Week 1 HW: Principles and Practices

Question 1: First, describe a biological engineering application or tool you want to develop and why. This could be inspired by an idea for your HTGAA class project and/or something for which you are already doing in your research, or something you are just curious about.
DNA is the rulebook of life! Imagine it as small lego pieces, building up the entire empire of a human body. But, what if we can pick out the lego pieces and assemble them in different, never-thought-before ways, to unlock new functions and outcomes ?! This is the potential of engineering synthetic genetic circuits! I am intrigued by the possibility of engineering our desired outcome by designing genetic circuits that can alter or control it.
My (Initial) Vision:
I would like to build a multi-input theragnostic circuit for ovarian cancer, given the fact that, inspite of being the one of the deadliest gynaecological cancers, it does not have a means for early detection, leading to late diagnosis and poor outcomes.
Where would it operate? The circuit can be delivered directly to cancer cells, by nanoparticles or viral vectors.
Where is it meant to be used? Upon successful testing in laboratories and passing clinical trials, it can be used as a part of clinical cancer treatments.
Desired Characteristics: The gene circuit must be able to…
1 Take in multiple inputs.
2 Detect the condition and inform us upon detection.
3 Inform us if the condition is not detected.
4 Secrete/produce chemicals that slow down/aid in the slowing down of the progression of the condition.


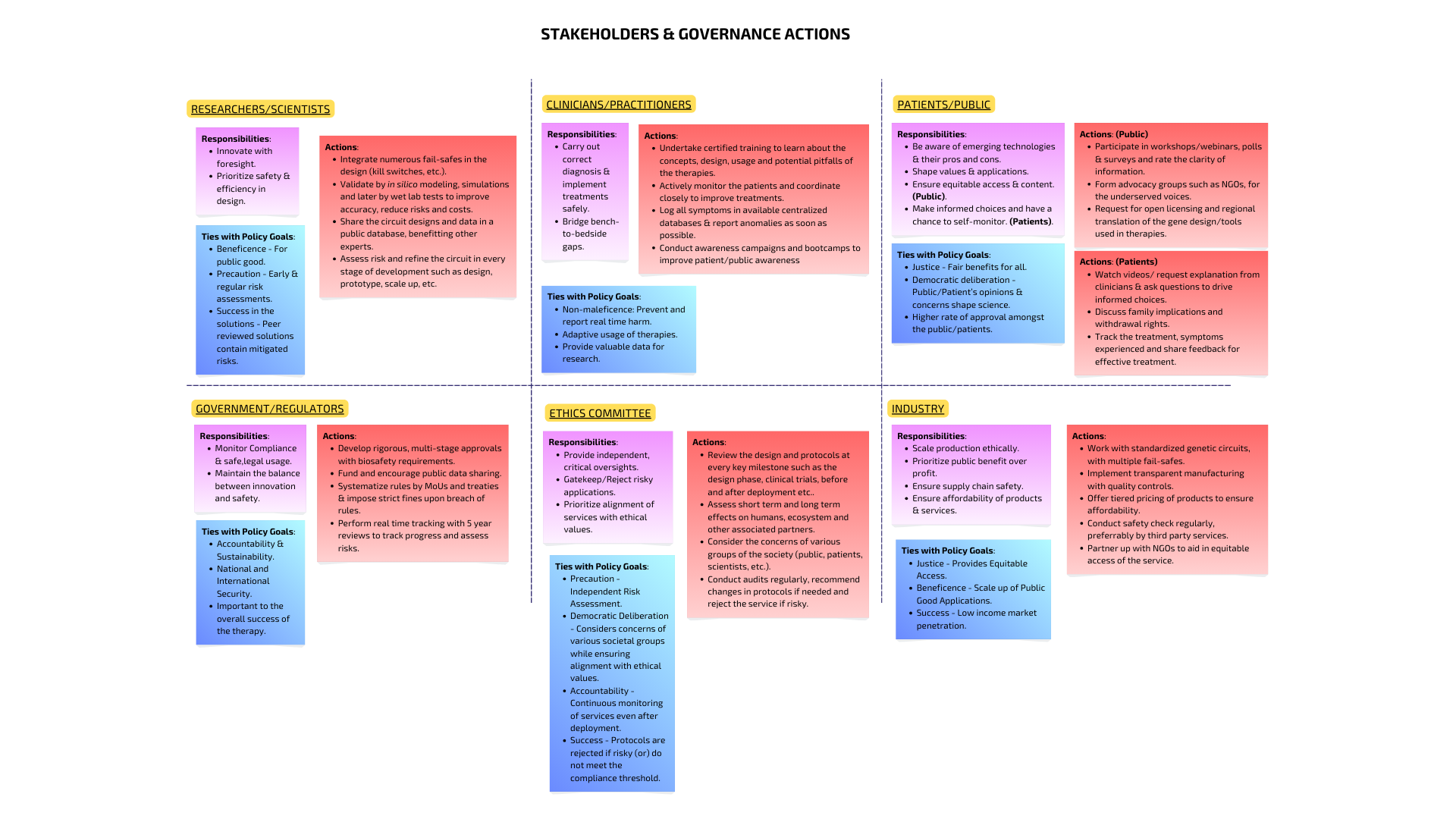

Question 2: Describe one or more governance policy goals related to ensuring this application contributes to an ethical future & prevents harm.
Some governance policy goals are described below. For others, kindly refer to the attached image!
Goal 1: Precaution/ Biosecurity
Rigorous audits of safety during each phase of design, validation and implementation, offers a high chance of mitigating risks and evaluating the feasibility of the therapy. Here’s how it might look:
Researchers and Industries can incorporate multiple fail-safes combined with regular safety monitoring to make the therapy harmless, incase it fails to perform.
Ethics Committee can provide an independent review of the therapy at each stage, critically assessing designs and recommending changes to reject risky therapies and promote the safe designs.
