Week 3 Lab: Lab Automation

 Image credit: HTGAA 2026
Image credit: HTGAA 2026
Lab: Opentrons Artwork
In this week’s lab, I made two custom Opentrons OT-2 protocols and used the liquid-handling robot to print Fluorescent E. coli onto black (charcoal) agar plates. I produced two pieces using different design workflows:


Design 1: Flower (Python protocol)
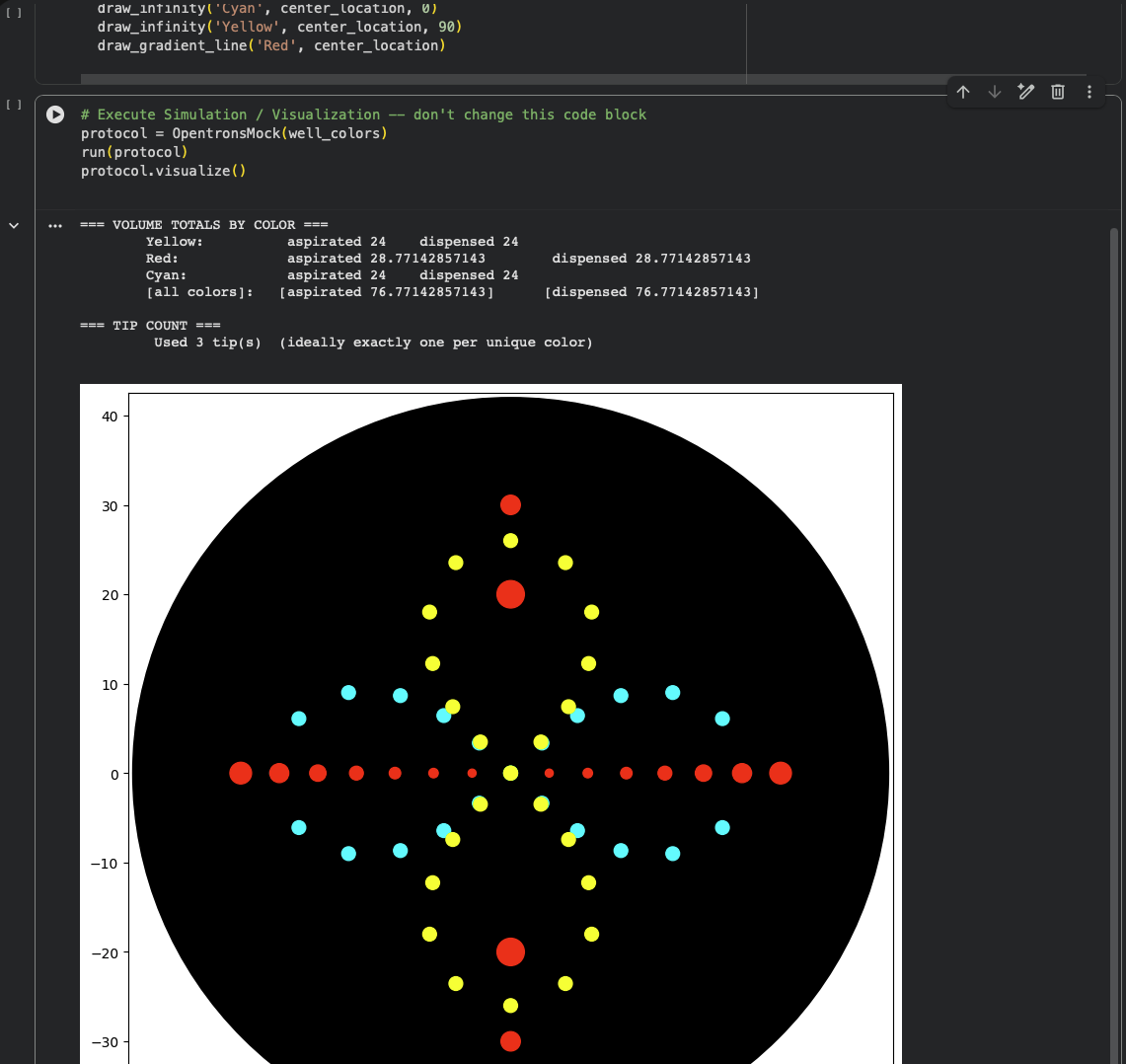
I explored how Python-defined coordinates translate into the OT-2 end-effector path, and how pipetting settings—especially volume—shape the deposited pattern and later bacterial growth. I used three fluorescent strains: Cyan (mTurquoise2), Yellow (Venus), and Red (mRFP). Cyan and Yellow followed two perpendicular ∞ (infinity) paths that together read as a flower-like form; their paths overlap at the center to test color mixing and spatial blending between strains. For Red, I mainly adjusted single-dispense volume to compare the “growth radius” suggested by the code with the actual colony spread on the plate. Two Red deposit points were placed to intersect with the Cyan/Yellow geometry to further test mixed-color effects at intersections.
