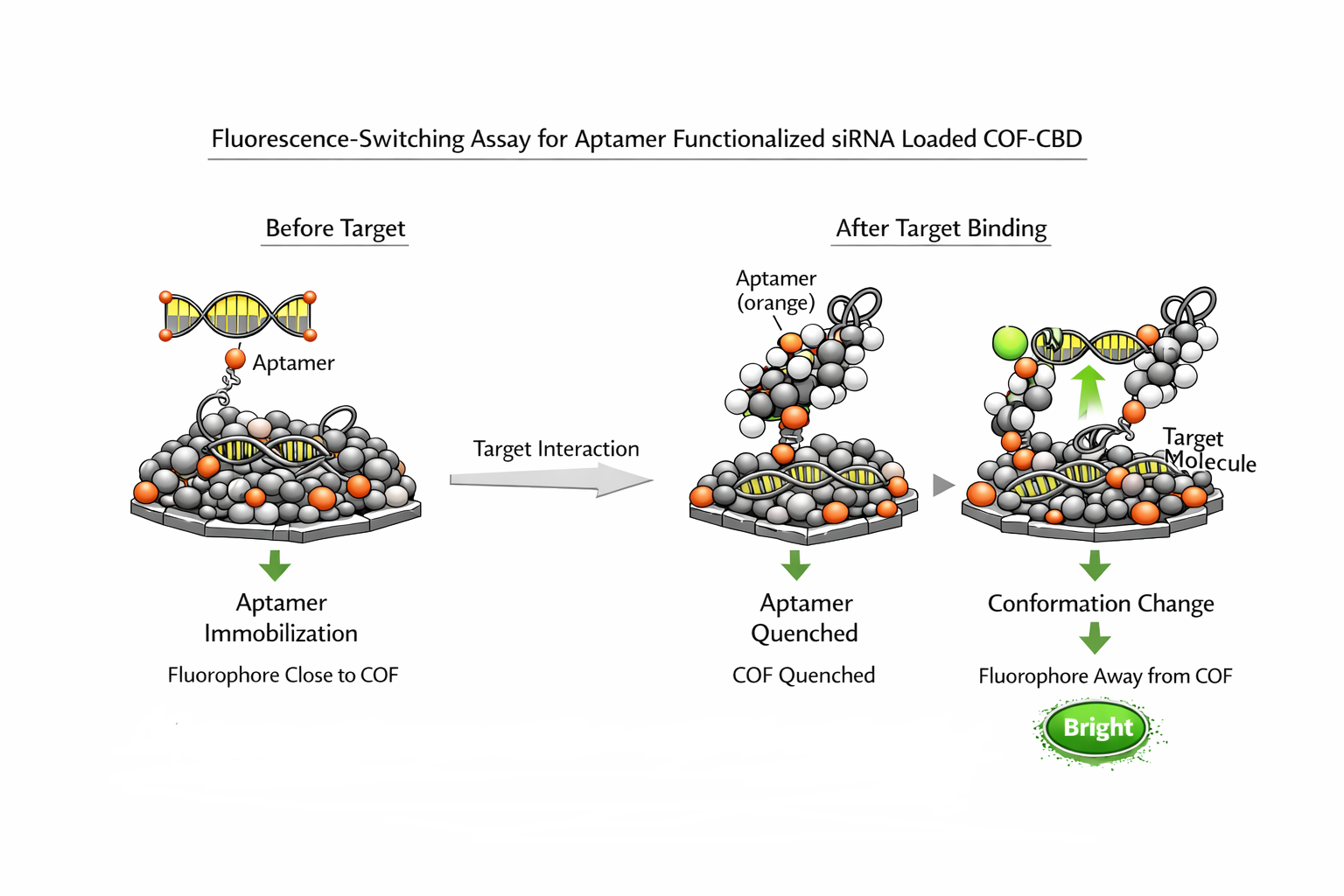

Figure created by copilot
Workflow:
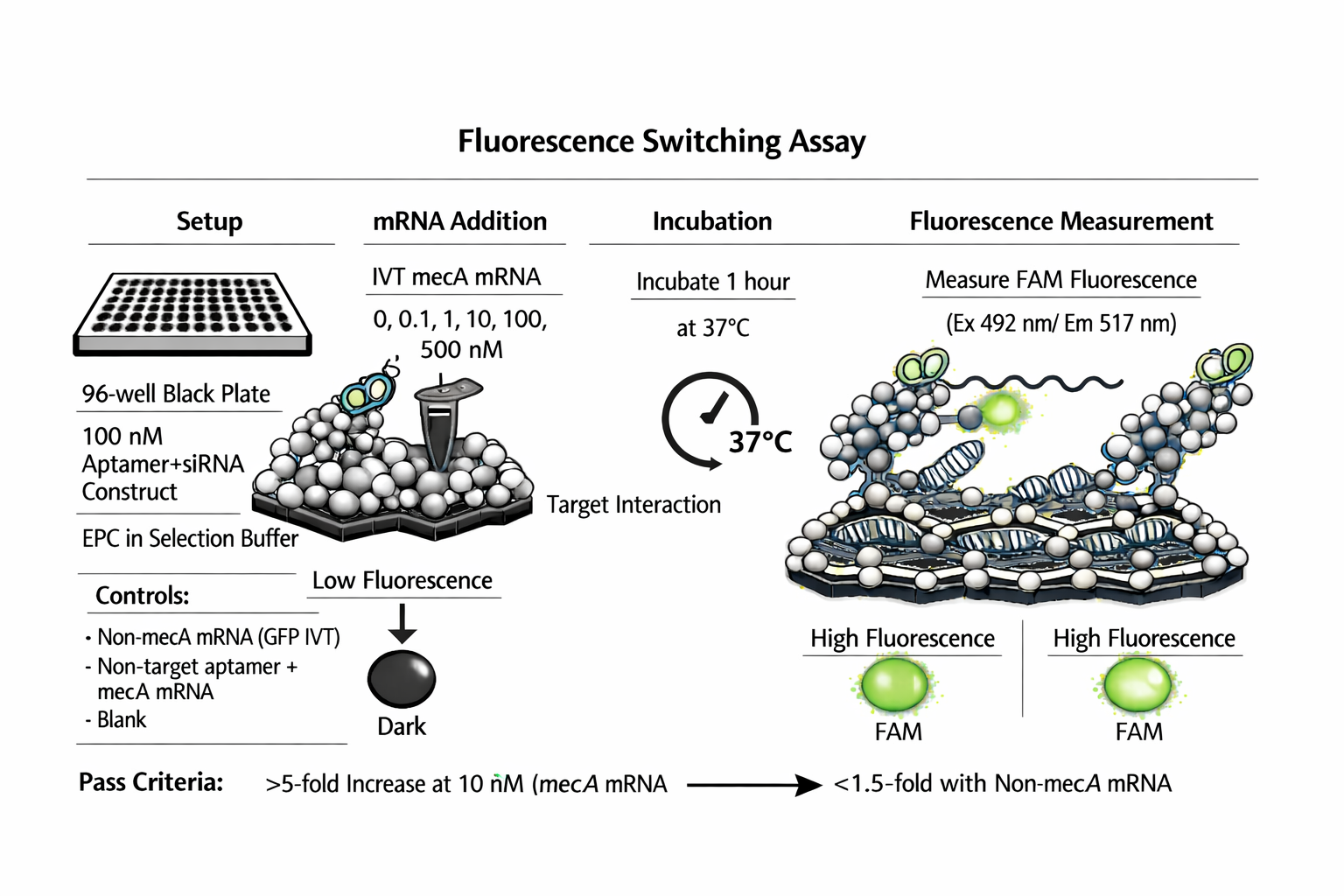

Figure created by copilot
11b — CBD binding specificity to S. aureus
Mix 100 nM complete construct with: (a) MRSA ATCC 33591; (b) MSSA ATCC 25923; (c) E. coli ATCC 25922; (d) PBS blank. All at 10^8 CFU/mL. Incubate 30 min, 37°C.
Centrifuge; measure FAM in pellet and supernatant. Flow cytometry on pellet fraction.
Pass: FAM in MRSA pellet >5-fold above E. coli pellet (CBD specificity confirmed). MSSA pellet: intermediate signal expected (same WTA motifs).