Government/Regulators can assess the design by subjecting it to tiered evaluations and systematize rules for such services, ensuring national security and safety.
Clinicians can verify the diagnosis multiple times before prescribing the treatment ensuring that it is the right fit for the patient.
Goal 2: Transparency
Transparency in the design of the circuit, the protocols adopted and the data used can bring safety improvements and enable collective oversight. Here is how it might look:
Researchers can share the data after screening in public databases, enabling other experts to spot missed flaws, improve the protocols and design or even create new knowledge based upon this data.
Clinicians logging symptoms in centralized/ government enabled databases with real time tracking, can aid in efficient treatment along with eaarly detection of abnormalities.
Public rating of the information clarity and consideration of public concerns by ethical committees can drive faster improval and a high acceptance rate from the public.
Patients can also be enabled to make informed choices, when they are clear in how their data is being used and how the therapy works.
Goal 3: Equitable Access
This goal ensures that the therapy reaches to all populations, avoiding unreasonable risk burdens on some/vulnerable groups. Here are some ways it can occur:
Industry can offer tiered pricing for different populations while also partnering with advocacy groups such as NGOs, to reach a large and wider market.
The government can mandate tiered pricing from industries, set price caps on services or offer subsidies by which they can reach out to low income populations.
Fun fact: Singaporean government already does this by subsidizing 75% of the gene therapy costs by bulk deals!
The public can request regional translations of the materials and aid in equitable access.
Question 3: Describe at least three different potential governance actions by considering the purpose, design, assumptions, and risks of failures & “success”. Draw upon your existing knowledge and a little additional digging, and feel free to use analogies to other domains such as 3D printing, drones, financial system, etc.
Purpose: What is done now and what changes are you proposing?
Design: What is needed to make it “work”? Consider the actor(s) involved - who must opt in, fund, approve, or implement?
Assumptions: What could you have wrong? Incorrect assumptions? Uncertainties?
Risks of Failures & Success: How might this fail, including any unintended consequences of the “success” of your proposed actions?