Python Script:
Simulation:
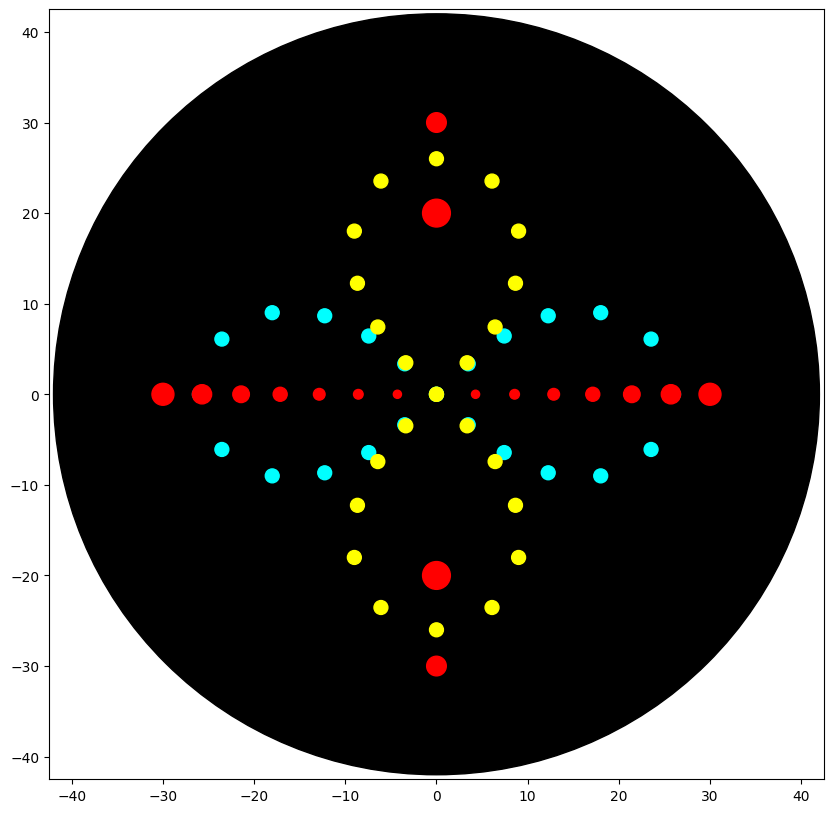

UV-illuminated Petri Dish:


Design 2: Birds (Automation Art)
To learn the Opentrons Arts pipeline, I used the Automation Art Interface: I imported an illustration, adjusted it in the web editor, published the design to the gallery, and then had the TA download and run it in the Opentrons App for automated pipetting. This workflow let me compare the constraints and benefits of a GUI-based image-to-path process versus direct protocol scripting.
Automation Art Design & Simulation:
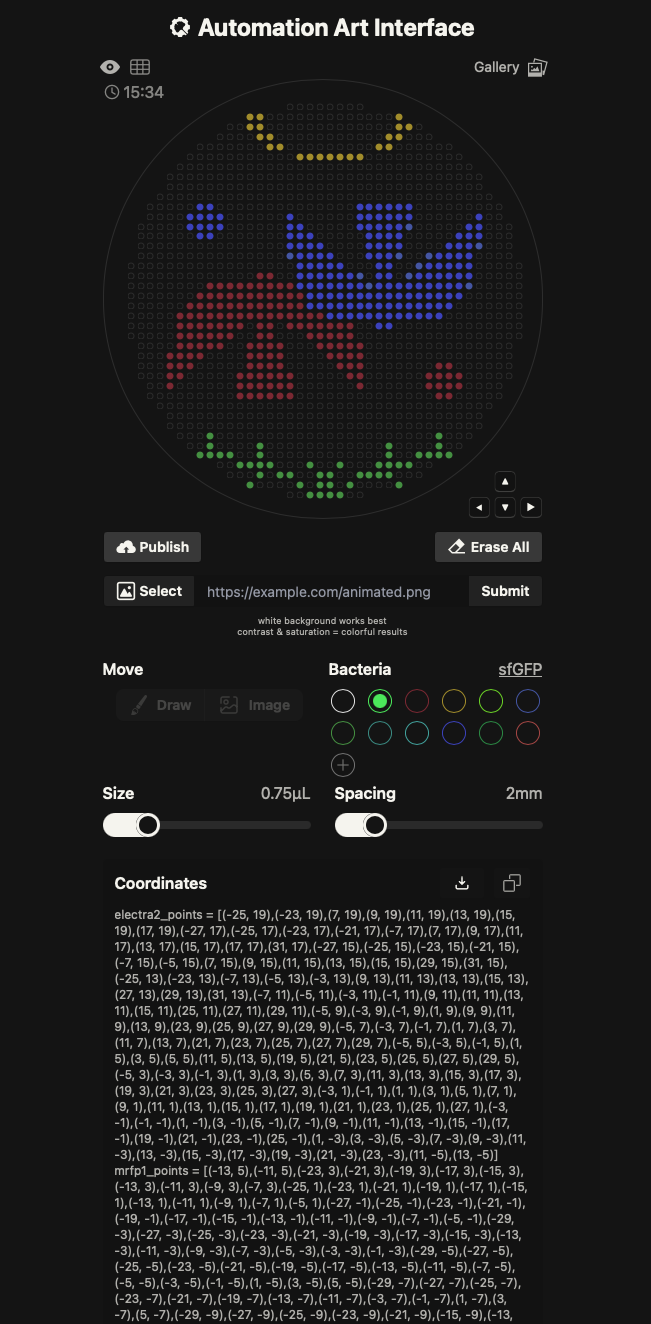

