Week 7 HW: Genetic Circuits Part II
Assignment Part 1: Intracellular Artificial Neural Networks (IANNs)
1. What advantages do IANNs have over traditional genetic circuits, whose input/output behaviors are Boolean functions?
A Boolean function is based on a binary system where it can only assign two values, such as "true" or "false", or as in numeric values: "0" or "1". This is akin to digital systems. Complexities in the biological systems cannot be adequately represented by binary input/output. Because signals in biological systems, such as concentrations of regulatory proteins, vary in gradation. So, a Boolean genetic circuit would have limitations in interpreting the complexities of a biological system.
IANNs are based on analog systems where weights are implemented. Examples of weights include variable concentrations of regulatory proteins, promoter strengths, and RBS efficiencies. These make positive or negative regulatory output. IANNs also integrate dose-response analysis, from inhibitory to non-inhibitory concentrations of a typical sigmoidal curve. IANNs consider biases such as taking into account whether promoters could be leaky. Advantageous parts are the ability to handle a great level of complexity due to the gradation that living systems have.
2. Describe a useful application for an IANN; include a detailed description of input/output behavior, as well as any limitations an IANN might face to achieve your goal.
IANNs for Forest Restoration
Several areas in the world face a threat of losing forests due to increased fungal pathogens and insect activity in a warming climate. Intracellular IANNs could have a useful application here by sensing the threats and producing defensins (antifungal peptides) and terpenes (insect deterrents).
Threats:
Amazon basin: Brazil, Peru, Colombia (Ceratocystis wilt, Phytophthora root rot, Leaf blight fungi, root rot complexes)
Europe: Scandinavia, Poland, Baltic states (Ash dieback, Bark-beetle-fungus complex, root rot)
Southeast Asia: Borneo, Sumatra, Mekong (Damping-off disease, vascular wilt, seedling fungal mortality)
Congo Basin: DRC, Cameroon, Gabon (Armillaria root disease, Cacao swollen shoot virus, termite crown damage)
North America: Eastern US, Pacific Northwest, Canada (Emerald ash borer, white nose syndrome, sudden oak death, chestnut blight)
The rescue plan:
A biosensor can be delivered via an endophyte microorganism (a commensal bacterial species such as Bacillus subtilis or Pseudomonas fluorescens or mycorrhizal fungi) inside trees, and an RNA-based biosensor can be additionally applied topically.
IANN Input/Output:
As an example, the IANN circuit can be designed to detect several markers indicative of pest activity, such as chitin, jasmonic acid (JA), and reactive oxygen species (ROS). Chitin would be found due to fungal activity. JA would be released due to insect feeding/damage to the plant. ROS would be detected at the infection site as the plant's response to stressors.
Overall, the IANN design in the example is responsive to 3 inputs (chitin, JA, and ROS), with an output to produce a synthetic regulator that turns the gene expression on simultaneously for 2 outputs, defensins and terpene production. The synthetic regulator is the middle layer. The synthetic regulator is designed to be expressed by a series of promoters, specific to the output of each sensor system. The amount of the synthetic regulator made would reflect being transformed from the input as a weighted function.
Chitin sensor. Plants sense chitin through the CERK1 receptor, which is bound to a membrane and has an extracellular sensing domain and a cytoplasmic kinase domain. Once chitin binds to the receptor, a signaling cascade leads to phosphorylation of a transcription factor, WRKY, which turns on gene expression. I would integrate the chitin sensor into the engineered microbe by building the synthetic hybrid CERK1 receptor and synthetic WRKY transcription factor. Synthetic WRKY would produce an output in response to chitin, that is, it will drive the expression of the synthetic regulator in the middle layer.
Jasmonate sensor. Jasmonates are phytohormones produced by the plant, and they trigger the degradation of proteins called JAZ repressors, which have a jas motif, a signature for degradation. The COI1 domain of the 26S proteasome mediates the degradation of JAZ repressors. The degradation of JAZ repressors enables JA-responsive gene expression as part of plant defense. Critically, a form of JA, JA-Ile, is required for the COI1 activity. A free form of JA is not the active ligand for COI1. A plant enzyme called JAR1 (JA amino acid synthetase) modifies JA by adding Ile. I would integrate the jasmonate sensor in the engineered microbe by creating a synthetic repressor with a jas-motif. COI1 and JAR1 are both also needed to be expressed in the engineered microbe. A synthetic repressor with a jas-motif would produce output in response to JA, driving the expression of the synthetic regulator in the middle layer.