Action 1: Incorporation of Multiple (atleast 2) Fail-Safes
Purpose: To prevent the activation of circuit outside specific conditions, preventing harm, contamination and weaponization.
Design: A proposed method is to include a light inducible activation/inactivation. We can also design circuits which stops gene expression if proteins/markers belonging exclusively to healthy cells are detected.
Assumptions: Independent kill switches, No cross reactions, function reliably in lab and in vivo conditions.
Risks of Failure:
Failure of one or both kill switches.
Failure to activate or inactivate circuits by light when needed.
Failure to choose exclusive proteins belonging to only healthy cells.
Success:
Key switches activate as intended.
Inactivation in healthy cells as intended.
Reliable and predictable behaviour in lab and in vivo conditions.
Action 2: Public Database
Purpose: Collective safety debugging enabling global experts to spot missed flaws and enable transparency, preventing bioweaponization.
Design: Centralized registry with tiered access where non-sensitive designs are public and risky sequences are screened.
Assumptions: Responsible actors, Biosecurity screening networks, Safety as transparency are balancedly prioritized.
Risks of Failure:
Misuse of the database leading to weaponization.
Success:
Less/No Biosecurity incidents.
Peer improvements leading to improval of therapy protocols and design.
High public acceptance and approval rates.
Action 3: Tiered Pricing
Purpose: Tiered pricing enables quality care for all populations of the society, regardless of their incomes and other differences.
Design: Indutry partnerships with NGOs, Government enforced bulk deals and subsidies and encouragement by ethical committees to develop community beneficial plans can be useful.
Assumptions: Industry prioritizes health and accepts low margins, Global coordination of governments.
Risks of Failure:
Unofficial sources release the therapy designs and services.
Corporate pressure causes price cap weakening by the government.
Therapy quality degrades in accordance to the prices fixed.
Success:
Significant low income market penetration within a short time span (e.g. 5 years)