UV-illuminated Petri Dish:


Part 2: Post Lab Questions
Q1: Find and Describe
HYdrogel Dispensing method with Robotic Automation (HYDRA) for HTS-compatible hydrogels.
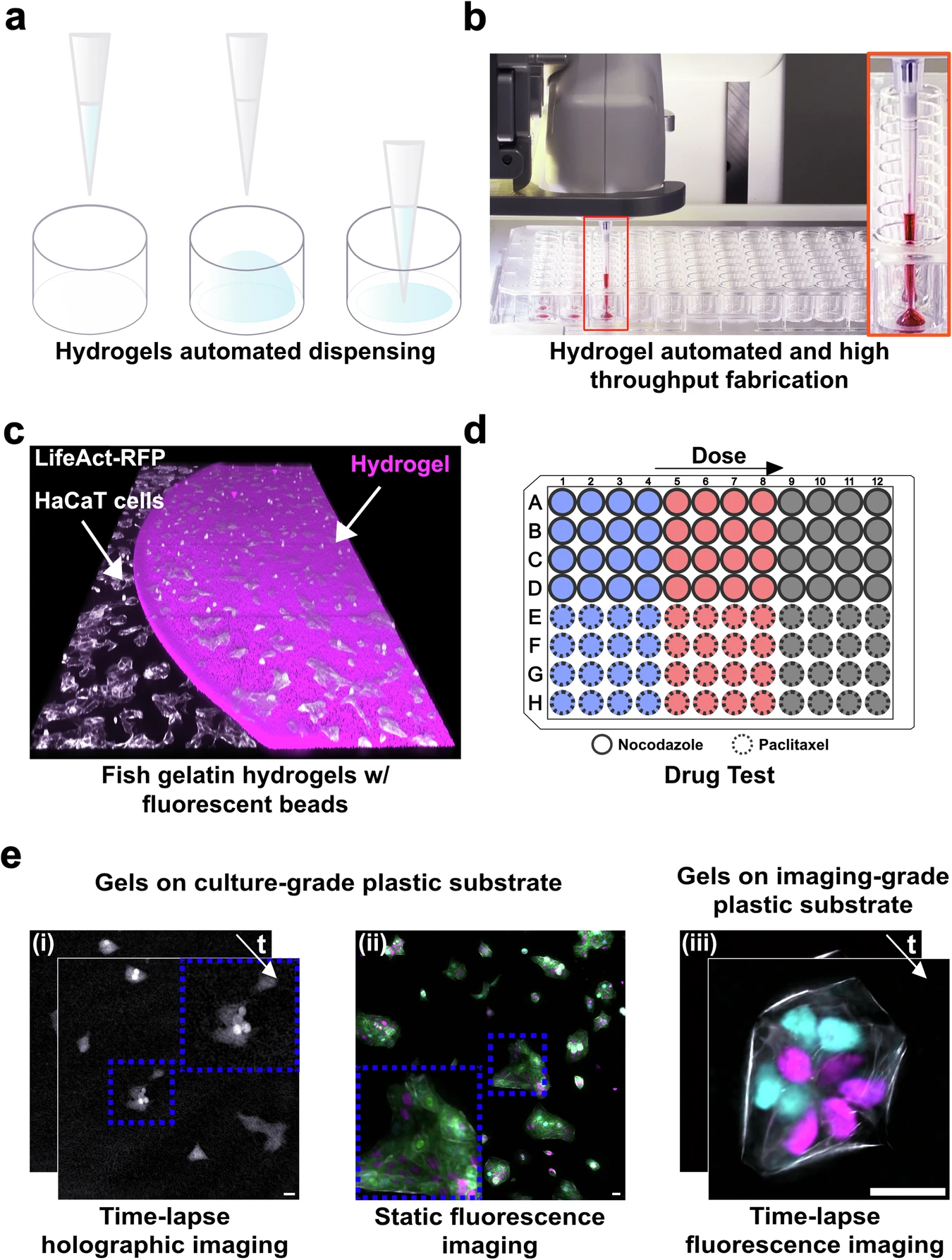
In this publication, the authors show how liquid handling automation can be used as a fabrication method, not just as a pipetting convenience. They introduce an automated workflow called HYDRA that makes thin and planar hydrogel films directly inside standard multiwell plates, so the resulting cell culture substrate is compatible with high throughput screening and imaging.