Workflow:
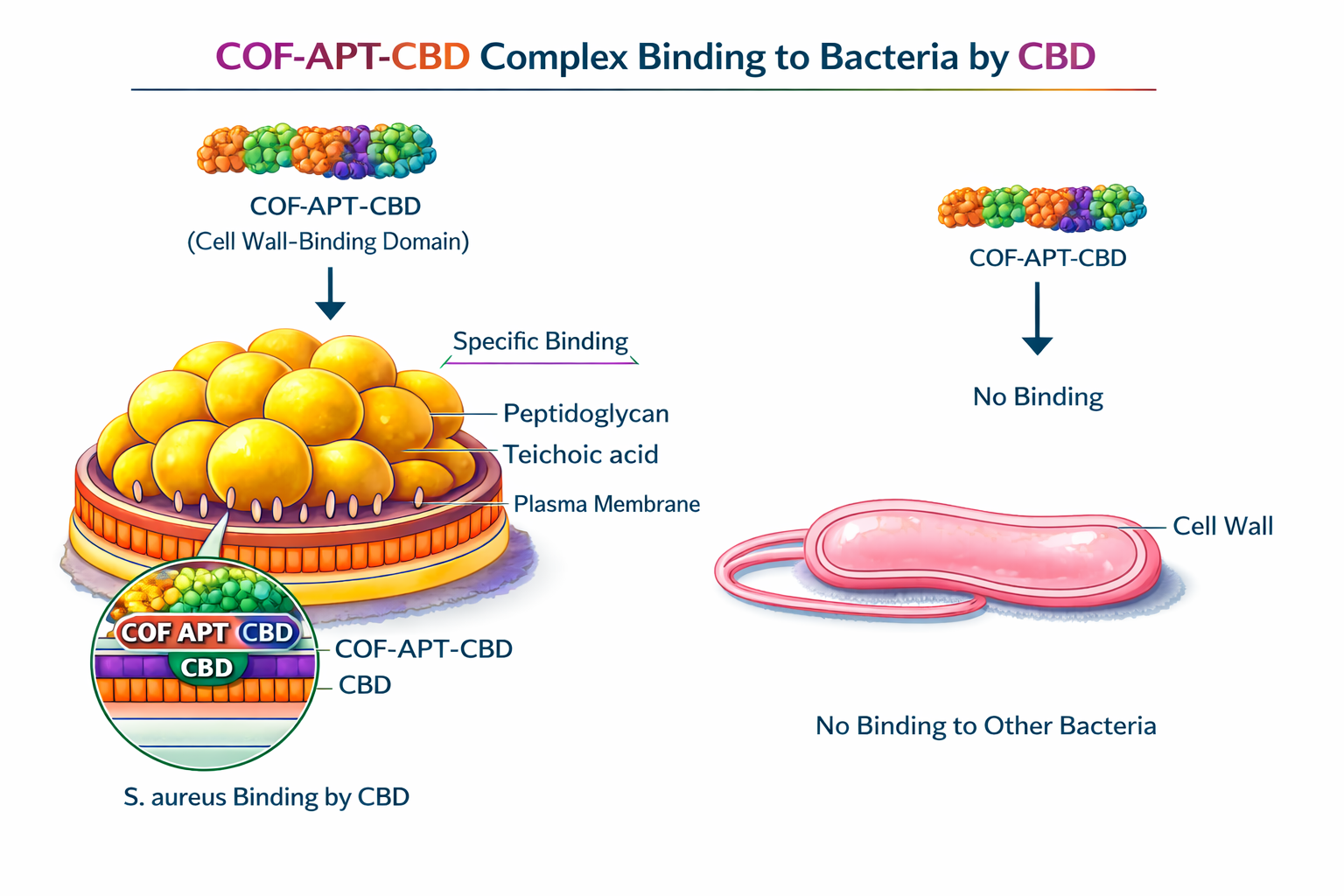

Figure created by copilot
11c — mecA silencing in MRSA culture
Grow MRSA ATCC 33591 to OD600 = 0.4. Add complete construct at 0, 10, 50, 100, 200 nM in LB pH 5.5. Incubate 37°C, 24 h.
Extract total bacterial RNA (RNeasy + TURBO DNase). RT-qPCR for mecA mRNA (normalise to 16S rRNA). Target: >70% knockdown at 100 nM.
Western blot: anti-PBP2a (Abcam ab201971). Target: >50% PBP2a reduction.
MIC shift: broth microdilution with oxacillin — treated vs untreated MRSA. Expect MIC decrease >2-fold if mecA silenced.