ROS sensor. The bacterial H2O2 sensor, OxyR, is a transcriptional regulator. I would integrate this as part of the IANN circuit with the output that will drive the expression of the synthetic regulator in the middle layer.
Output, defensin.
These are small peptides, rich in cysteine. Several defensins are described, such as the MtDef4 class for ash dieback. Some defensins could also have antibacterial activity. Given that a choice of defensin does not kill the host, integration into the IANN would require that the expression depend on the synthetic regulator from the middle layer.
Output, terpenes.
Volatile compounds produced by the terpene synthase gene have an insect-repelling effect. These can be toxic to bacterial cells as well. Given that the choice of terpene is not bactericidal, integration into the IANN circuit is the same as defensin. The terpene expression is driven by the synthetic regulator from the middle layer.
Limitations:
Colonization of the endophyte in trees is a complex process. The stability of the IANN circuit in the endophyte and the succession in colonization are both unknown.
A massive delivery operation would be needed since forests are fairly large areas. Spray treatments could work for crops, but it is not certain that the spray delivery method would work for trees in forests.
Interaction with the soil microbiome, which is unknown, can be a concern since a genetically engineered organism is being released to the environment.
Public perception and regulatory barriers can be a limitation to using genetically engineered organisms in the environment.
Birds live in trees and feed on insects. An environmental safety assessment needs to be made to ensure the engineered endophyte does not pose a threat to birds and other animals.
3. Below is a diagram depicting an intracellular single-layer perceptron where the X1 input is DNA encoding for the Csy4 endoribonuclease and the X2 input is DNA encoding for a fluorescent protein output whose mRNA is regulated by Csy4. Tx: transcription; Tl: translation.
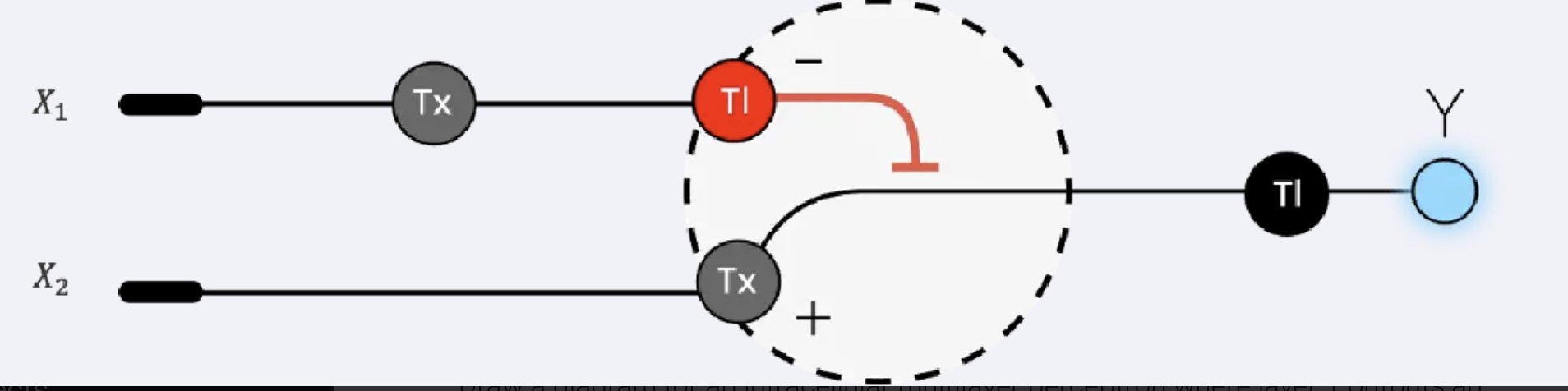

Draw a diagram for an intracellular multilayer perceptron where layer 1 outputs an endoribonuclease that regulates a fluorescent protein output in layer 2.
Assignment Part 2: Fungal Materials
1. What are some examples of existing fungal materials, and what are they used for? What are their advantages and disadvantages over traditional counterparts?
2. What might you want to genetically engineer fungi to do and why? What are the advantages of doing synthetic biology in fungi as opposed to bacteria?
Assignment Part 3: First DNA Twist Order
0. Review the Individual Final Project documentation guidelines.
1. Submit this Google Form with your draft Aim 1, final project summary, HTGAA industry council selections, and shared folder for DNA designs. DUE MARCH 20 FOR MIT/HARVARD/WELLESLEY STUDENTS
2. Review Part 3: DNA Design Challenge of the week 2 homework. Design at least 1 insert sequence and place it into the Benchling/Kernel/Other folder you shared in the Google Form above. Document the backbone vector it will be synthesized in on your website.