Week 1 : Principles and Practices
1. Describe a biological engineering application or tool you want to develop and why.
The oximeter, used to detect oxygen saturation, has known disparities accross different skin tones, due to the melanins ability to absorb near infered light (NIR) (Clarice). These disparaiteis result in patients or darker skin tone showing higher oxigen saturation and appearing healthier than they are. This resulted in black and hispanic patients recived delayed treated for COVID 19, due to systematic overestmation of oxygen saturation (Fawzy et al.) Genetic circuits in a synthetic biological application, that engineers DNA, RNA and protiens to programmable; functions such as biosensors that can be programmed to react to enviromental factors. I want to use this biological engineering application to engineer genetic circuits to sense a CO2 or O2 saturations in the epidermis. Using a biosensor that does not depend on NIR will increase the accuracy of oxygen readings and result in more equitable care
2. Describe one or more governance/policy goals related to ensuring that this application or tool contributes to an “ethical” future
Loosly refering the TAPIC framework from the World Health Organisation, focusing on transparency, acccountability, participation, intergrity and Capacity. The below are three goals that are built on the TAPIC framework(“TAPIC”).
1. Reduce Physical and Ecological Harm
Governance should ensure that engineered organisms using genetic circuits are designed with robust biosafety and biosecurity features such as kill switches that trigger cell death in inappropriate environments, or activation only under specific conditions such as defined temperature or oxygen saturation.
In addition, long‑term post‑deployment monitoring of surrounding environments and exposed people, coupled with clear procedures for recalls and adverse event reporting, should be required so that emerging harms can be detected and mitigated early.
2. Reduce Risk of Misuse
Governance should aim to professionalise work with genetic circuits by requiring practitioners to belong to a professional body that manages licensing, training, ongoing awareness, and a code of conduct, similar to a Hippocratic‑style commitment to beneficence and non‑malfeasance.
Manufacturers and design platforms could also be required to register with this body and to participate in systematic flagging of genetic designs that have plausible dual‑use or biological‑weapon potential, creating a structured mechanism to prevent intentional misuse.
3. Transparency and Acccountability
Governance should prioritise clear, accessible education for consumers and end‑users of products containing engineered organisms, to reduce misunderstanding, misuse, and conspiracy‑driven opposition that can undermine legitimate, beneficial applications. Legal and regulatory policies should be evidence‑based and explicitly focused on reducing physical and ecological harm, with expert‑led advisory structures to minimise the lag between innovation and oversight and to prevent premature or inappropriate deployment of genetic‑circuit applications.
3. Describe at least three different potential governance “actions” by considering the four aspects (Purpose, Design, Assumptions, Risks of Failure & “Success”)
A Mandatory Professional Board
Purpose The closest board that currently does this is European Board of Medical Genetics, but they prioritise standardising biological engineering laboratories. Multiple regional boards specifically for synthetic engineering can be developed to standardise and regulate protein designs.
Design Governmental bodies such as the NHS, MHRA, academics and researchers would all have to opt in. Research institutions such as the Research Council, could fund research in safety and regulations of synthetic biology.
Assumptions
That professionals would want have regulations on a relatively new field.
Regulations would keep up with the pace of the research
Risk of Failure or Success
Failure - knowledgable experts can opt to manufacture their designs abroad to skip legal or regulatory requirements. Too much regulations could stiffen innovation in the field
Success - knowledgable experts only work on designs that are for the betterment of humanity and design intentional and reliable bio-securities
Expert Led Legalisation
Purpose Currently the legislation is dated compared to current technologies and research. An Expert led legislation task force can consult, draft and spearhead legislation to create current guard rails within the field.
Design The most suitable experts in synthetic biology, biosecurity, environmental scientists and biology will need to be bought together.
The government would need to establish this task force alongside the regulators and legal bodies.
Assumptions
A small task force would increase and improve the legislations and not create legislation from a narrow perspective
Experts will keep up to date with technologies in the overall field, outside of their expertise.
Risk of Failure or Success
Failure - Expert bias, legislation could be geared to benefiting specific interest and funding instead of safety
Success - Unintended results maybe that funding only goes to research that aligns with the legislation, leaving little room for explorations of the field. There may also be an over standardisation of procedures, which stiffens innovation and development.
Longterm Post Deployment Observations
Purpose Currently the monitoring of engineered organisms can be short term and project specific. The proposed change would mandate schemes for all applications utilising engineered organism to observe humans bodies, workplaces and the environment long term.
Design
This will require a clear triggers for when monitoring is mandatory (any use of genetic circuits that could affect health, ecosystems, or critical infrastructure over time).
Defined institutional roles, where developers design monitoring plans, regulators set and approve standards, and health/environmental authorities collect and report data.
Supporting infrastructure, including registries linking deployments to surveillance systems and predefined protocols for incident reporting, investigation, and corrective actions such as recalls or added containment.
