Week 2 Lab: DNA Gel Art
Louisa, Jasmine, Yutong and I worked on this lab together.
TAE Buffer Preparation
We mixed 8mL of TAE (50% concentration) with 492mL of deionized water to make 500mL of TAE buffer (1% concentration). We also added dye to the buffer to make the electrophoresis process traceable.

Agarose Electrophoresis Gel Preparation
First, we added 0.75g of agarose powder and 75mL of TAE buffer into a microwavable flask. We shaked the flask to mix the powder and the buffer.
Next, we microwaved the flask for multiple rounds of 20 seconds each until the agarose powder was completely dissolved.
After that, we waited for the agarose solution to cool down for about 20 minutes. We then added 7.5uL of SYBR Safe DNA stain to the solution.
Finally, we poured the agarose solution into a gel mold, inserted the comb, and let it solidify for about 30 minutes. After the gel solidified, we removed the comb and placed the gel.




Digestion
According to the gel art designed, we used EcoRV, SacI, BamHI, and KpnI. We created the mixture of Lambda DNA, the restriction enzyme, and the buffer according to the protocol. After mixing the solution, we incubated it at 37C for 30 minutes to allow the digestion to occur.


Running the Gel
First, we poured the TAE buffer into the gel box until the gel was submerged. Next, we pipetted the digested DNA samples into the wells of the gel.
We then connected the gel box to the power supply and ran the gel at 120V for about 30 minutes.
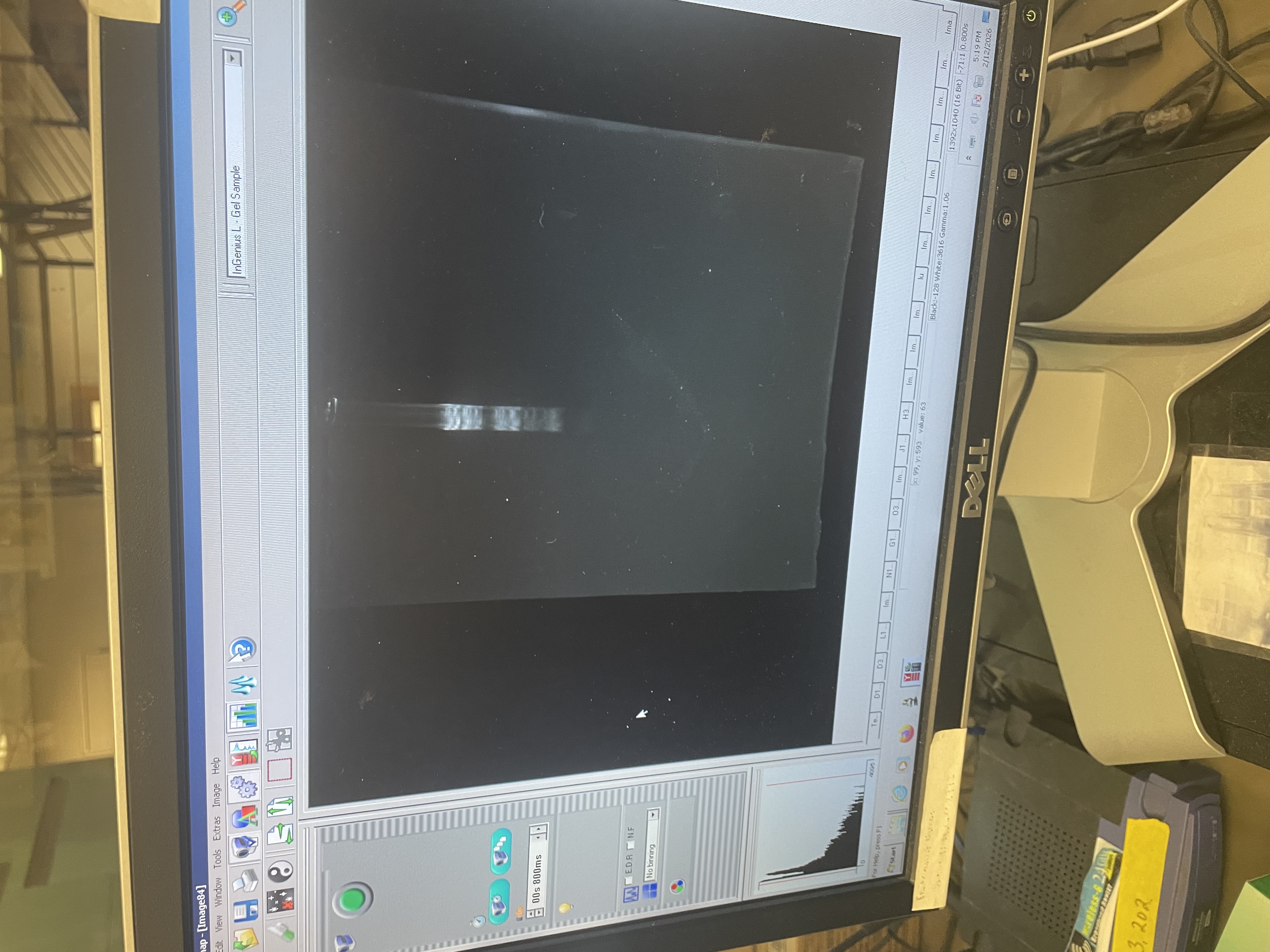
Result
Finally, we visualized the gel in the imaging system. The gel art is shown below. It does not look exactly like the design we created, but at least we gave it a try and had fun in the process!