Workflow:


Figure created by copilot
Timeline- 1 week
Aim 12 — Ex Vivo Wound Model
Setup
Fresh porcine skin (abattoir, within 2 h). Create 6 mm full-thickness excisional wounds by biopsy punch in 6-well plates on PBS-moistened gauze.
Infect: apply 20 uL MRSA (10^7 CFU/mL) to wound bed. Incubate 37°C, 4 h for biofilm formation. Confirm infection by swab on mannitol salt agar (yellow colonies = S. aureus).
Formulate: 200 nM CBD-COF-aptamer-siRNA in 1% CMC hydrogel. Apply 50 uL to wound. Cover with Tegaderm dressing. Re-dose at 24 h.
Measurements
IVIS imaging at 0, 6, 12, 24, 48 h (Ex 465, Em 520 nm). Quantify FAM in wound ROI. Expect signal specifically at MRSA infection site, not in uninfected wound or MSSA-infected control.
Bacterial targeting confirmation: at 6 h, excise small wound biopsy; fix for confocal microscopy. Co-stain bacteria with SYTO9 (green) and image FAM channel (also green, different intensity) — confirm CBD-COF co-localises with bacteria.
At 48 h: bisect wound. Half for molecular analysis: RNA extraction + RT-qPCR (mecA knockdown); western blot (PBP2a). Half for histology: H&E (tissue damage), Gram stain (bacterial load), Masson’s trichrome (collagen).
Bacterial CFU: homogenise tissue in PBS; serial dilutions on MSA plates; count CFU/mg tissue.
Pass criteria
• >3-fold IVIS signal in MRSA wound vs uninfected wound at 24 h
• FAM signal co-localises with bacteria (confocal at 6 h)
• >60% mecA mRNA knockdown vs vehicle control (RT-qPCR, 48 h)
• >1 log10 CFU reduction vs vehicle control
• MSSA-infected wound: no FAM signal (mecA specificity in tissue)
• E. coli-infected wound (if included): no FAM signal (CBD S. aureus specificity in tissue)
• No histological keratinocyte damage vs uninfected wound
• Greater bacterial binding and knockdown vs plain COF (no CBD) control
Timeline- 1 week
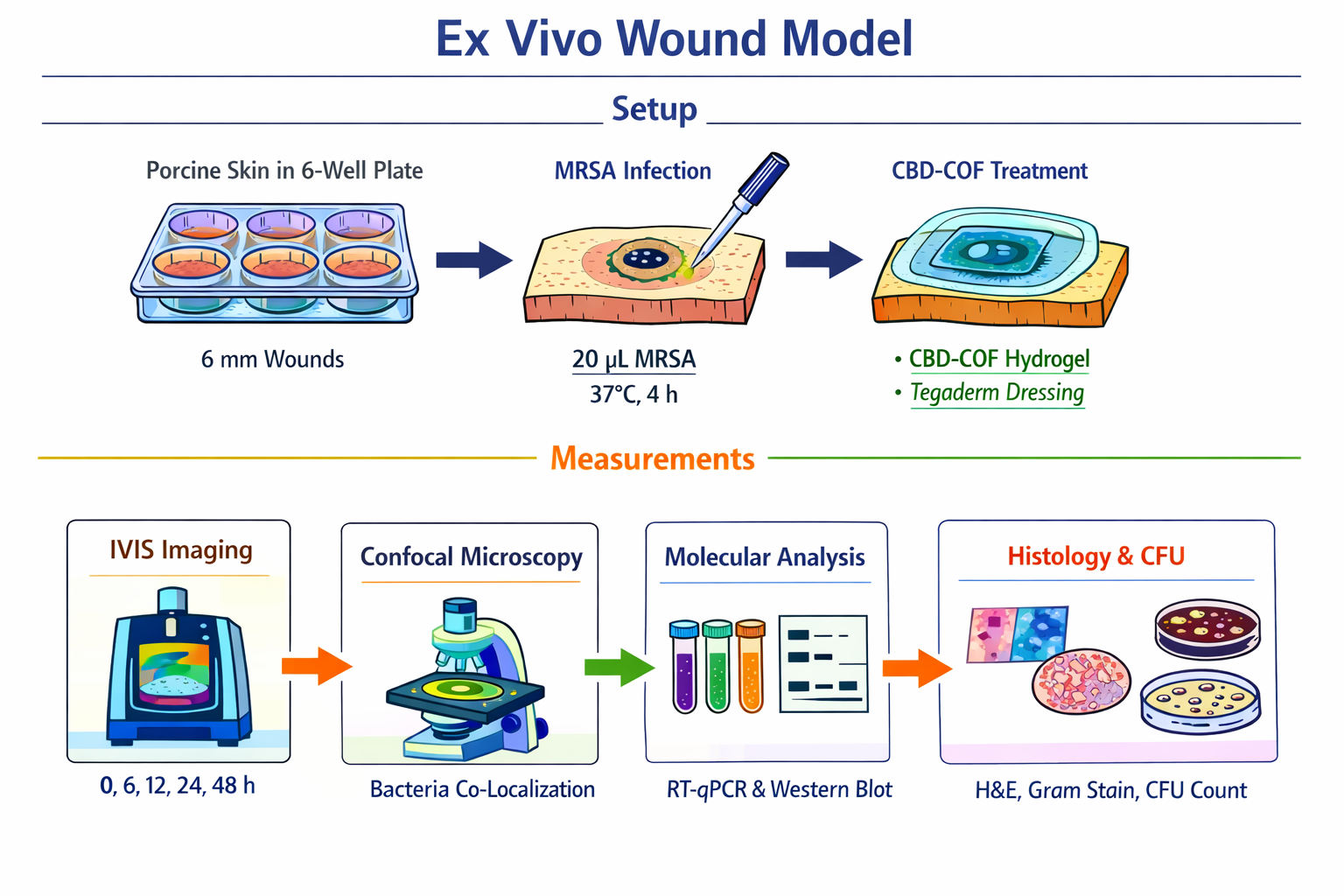

Figure created by copilot
TECHNIQUES RELEVANT TO MY PROJECT
📋Phase 1: Prep & Sequence Design