Assumptions
Funding will be sustained for a long period of time
Consumers would be comfortable with long term monitoring
Risk of Failure or Success
Failure - Poorly design monitoring forms/questions that do not accurately may miss key interactions
Poorly analysis of the collected data
Success - Unintended results maybe long term observations increases the cost of research and costs out SME and smaller researching companies.
May put of researchers exploring products using engineered organisms.
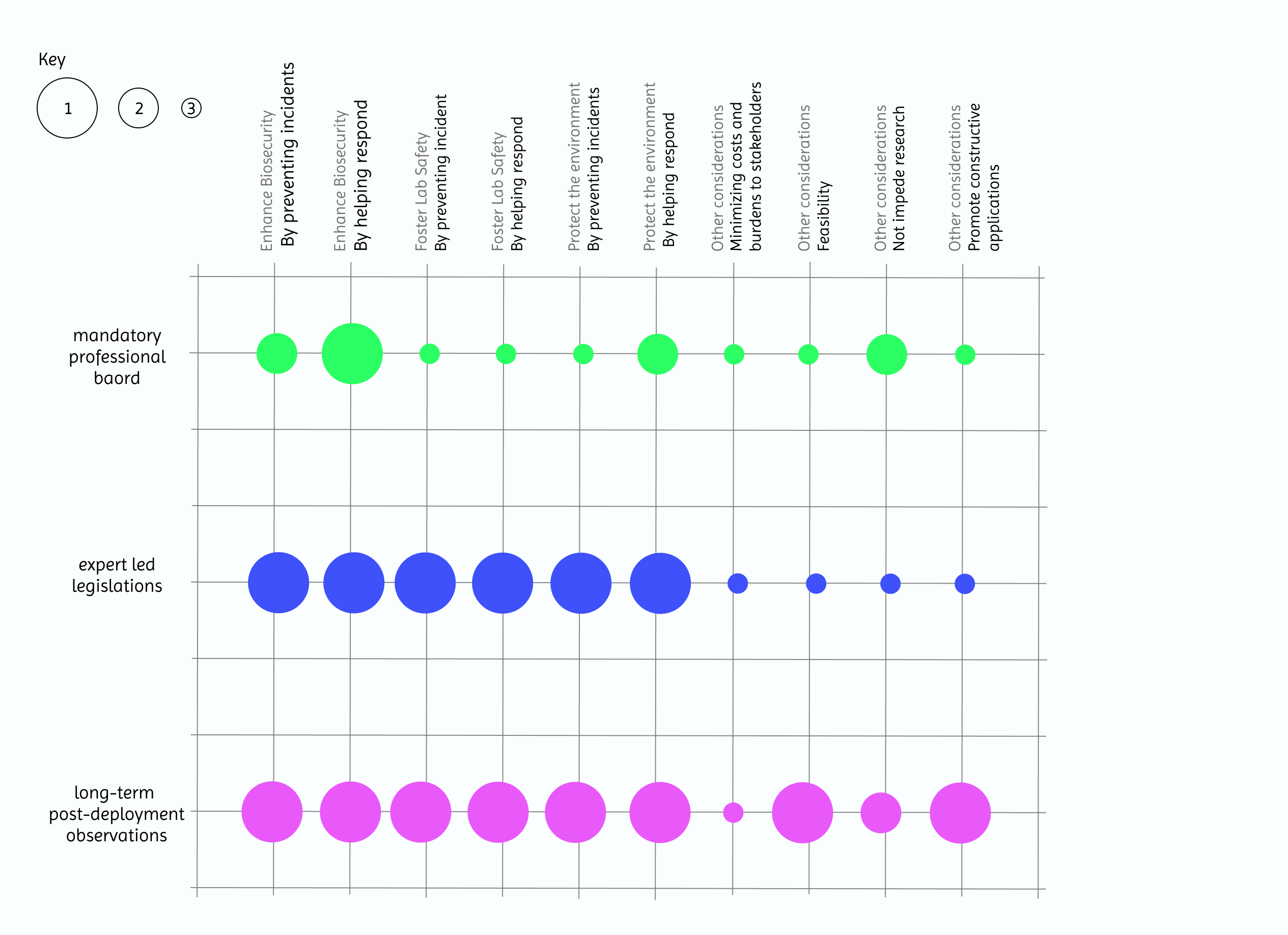
4. Score (from 1-3 with, 1 as the best, or n/a) each of your governance actions against your rubric of policy goals.
5. Drawing upon this scoring, describe which governance option, or combination of options, you would prioritize, and why
I would prioritise a combination of expert‑led legislation as the backbone, complemented by mandatory professional board. This pairing best balances safety, adaptability, and innovation by using law to set system‑wide guard rails and the board to shape day‑to‑day professional practice.
Expert‑led legislation should be the primary mechanism because it can create clear, democratically anchored guard rails that apply to all actors, including companies and international collaborators, not just individual professionals. A standing task force can iteratively update rules as genetic circuit technologies evolve, reducing the risk that regulation becomes outdated, and it provides enforcement tools such as licensing conditions and penalties that a professional board alone would lack.
