Week 4 HW: Protein Design Part I
A
Answer any NINE of the following questions from Shuguang Zhang: (i.e. you can select two to skip)
1. How many molecules of amino acids do you take with a piece of 500 grams of meat? (on average an amino acid is ~100 Daltons)
A Dalton is equal to 1/12 atom of carbon, so aprox. 1.66x10-27kg. I will chose beef , because the type isn’t specified . 100g of beef has aporx. 25g protein, so 500g has 125g protein. 1g=6.022x1023 atomic mass units =>125g=7,5275 x 10^25 Dalton
2.Why do humans eat beef but do not become a cow, eat fish but do not become fish?
Humans eat beef but do not become a cow, eat fish but do not become fish, because these foods are decomposed in substances that are used or throw away . They don’t have an impact on our DNA so we don’t become cows or fishes.
3. Why are there only 20 natural amino acids?
There are only 20 natural amino acids because like that they are more stable and it can provide a big variety of proteins.
4. Can you make other non-natural amino acids? Design some new amino acids.
Yes, it is possible , but I won’t do it.
5. Where did amino acids come from before enzymes that make them, and before life started?
Before enzymes that make them, and before life started amino acids come from abiotic synthesis. Simple inorganic molecules reacted under high-energy conditions.
6. If you make an α-helix using D-amino acids, what handedness (right or left) would you expect?
7. If you make an α-helix using D-amino acids , there will be a left handedness.
8. Why are most molecular helices right-handed?
9. Why do β-sheets tend to aggregate? • What is the driving force for β-sheet aggregation?
10. Why do many amyloid diseases form β-sheets? • Can you use amyloid β-sheets as materials?
11. Design a β-sheet motif that forms a well-ordered structure.
Part B: Protein Analysis and Visualization
In this part of the homework, you will be using online resources and 3D visualization software to answer questions about proteins. Pick any protein (from any organism) of your interest that has a 3D structure and answer the following questions:
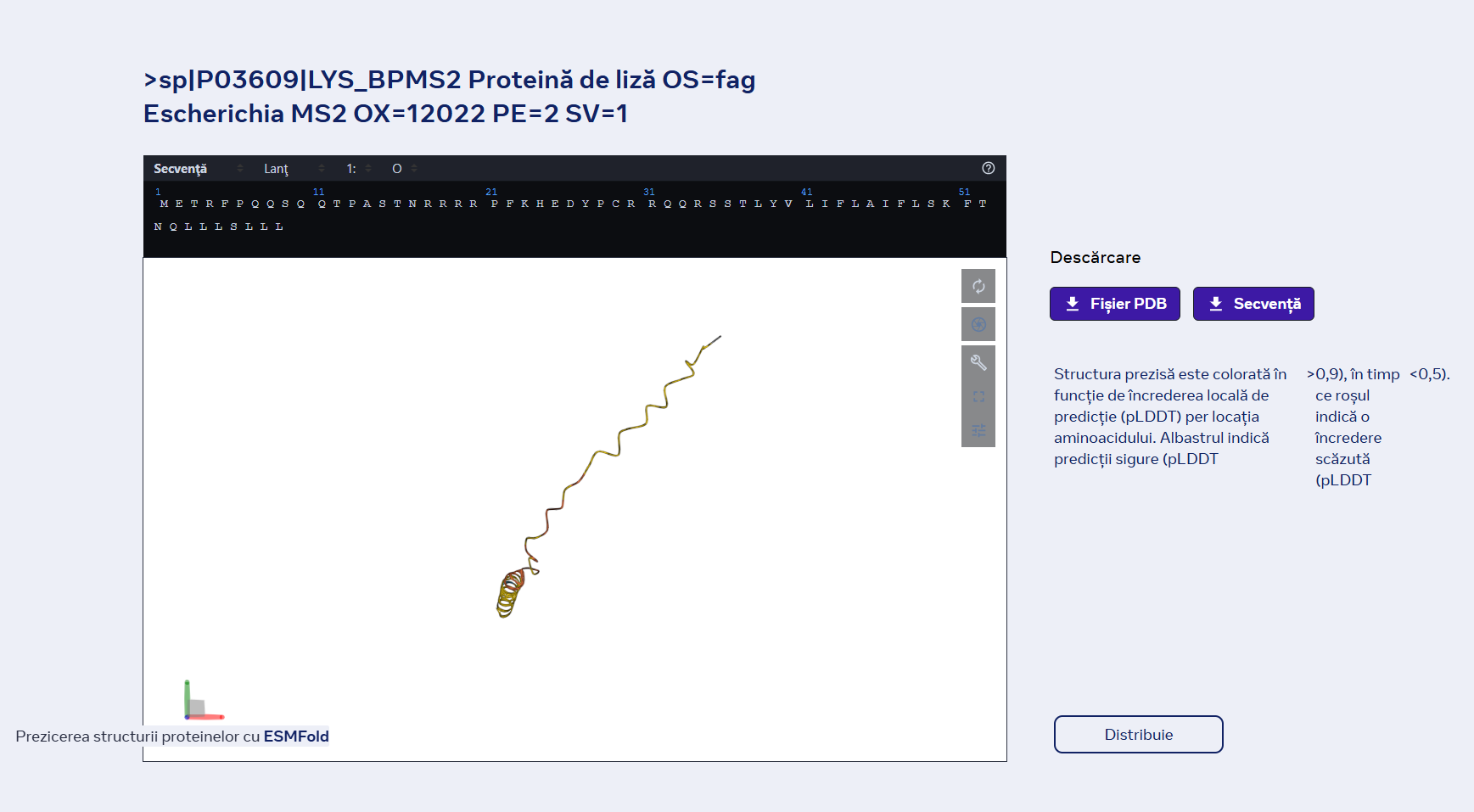
1Briefly describe the protein you selected and why you selected it.
I chose the same protein that I used in homework two, because Dsup is very interseting and I like very uch tardigrades.
2 Identify the amino acid sequence of your protein.
MASTHQSSTEPSSTGKSEETKKDASQGSGQDSKNVTVTKGTGSSATSAAIVKTGGSQGKDSSTTAGSSSTQGQKFSTTPTDPKTFSSDQKEKSKSPAKEVPSGGDSKSQGDTKSQSDAKSSGQSQGQSKDSGKSSSDSSKSHSVIGAVKDVVAGAKDVAGKAVEDAPSIMHTAVDAVKNAATTVKDVASSAASTVAEKVVDAYHSVVGDKTDDKKEGEHSGDKKDDSKAGSGSGQGGDNKKSEGETSGQAESSSGNEGAAPAKGRGRGRPPAAAKGVAKGAAKGAAASKGAKSGAESSKGGEQSSGDIEMADASSKGGSDQRDSAATVGEGGASGSEGGAKKGRGRGAGKKADAGDTSAEPPRRSSRLTSSGTGAGSAPAAAKGGAKRAASSSSTPSNAKKQATGGAGKAAATKATAAKSAASKAPQNGAGAKKKGGKAGGRKRK