 HYdrogel Dispensing method with Robotic Automation (HYDRA) for HTS-compatible hydrogels. Torchia, E., Di Sante, M., Horda, B. et al. Fabrication of cell culture hydrogels by robotic liquid handling automation for high-throughput drug testing. Commun Eng 4, 222 (2025). https://doi.org/10.1038/s44172-025-00575-3
HYdrogel Dispensing method with Robotic Automation (HYDRA) for HTS-compatible hydrogels. Torchia, E., Di Sante, M., Horda, B. et al. Fabrication of cell culture hydrogels by robotic liquid handling automation for high-throughput drug testing. Commun Eng 4, 222 (2025). https://doi.org/10.1038/s44172-025-00575-3
Research Question
High-throughput screening (HTS) usually uses rigid plastic/glass, which reduces physiological relevance. Hydrogels improve biomimicry, but in multiwell plates they often form curved menisci that interfere with uniform seeding and microscopy, and many hydrogel approaches end up too thick for high-resolution imaging.
Lab Automation
HYDRA’s key trick is a dispense + immediate re-aspiration routine: the robot dispenses a sub-contact volume of hydrogel precursor (avoiding sidewall wetting), then re-aspirates to “pin” the contact line and leave a uniform micrometric film on the well bottom (reported ~10–50 µm). This produces meniscus-free coatings that remain compatible with standard 96- and 384-well workflows.
In their methods, the authors explicitly use an OT-2 as an “entry-level” open-source platform to demonstrate accessibility for academic labs: they implement casting via Opentrons Protocol Designer, using controlled dispense/aspirate heights and flow rates (e.g., dispensing at hundreds of microns above the bottom and aspirating closer to the surface) to reliably leave a thin residual layer that later crosslinks.
Biological Application
They fabricate fish-gelatin hydrogels crosslinked with microbial transglutaminase, then validate the platform with imaging-based dose–response assays (e.g., nocodazole and paclitaxel) on engineered epithelial cells, showing the hydrogel substrates support long-term holographic imaging and fluorescence microscopy while preserving expected pharmacological readouts.
Q2: Lab Automation Plan for Final Project
For my final project on improving extreme-environment tolerance for moss-based DNA storage, I plan to use a cloud laboratory workflow (Ginkgo Nebula) to run a high-throughput screening matrix with strong reproducibility and minimal manual pipetting.
Cloud-lab automation plan:
To systematically identify the most resilient variants and protective environments for our moss-based sensors, I utilize an automated screening workflow to test multiple tolerance-enhancement conditions in parallel:
- Precision Arraying (Echo Transfer): I will use the Echo Acoustic Liquid Handler to precisely array various tolerance-enhancement sets—including diverse pHluorin variants, protective additives (e.g., trehalose), and specific cofactors—into designated wells of 96 or 384-well plates.
- Stressor Distribution (Bravo & MultiFlo): The Bravo Automated Liquid Handling Platform and MultiFlo FX Dispenser are employed to distribute master mixes and stressor reagents (such as $H_2O_2$ for oxidative stress) across the plates, creating controlled gradients of UV, desiccation, and chemical stressors.
- Environmental Control (PlateLoc & Inheco): Plates are sealed using the PlateLoc Thermal Microplate Sealer to prevent evaporation, followed by incubation in Inheco modules. This ensures rigorous control over temperature and timing during both the stress-induction and recovery phases.
- Automated Access (XPeel): Between multi-step stages, transitioning from stress to recovery or preparing for measurement, the XPeel system automates the removal of seals, allowing for seamless integration without manual intervention.
- Performance Analysis (PHERAstar): Finally, the PHERAstar Plate Reader measures fluorescence (specifically the pH-dependent excitation shifts of pHluorin) and absorbance. This serves as a post-stress performance proxy, allowing me to rank conditions based on signal retention and metabolic recovery.