A professional board then works as a complementary layer rather than the sole gatekeeper, helping to standardise training, codes of conduct, and norms of good practice that go beyond what law can specify in detail. Positioned as mandatory for certain high‑risk roles but lighter‑touch for early‑stage research, it can translate high‑level legal requirements into everyday standards without over‑burdening the entire field.
Key Trade Offs
- Legislation reaches a broader set of actors (industry, international partners, “grey zone” labs), while a board mainly governs recognised professionals.
- Law can be slower and more rigid; boards are more agile but narrower in reach.
- A strong board, if too rigid, can discourage unconventional research paths or push work abroad.
- A purely legal approach, if too heavy, can slow innovation and make investment less attractive.
- Combining them allows: laws to set minimum safety baselines, while the board supports safe experimentation within those boundaries.
- If the board is the main gatekeeper, experts might relocate or collaborate in jurisdictions without such requirements.
- If legislation is too detailed and centralised, it may lock in certain approaches and make it hard for new methods or smaller researchers to compete.
- The combination allows the law to focus on outcomes (e.g., risk thresholds, accountability) and the board on evolving best practice, reducing the pressure on either to micromanage everything.
Assumptions
Governments are willing to create and properly resource an expert task force, and to revisit legislation regularly rather than treating it as “one‑and‑done.”
The task force includes not just technical experts but also ethicists and public-interest voices, reducing the risk of narrow or captured perspectives.
Professionals in synthetic biology see value in collective standards and reputation, so they opt into (and help shape) a board rather than treating it purely as a burden.
Both mechanisms can coordinate: legal frameworks recognise the board, and the board’s standards are compatible with statutory rules.
Remaining uncertainties
It’s uncertain how much influence a national or regional board can have when work, code, and designs are easily shared across borders.
Both the task force and the board risk lagging behind rapid technical change; success depends on continuous renewal and good links to cutting‑edge research, not just one‑off appointments.
There is a risk that combined requirements (membership + compliance with evolving law) burdens small labs, start‑ups, or institutions in lower‑income settings, unless support mechanisms are built in.
Lecture Prep
Homework Questions from Professor Jacobson
- The error rate is 1:10*6 with 3.2 million base pairs in the human genome. MutS repair system
- Theres approximatly 1,036 base pairs and some reasons why all of these different codes don’t work to code for the protein of interest is because high GC content creates stable structures that can interfere with transcription and translation, Some sequences create RNase cleavage sites, repetitive sequences, extreme GC content, and secondary structures make both chemical synthesis and PCR assembly difficult and different organisms prefer certain codons over others, affecting translation efficiency.
Homework Questions from Dr. LeProust
- Phosphoramidite chemistry is the dominant method which involves: Deprotection, Base coupling, Capping and Oxidation. Each cycle takes approximately 5 minutes, and a single machine can produce roughly 0.5 Mbp per year.
- The yeild decreases, due to decoupling effects when the oligos exceed 200nt
- The yield would make it redundant and the error rate would be too high.
Homework Question from George Church
For question three answer click here
Referece
Works CitedBrophy, Jennifer A N, and Christopher A Voigt. “Principles of Genetic Circuit Design.” Nature Methods, vol. 11, no. 5, 29 Apr. 2014, pp. 508–520, https://doi.org/10.1038/nmeth.2926.
Clarice.“Inaccurate Oxygen Readings: The Problem with Pulse Oximeters.” Baylor College of Medicine Blog Network, 19 Aug. 2022, blogs.bcm.edu/2022/08/19/inaccurate-oxygen-readings-the-problem-with-pulse-oximeters/.
Fawzy, Ashraf, et al. “Racial and Ethnic Discrepancy in Pulse Oximetry and Delayed Identification of Treatment Eligibility among Patients with COVID-19.” JAMA Internal Medicine, vol. 182, no. 7, 1 July 2022, pp. 730–738, jamanetwork.com/journals/jamainternalmedicine/fullarticle/2792653#:~:text=Conclusions%20and%20Relevance%20The%20results, https://doi.org/10.1001/jamainternmed.2022.1906.
Kelle, Alexander. “Ensuring the Security of Synthetic Biology—towards a 5P Governance Strategy.” Systems and Synthetic Biology, vol. 3, no. 1-4, 10 Oct. 2009, pp. 85–90, https://doi.org/10.1007/s11693-009-9041-8.Müller, Marik M., et al.
“Genetic Circuits in Synthetic Biology: Broadening the Toolbox of Regulatory Devices.” Frontiers in Synthetic Biology, vol. 3, 7 Mar. 2025, https://doi.org/10.3389/fsybi.2025.1548572.“TAPIC.” Eurohealthobservatory.who.int, eurohealthobservatory.who.int/themes/observatory-programmes/governance/tapic.