How long is it? What is the most frequent amino acid? You can use this Colab notebook to count the frequency of amino acids.
THe protein ha 445 aminoacids.The most frequent aminoacid is Serine. It apears 84 times.
How many protein sequence homologs are there for your protein? Hint: Use Uniprot’s BLAST tool to search for homologs.
I searched for homologs in Uniprot and I think I managed to find them . So, I opened the Uniprot link is on the htgaa website at week 4 . I paste the aminoacid sequence and then I waited it to run.
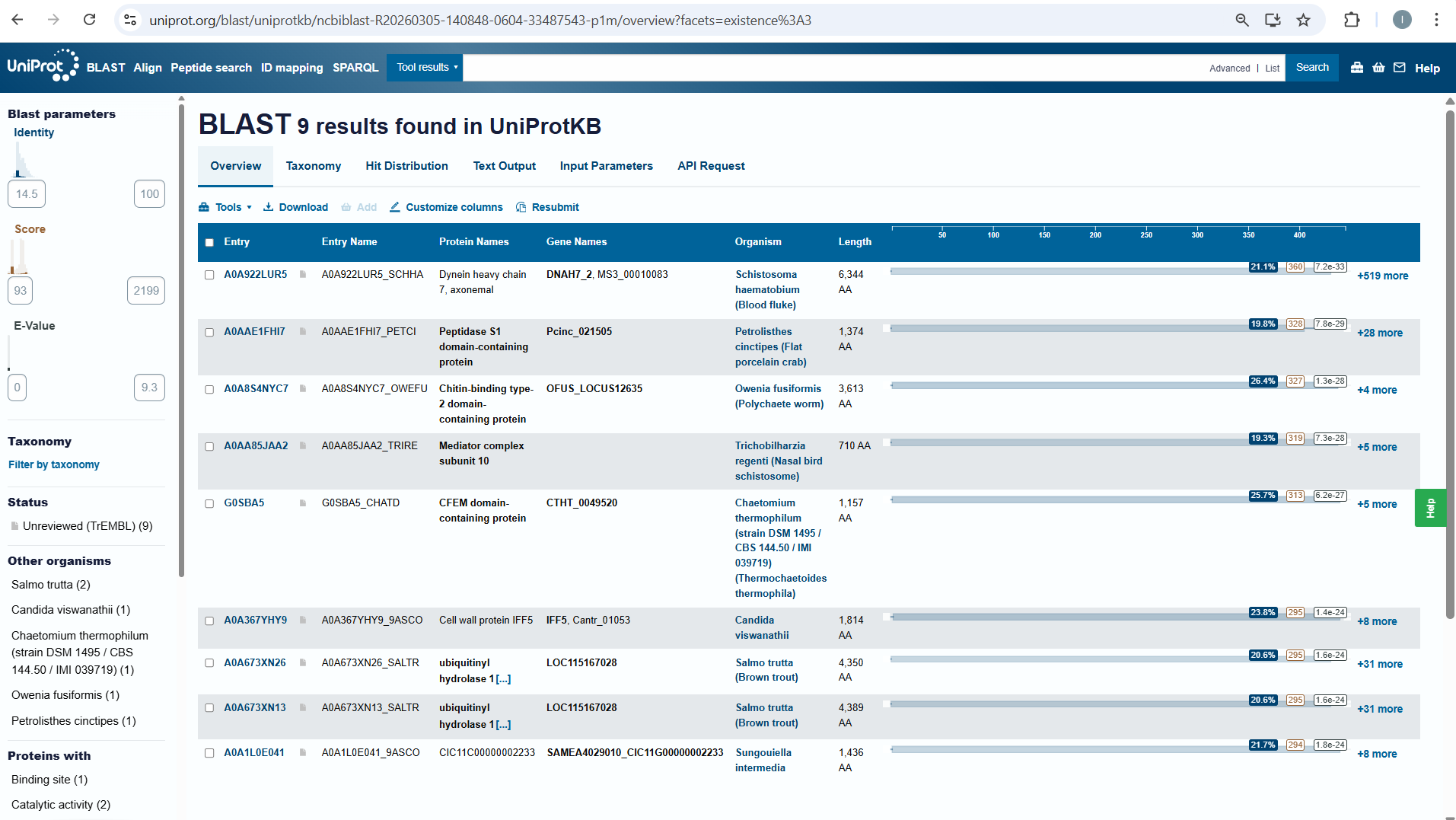

Does your protein belong to any protein family?
NO, it doesn’t belong to any protein family.
**3 Identify the structure page of your protein in RCSB
When was the structure solved? Is it a good quality structure? Good quality structure is the one with good resolution. Smaller the better (Resolution: 2.70 Å)
Are there any other molecules in the solved structure apart from protein? Does your protein belong to any structure classification family?
Nucleosome-Binding Protein
3 Open the structure of your protein in any 3D molecule visualization software: Visualize the protein as “cartoon”, “ribbon” and “ball and stick”.