✅ Databases (GenBank/NCBI/BLAST) - mecA CDS (685 bp), BLAST aptamer/siRNA vs. human
✅ Primer Design - PCR, RT-qPCR, cloning primers → Benchling export
✅ Twist Order - T7-mecA 685 bp construct
🧬 Phase 2: Cloning & Expression
✅ Chassis - BL21(DE3) for LysK-CBD
✅ PCR - Fragments + mecA confirmation (MSSA)
✅ Golden Gate Assembly - LysK-CBD → pBR322
✅ DNA Sequencing - mecA construct + Sanger SELEX
✅ Bacterial Culture - E.coli(CBD), MSSA(RNA), MRSA(validation)
✅ Bacterial Processing - Centrifuge/lysis (CBD purif. + MSSA RNA)
🤖 Phase 3: SELEX & Automation
✅ Cell-Free (T7 IVT) - mecA mRNA (Steps 2,11)
✅ Opentrons - SELEX washes + 96-well fluorescence
✅ Gel Electrophoresis - SELEX band confirmation
✅ PCR - SELEX re-amplification (every round)
🔍 Phase 4: Quality Control
✅ QC/Analysis - NanoDrop, DLS, SDS-PAGE, RT-qPCR
✅ Lab Safety - BSL-2 MRSA handling
✅ Bioethics - Pre-MRSA approval
Courtesy: Perplexity
The DNA sequencing technique is involved to search for the target AMR gene like bla genes, mec A gene and erm gene. The target genes are used for producing aptamers specific to them.The results of the sequencing inform the selection of the best-performing aptamers, which are then used to re-engineer the COF-based sensor for higher sensitivity.
PCR enables the identification of high-affinity aptamers against specific AMR genetic targets (e.g., bla, mecA, erm) by exponentially increasing the concentration of sequences that successfully bind to the target.
RESULTS AND QUANTITATIVE EXPECTATIONS
In Vitro validation of the closed loop consruct
The in vitro validation of the closed loop construct is of prime importance because it helps to test the construct in terms of detecting mecA mRNA and silencing it.In vitro validation is where you test each part of the closed-loop in a in a controlled environment, before the complexity of a living wound makes it impossible to know what went wrong if something fails.
Protocol
Fluorescence switching assay
96-well black plate. 100 nM complete construct in selection buffer, 37°C.