Therapy is considered a public good and not a luxury.
Please refer to the other governance actions in the image!
Question 4: Score each of your governance actions against your rubric of policy goals.
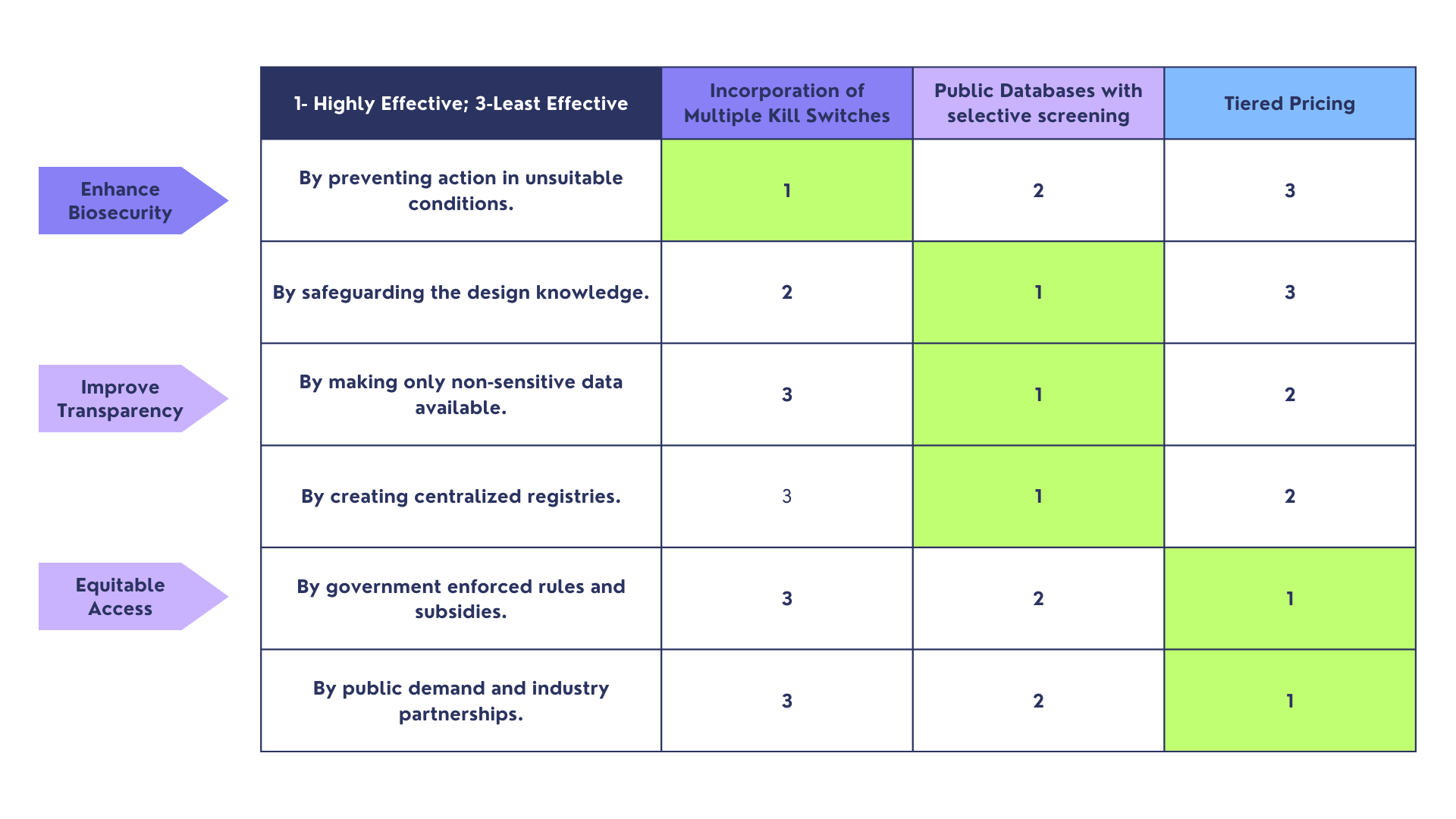

Question 5: Based on scores, describe which governance option or combination of options, you would prioritize, and why. Outline any trade-offs you considered as well as assumptions and uncertainties. Think about your audience - very local (MIT, Cambridge Mayoral Office), to national (President or Head of a Federal Agency), to international (United Nations Office of the Secretary-General)
Based on the table, I would prioritize incorporation of multiple kill switches for enhancing biosecurity, creating public databases with selective screening to achieve biosecurity and improve transparency and opt for tiered pricing to ensure equitable access of the therapy to all.
References:
1 WHO Health Ethics and Governance: https://r.search.yahoo.com/_ylt=Awrx.ztbaYtpOQIAv7O7HAx.;_ylu=Y29sbwNzZzMEcG9zAzEEdnRpZAMEc2VjA3Ny/RV=2/RE=1771953756/RO=10/RU=https%3a%2f%2fwww.who.int%2fteams%2fhealth-ethics-governance%2fgovernance/RK=2/RS=wajsNkQcjNltVnvz6DvebVhvzic-
2 Genetically stable kill-switch using “demon and angel” expression construct of essential genes: https://doi.org/10.3389/fbioe.2024.1365870
3 Advances in Synthetic Biology and Biosafety Governance: https://doi.org/10.3389/fbioe.2021.598087
Tools Used:
1 Canva - For preparing the Stakeholders & Governance Actions chart and Ranking chart.
2 Pinterest - For obtaining the Cover Images.
3 Perplexity AI - To understand the questions & their concepts, and for refining answer phrases. (Ethics in biology is a very new area for me, so I used AI to understand the field, concepts and given questions. I also used AI to polish the answers and ensure usage of correct terminology. However, the essence of the answer was not prepared using AI.)
Prompt Used: I want to explore ethics and governance policies for a novel genetic circuit design. However, I am a beginner to these fields. Give me a general overview of the subject and give me examples of concepts to begin with, and also tell me the ways in which I can structure a writeup.
Thank you!