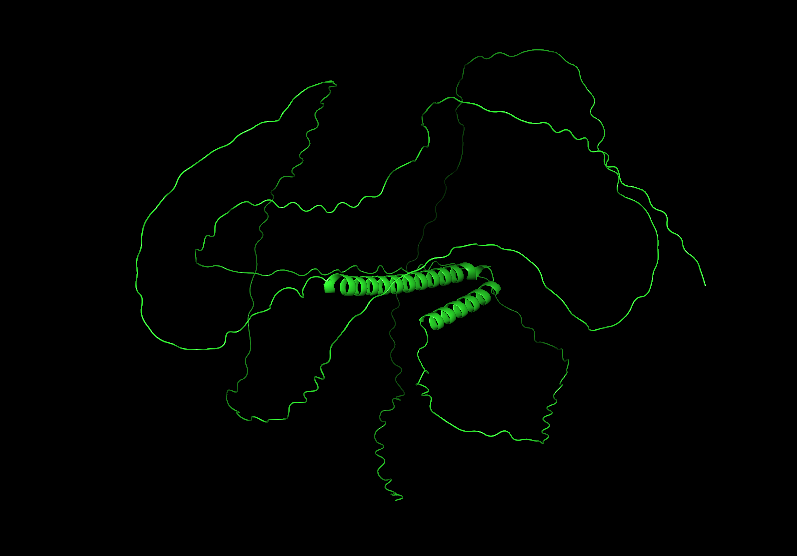

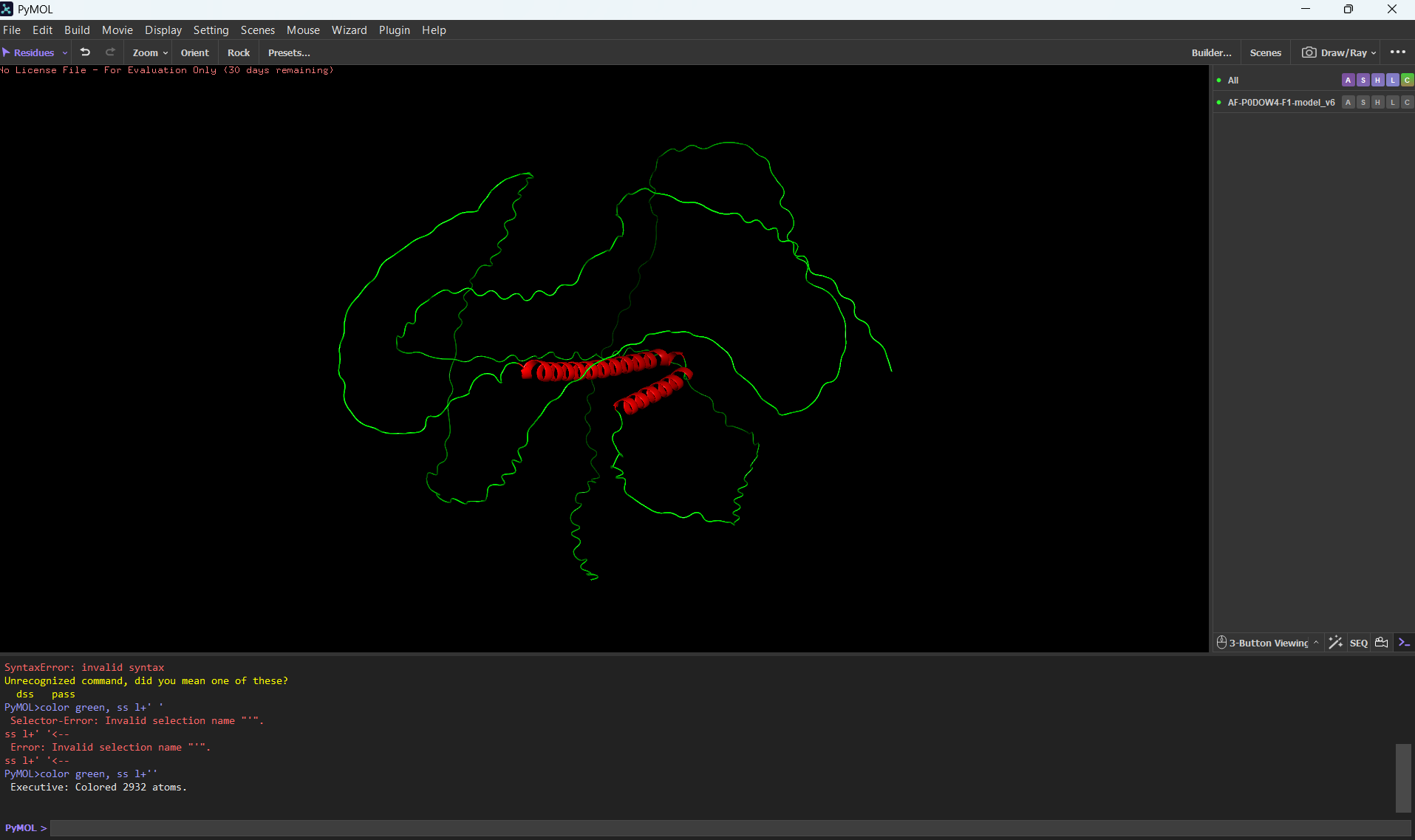

Color the protein by secondary structure. Does it have more helices or sheets?
It has more helices than sheets.
Color the protein by residue type. What can you tell about the distribution of hydrophobic vs hydrophilic residues?
Most of the protein will remain green, with a few small cylinders representing transient alpha-helices.
Visualize the surface of the protein. Does it have any “holes” (aka binding pockets)?
To color by secondary structure I used color red, ss h; color yellow, ss s; color green, ss l+’'.