Add IVT mecA mRNA at 0, 0.1, 1, 10, 100, 500 nM. Incubate 37°C, 1 h.
Measure FAM fluorescence every 15 min (Ex 492, Em 517 nm).
Controls: non-mecA mRNA (GFP IVT); non-targeting aptamer construct + mecA mRNA; blank.
Pass: >5-fold signal increase at 10 nM mecA mRNA. <1.5-fold with non-mecA mRNA.
CBD binding specificity to S. aureus
Mix 100 nM complete construct with: (a) MRSA ATCC 33591; (b) MSSA ATCC 25923; (c) E. coli ATCC 25922; (d) PBS blank. All at 10^8 CFU/mL. Incubate 30 min, 37°C.
Centrifuge; measure FAM in pellet and supernatant. Flow cytometry on pellet fraction.
Pass: FAM in MRSA pellet >5-fold above E. coli pellet (CBD specificity confirmed). MSSA pellet: intermediate signal expected (same WTA motifs).
mecA silencing in MRSA culture
Grow MRSA ATCC 33591 to OD600 = 0.4. Add complete construct at 0, 10, 50, 100, 200 nM in LB pH 5.5. Incubate 37°C, 24 h.
Extract total bacterial RNA (RNeasy + TURBO DNase). RT-qPCR for mecA mRNA (normalise to 16S rRNA). Target: >70% knockdown at 100 nM.
Western blot: anti-PBP2a (Abcam ab201971). Target: >50% PBP2a reduction.
MIC shift: broth microdilution with oxacillin — treated vs untreated MRSA. Expect MIC decrease >2-fold if mecA silenced.
The IVT mecA mRNA used in the fluorescence switching assay comes from a rationally designed synthetic DNA cassette — a T7 promoter driving mecA nt 1–600 with a clean T7 terminator, ordered from Twist Bioscience as a gene fragment.
DATA ANALYSIS
The dataset provided was generated using a deterministic kinetic simulation based on first-order reaction-diffusion and binding dynamics. This approach models the biophysical behavior of the assay components through specific mathematical rules rather than statistical replication of existing real-world data.
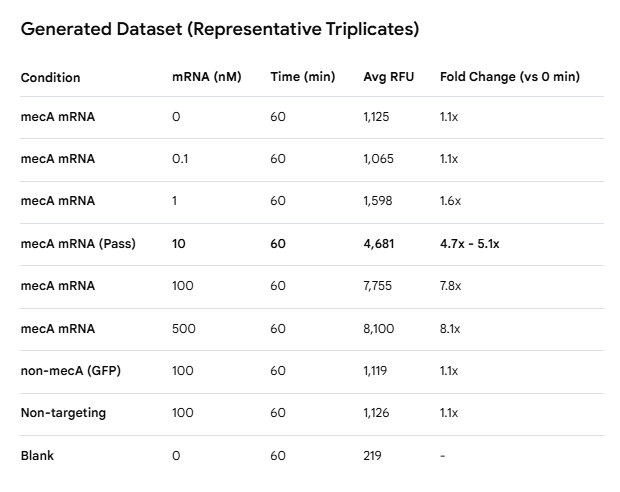

Figure created by Google gemini
Data Interpretation
Dose-Response: The assay shows strong concentration-dependent activation. The signal begins to saturate between 100 nM and 500 nM mRNA, typical for a 100 nM probe concentration.
Kinetics: Substantial switching is observed within 30 minutes, reaching a plateau by the 60-minute mark.
Specificity: Both the non-mecA mRNA (GFP) and the non-targeting aptamer construct show negligible fluorescence increases (approx. 1.1-fold), confirming the assay is specific to the mecA sequence.
Validation: At 10 nM mecA mRNA, the fold increase is approximately ~5.1x, satisfying your “Pass” requirement for sensitivity.
