Week 6 HW: Genetic Circuits Part I: Assembly Technologies
Assignment: DNA Assembly
1. What are some components in the Phusion High-Fidelity PCR Master Mix and what is their purpose?
- Phusion High‑Fidelity DNA Polymerase. This is the key enzyme that copies DNA during PCR.
- Reaction Buffer (HF Buffer). The buffer provides the chemical environment in which the enzyme works best.
- Deoxynucleoside Triphosphates (dNTPs). These are the building blocks of DNA (A, T, G, C).
- Optional Additives
- Water. Used to bring the reaction to the final volume and ensure proper concentrations of all components once primers and DNA template are added.
2. What are some factors that determine primer annealing temperature during PCR?
The primer annealing temperature (Ta) in PCR is critical because it determines how specifically your primers bind to the target DNA. If the temperature is too low, primers may bind nonspecifically; if too high, they may not bind efficiently.
- Primer sequence composition
- Primer melting temperature
- Salt concentration in the reaction
- Primer-target complementarity
- Presence of additives
- PCR polymerase used Some polymerases are more tolerant to mismatches or high GC content; high-fidelity polymerases like Phusion often work better with slightly higher annealing temperatures.
3. What are some components in the Phusion High-Fidelity PCR Master Mix and what is their purpose?
Protocol differences
| Aspect | PCR | Restriction Enzyme Digest |
|---|---|---|
| Input material | Template DNA containing the target sequence | DNA containing one or more recognition sites for the enzyme |
| Reaction components | Primers, DNA polymerase, dNTPs, buffer, template | Restriction enzyme(s), buffer, template, Mg²⁺ |
| Temperature control | Requires thermocycling (denaturation → annealing → extension, repeated 25–35×) | Single incubation at optimal temperature for enzyme (usually 37°C) |
| Time | Typically 1–3 hours depending on fragment length and cycles | Usually 30 min–2 hours |
When one method is preferable
Use PCR
- to need highly specific fragments from a complex genome
- to want to amplify from very little starting material
- to want to add custom sequences, like restriction sites, tags, or mutations
Use Restriction Enzyme Digest
- to have large amounts of DNA and want fragments exactly defined by natural restriction sites
- to want to cut plasmids or genomic DNA into predictable pieces for cloning, mapping, or analysis
- to prefer a simpler, single-temperature protocol without thermal cycling
4. How can you ensure that the DNA sequences that you have digested and PCR-ed will be appropriate for Gibson cloning?
- Understand the requirements of Gibson Assembly
Gibson Assembly joins two or more DNA fragments in a single isothermal reaction using: Exonuclease – chews back the 5′ ends to create single-stranded 3′ overhangs. DNA polymerase – fills in the gaps after fragments anneal. DNA ligase – seals the nicks to make a continuous DNA molecule.
- Designing PCR products for Gibson cloning
When using PCR to generate fragments: Forward primer for fragment A should include a 5′ extension that is complementary to the start of fragment B. Reverse primer for fragment B should include a 5′ extension complementary to the end of fragment A. These extensions become the overlapping regions for Gibson Assembly.
5. How does the plasmid DNA enter the E. coli cells during transformation?
Plasmid DNA enters E. coli during transformation because the cells are made temporarily permeable (competent). In chemical transformation, DNA binds to the membrane and slips in during heat shock. In electroporation, DNA is driven through pores formed by a brief electric pulse. Once inside, plasmid DNA replicates and can express its genes, including selectable markers.
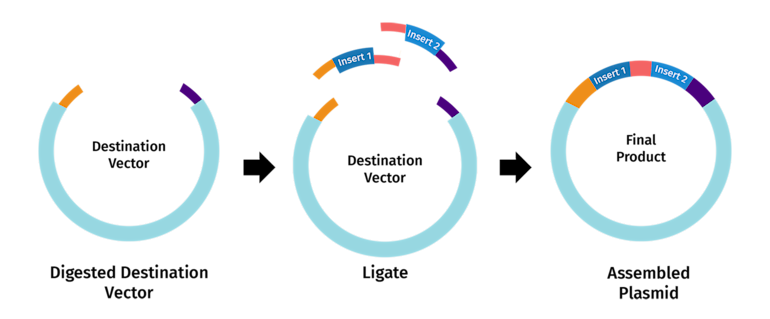
6. Describe another assembly method in detail
Golden Gate Assembly is a modular DNA assembly method that allows multiple DNA fragments to be joined in a defined order in a single reaction. It uses type IIS restriction enzymes, which cut outside of their recognition sequences, creating custom overhangs. Each fragment is designed with specific overhang sequences that match the adjacent fragment, so they ligate in the correct order. The reaction is performed simultaneously with a DNA ligase, so the fragments are cut and joined in cycles, and the recognition sites are eliminated in the final product, leaving seamless DNA. This method is very efficient for constructing multi-gene pathways or synthetic constructs because it allows precise assembly without leaving extra sequences. Golden Gate is often faster and more flexible than traditional cloning, especially when assembling more than 3–4 fragments. By careful design of overhangs, you can assemble fragments in a predefined order with high fidelity.

Assignment: Asimov Kernel
I don’t have access to the Asimov Kernel Software yet