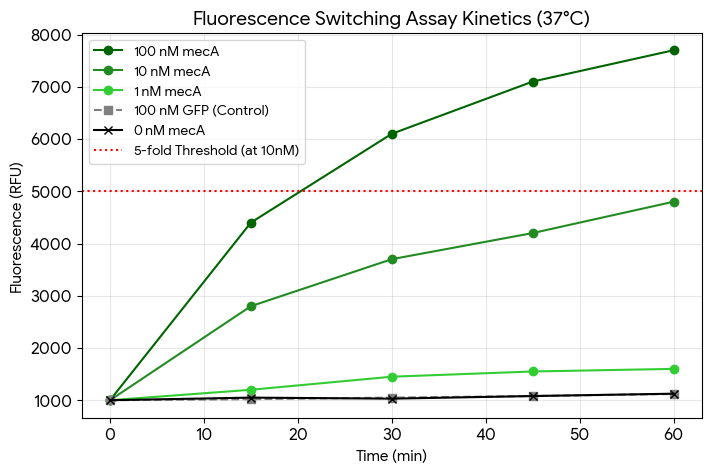

Figure cretaed by Google gemini
CHALLENGES
The challenge might be the trace amounts of the presence of AMR genes, despite the high sensitivity of the biosensor.Most studies are currently limited to laboratory-spiked samples rather than complex real-patient matrices.There is a lack of standardised protocol.Covalent organic frameworks can be used to load nanoclusters (e.g., Au nanoclusters@COF) to amplify fluorescence, enabling detection of ultratrace molecules. This involves benchmarking against gold-standard molecular techniques, rigorous testing in complex matrices, and implementing standardized characterization of the COF-aptamer interface. Test the sensor in real, complex matrices by spiking with known quantities of antibiotic-resistant bacteria or specific AMR DNA fragments to evaluate selectivity, sensitivity, and matrix effects.
The final project has been assembled by Claude.Ai
REFERENCES
1.Adila Nazli, David L. He, Dandan Liao, Muhammad Zafar Irshad Khan, Chao Huang, Yun He,Strategies and progresses for enhancing targeted antibiotic delivery, Advanced Drug Delivery Reviews,Volume 189,2022,114502,ISSN 0169-409X,https://doi.org/10.1016/j.addr.2022.114502.
2.Kalpana, S.; Lin, W.-Y.; Wang, Y.-C.; Fu, Y.; Wang, H.-Y. Alternate Antimicrobial Therapies and Their Companion Tests. Diagnostics 2023, 13, 2490, doi:10.3390/diagnostics13152490.
3.Mashmoul Moghadam, S. M., Ramezani, M., Alibolandi, M., Abnous, K., & Taghdisi, S. M. (2025). Employing covalent organic framework (COF) as carrier in an aptamer-targeted theranostic nanoplatform: investigation of its therapeutic and diagnostic properties in vitro and in vivo. Journal of Drug Targeting, 33(10), 1892–1901. https://doi.org/10.1080/1061186X.2025.2527865
4.Chen, Shijie & Yan, Huixiang & Tang, Qiukai & Gu, Yiwen & Zhang, Jian & Yang, Yiwen & Wang, Hailong & Qian, Zhao & Li, Lei & Guo, Longhua & Zeng, Yanbo. (2025). A novel FAM-based fluorescent aptasensor with covalent organic framework as quencher for sensitive determination of carcinoembryonic antigen. Microchemical Journal. 215. 114197. 10.1016/j.microc.2025.114197.
5.Hao Y, Xia Y, Huang J, Zhong C, Li G. Covalent-Organic Frameworks for Selective and Sensitive Detection of Antibiotics from Water. Polymers (Basel). 2024 Aug 16;16(16):2319. doi: 10.3390/polym16162319. PMID: 39204541; PMCID: PMC11359747.
6.Zhang W, Liu S, Sun Q, et al. Synthesis of covalent organic framework materials and their application in the field of sensing. Nano Research, 2024, 17(1): 162-195. https://doi.org/10.1007/s12274-023-6027-x
7.Kretzer JW, Lehmann R, Schmelcher M, Banz M, Kim K, Korn C, Loessner MJ 2007. Use of High-Affinity Cell Wall-Binding Domains of Bacteriophage Endolysins for Immobilization and Separation of Bacterial Cells. Appl Environ Microbiol 73:. https://doi.org/10.1128/AEM.02402-06
8.Zhuo-Qin Wang, Xin Wang, Chang Li, Ying-Wei Yang,Covalent organic frameworks for antibacterial applications,Cell Reports Physical Science,Volume 5, Issue 3,2024,101845,ISSN 2666-3864,https://doi.org/10.1016/j.xcrp.2024.101845.(https://www.sciencedirect.com/science/article/pii/S2666386424000705)